
- nature
- npj science of food
- review articles
- article
The physiological characteristics and applications of sialic acid
- Review Article
- Open access
- Published: 26 February 2025
The physiological characteristics and applications of sialic acid
npj Science of Food volume 9, Article number: 28 (2025) Cite this article
Abstract
Sialic acid (SA) is widely present at the end of the sugar chain of glycoproteins and glycolipids on the surface of animal and microbial cells and is involved in many physiological activities between microbial and host cells. Notably, these functions are attributed to the diversity of these SA types, their different transformation pathways, and their metabolic actions within the host, which are considered potential targets for affecting various diseases. However, developing disease mitigation strategies is often limited by an unclear understanding of the mechanisms of interaction of the causative agents with their hosts. This review mainly focuses on three types of SA: Neu5Ac, Neu5Gc, and KDN. The sources, main types, and distribution of these SAs are discussed in detail, emphasizing the metabolic processes of different SAs and their interaction mechanisms with the host. This review will help lay a foundation for developing functional foods and SA-targeted intervention strategies.
시알산(SA)은
동물 및 미생물 세포 표면의 글리코프로틴과 글리콜리피드 당 사슬의 말단에 널리 존재하며,
미생물과 호스트 세포 간의 다양한 생리적 활동에 관여합니다.
특히, 이러한 기능은
SA 유형의 다양성,
서로 다른 변환 경로,
호스트 내 대사 작용에 기인하며,
이는 다양한 질병에 영향을 미치는 잠재적 표적으로 간주됩니다.
그러나
질병 완화 전략을 개발하는 것은
원인체와 호스트 간의 상호작용 메커니즘에 대한 명확한 이해 부족으로 인해 종종 제한됩니다.
이 리뷰는
주로 세 가지 유형의 SA인 Neu5Ac, Neu5Gc, 및 KDN에 초점을 맞춥니다.
이 SA의 원천, 주요 유형, 분포가 상세히 논의되며,
다양한 SA의 대사 과정과 호스트와의 상호작용 메커니즘에 중점을 둡니다.
이 리뷰는 기능성 식품 개발 및 SA를 표적으로 한
개입 전략 수립을 위한 기반을 마련하는 데 기여할 것입니다.
Similar content being viewed by others
Identification of an N-acetylneuraminic acid-presenting bacteria isolated from a human microbiome
Article Open access26 February 2021
Biological function of sialic acid and sialylation in human health and disease
Article Open access30 September 2024

Sucrose-preferring gut microbes prevent host obesity by producing exopolysaccharides
Article Open access29 January 2025
Introduction
Sialic acid (SA), a crystalline reducing acid, was first isolated from bovine submaxillary mucin1. Nowadays, approximately 80 neuraminic acid derivatives have been classified2. They are widely found in animals (from echinoderms to higher animals), microorganisms, and insects, but not in higher plants2. Despite the size of the family, current studies have mainly focused on three core structural derivatives: N-acetylneuraminic acid (Neu5Ac), N-glycolylneuraminic acid (Neu5Gc), and deaminoneuraminc acid (KDN). They usually occupy the far end of the glycan chain, making them particularly susceptible to interactions with other cellular and environmental factors. Host SA is mainly derived from endogenous synthesis and exogenous intake. Some endogenous SAs are synthesized by the liver, exist in large quantities in various parts of the body (such as the brain and colon)3, and play roles in promoting host neural development, enhancing memory, and regulating immunity. Neu5Gc is hydroxylated from CMP-Neu5Ac to CMP-Neu5Gc by CMP-Neu5Ac hydroxylase (CMAH)4. However, because of mutations in CMAH, humans lose the ability to biosynthesize Neu5Gc, which is in trace amounts in healthy individuals4, except in some patients. Therefore, foods such as red meat, milk products, and bird nests have become important sources of exogenous SA in the host5,6,7.
The important role of SA in affecting host physiological and pathological processes includes information transfer between cells, cell adhesion between, regulation of the life cycle, mediation of bacterial and viral infections, and involvement in tumor growth and metastasis. Of note, the diversity of SAs determines the diversity of their biological functions in human nutrition, and they have become potential targets for ameliorating some diseases. The presence of Neu5Gc is often closely related to the occurrence and development of diseases, such as cancer8. Neu5Ac is an important source of nutrients for brain development and cognition, particularly in infants and young children9. These results indicate that different types of SA have specific effects on host health. Considering the current lack of SA safety evaluation data, a detailed understanding of the source, structure, and physiological functions of different SA types is of great significance for developing SA products.
In this review, the three most widely studied types of SA, namely Neu5Ac, Neu5Gc, and KDN, were selected as the main research objects, focusing on their distribution in the host. Their metabolic processes and physiological functions in the human body are also discussed. In addition, the contents of SA in the external environment, especially in food, are summarized to clarify the application potential of different SAs in developing functional foods. Finally, the safety evaluation, application status, and future application prospects of SAs are analyzed, to facilitate the rational utilization of SA and the development of functional food components.
소개
시알산(SA)은
결정성 환원산으로,
처음에는 소의 하악선 점액에서 분리되었습니다1.
현재 약 80종의 뉴라민산 유도체가
분류되어 있습니다2.
이들은
동물(해면동물부터 고등동물까지), 미생물, 곤충 등에서 널리 발견되지만,
고등식물에서는 발견되지 않습니다2.
가족의 규모에도 불구하고, 현재 연구는
주로 세 가지 핵심 구조 유도체에 집중되어 있습니다:
N-아세틸뉴라민산(Neu5Ac),
N-글리콜릴뉴라민산(Neu5Gc), 및
데아미노뉴라민산(KDN).
이들은
일반적으로 글리칸 사슬의 끝부분에 위치하여
다른 세포 및 환경 요인과 상호작용에 특히 취약합니다.
호스트 SA는
주로 내인성 합성과 외인성 섭취에서 유래합니다.
일부 내인성 SA는 간에서 합성되며,
신체 여러 부위(예: 뇌와 대장)에 대량으로 존재하며3,
호스트 신경 발달 촉진, 기억력 강화, 면역 조절 등에 역할을 합니다.
Neu5Gc는
CMP-Neu5Ac 수산화효소(CMAH)에 의해 CMP-Neu5Ac에서 CMP-Neu5Gc로 수산화됩니다4.
그러나
CMAH의 돌연변이로 인해
인간은 Neu5Gc를 생합성하는 능력을 상실했으며,
건강한 개인에서는 극미량으로 존재합니다4,
일부 환자를 제외하고는.
따라서 적색 육류, 유제품, 새 둥지 등은
호스트에게 외인성 SA의 중요한 공급원이 되었습니다5,6,7.
SA가 호스트의 생리적 및 병리적 과정에 미치는 중요한 역할에는
세포 간 정보 전달, 세포 간 접착, 생명 주기 조절, 세균 및 바이러스 감염 매개,
종양 성장 및 전이에의 관여 등이 포함됩니다.
특히, SA의 다양성은
인간 영양에서의 생물학적 기능 다양성을 결정하며,
일부 질환 개선을 위한 잠재적 표적으로 부상했습니다.
Neu5Gc의 존재는
암8과 같은 질환의 발생 및 진행과 밀접하게 연관되어 있습니다.
Neu5Ac는 특히 영유아의 뇌 발달과 인지 기능에 중요한 영양소 공급원입니다9.
이러한 결과는 다양한 유형의 SA가 호스트 건강에 특정 효과를 갖는다는 것을 시사합니다. 현재 SA 안전성 평가 데이터가 부족하다는 점을 고려할 때, 다양한 SA 유형의 원천, 구조, 생리적 기능을 상세히 이해하는 것은 SA 제품 개발에 큰 의미를 갖습니다.
본 리뷰에서는 가장 널리 연구된 세 가지 SA 유형인 Neu5Ac, Neu5Gc, KDN을 주요 연구 대상으로 선정했으며, 호스트 내 분포에 초점을 맞췄습니다. 또한 인체 내 대사 과정과 생리적 기능도 논의되었습니다.
또한 외부 환경, 특히 식품 내 SA의 함량을 요약하여 다양한 SA의 기능성 식품 개발 잠재력을 명확히 합니다.
마지막으로 SA의 안전성 평가, 적용 현황, 미래 적용 전망을 분석하여 SA의 합리적 활용과 기능성 식품 성분 개발을 촉진합니다.
Distribution of SA
The distribution of different SAs in nature (especially in the host’s body) is significantly different, and this is attributable to the variation in external factors, such as diet types, and the physiological and developmental state of the host. Therefore, understanding the distribution of different SAs in the host and external environment is of great significance for the rational utilization and targeted regulation of different SAs (Fig. 1).
SA의 분포
자연계(특히 숙주 체내)에서 다양한 SA의 분포는 크게 다르며, 이는 식이 유형과 같은 외부 요인의 변동 및 숙주의 생리적·발달 상태에 기인합니다. 따라서 숙주와 외부 환경에서의 다양한 SA 분포를 이해하는 것은 다양한 SA의 합리적 활용과 표적 조절에 큰 의미를 갖습니다(그림 1).
Fig. 1: The distribution of three main types of sialic acid: Neu5Ac, Neu5Gc, and KDN in the human body (left) and in foods (right).
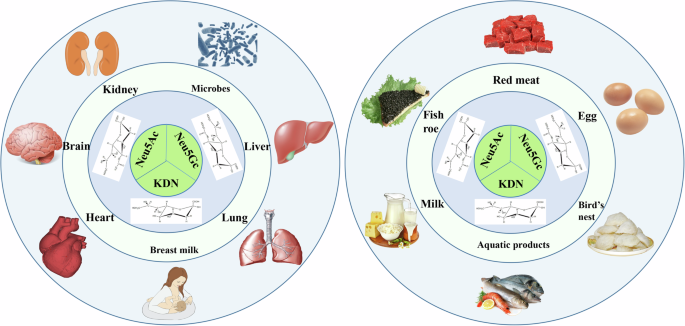
Neu5Ac N-acetylneuraminic acid, Neu5Gc N-glycolylneuraminic acid, KDN deaminoneuraminc acid, SA sialic acid. The image is created by the author and there is no copyright or conflict of interest.
Neu5Ac
Neu5Ac is widely distributed in nature, mainly as a component sugar in animal cell membranes, secreted fluid glycoproteins, glycolipids, and bacterial capsule substances. In the human body, Neu5Ac is primarily concentrated in the central nervous system, especially in the brain’s gray matter, where it combines with gangliosides and glycoproteins. It is an important structural and functional ganglioside component and correlates with brain growth and memory formation10,11. Neu5Ac could be detected in almost all tested organs and was the predominant type of SA distributed in animals12. In the gut, Neu5Ac plays a variety of biological roles at the interface of host epithelial cells and the microbiota. It is the most abundant SA type in adult and fetal mucins13. Many bacterial pathogens can mimic the host through cell surface decoration and provide structural building blocks to evade the host immune system14.
In food, SA exists mainly in a conjugated state and it binds to complex polysaccharides on the cell surface. Human milk is one of the richest sources of SA, primarily Neu5Ac. Most SA in breast milk is present in a conjugated state (oligosaccharide-conjugated), with a small amount remaining in the free state15. Edible bird nests are the main source of Neu5Ac7. In addition, it exists in large quantities in poultry eggs16, livestock meat17, and other foods, which is the most widely distributed SA type in food.
Neu5Ac
Neu5Ac는
자연에 널리 분포되어 있으며,
주로 동물 세포막의 구성 당분, 분비액 글리코프로틴, 글리콜리피드, 세균 캡슐 물질의 구성 성분으로 존재합니다.
인간 몸에서는 Neu5Ac가
주로 중추 신경계에 집중되어 있으며,
특히 뇌의 회백질에서 글리코사미노글리칸과 글리코프로틴과 결합합니다.
이는 중요한 구조적 및 기능적 글리코지드 성분으로
Neu5Ac는
거의 모든 검사된 장기에서 검출되었으며
동물에서 분포된 SA의 주요 유형이었습니다12.
장에서는
호스트 상피 세포와 미생물군집의 인터페이스에서
다양한 생물학적 역할을 수행합니다.
성인 및 태아 점액에서
가장 풍부한 SA 유형입니다13.
많은 세균성 병원체는
세포 표면 장식을 통해 호스트를 모방하고
호스트 면역 체계로부터 회피하기 위해 구조적 구성 요소를 제공합니다14.
식품에서 SA는
주로 결합된 상태로 존재하며
세포 표면의 복잡한 다당류에 결합됩니다.
인간 모유는
SA의 가장 풍부한 원천 중 하나로,
주로 Neu5Ac로 구성되어 있습니다.
모유에 존재하는 SA의 대부분은
결합 상태(올리고사카라이드 결합형)로 존재하며,
소량은 자유 상태로 남아 있습니다15.
식용 새 둥지는
Neu5Ac의 주요 원천입니다7.
또한 가금류 계란16, 가축 고기17 및 기타 식품에 대량으로 존재하며,
식품에서 가장 널리 분포된 SA 유형입니다.
Neu5Gc
Neu5Gc is present in non-human primate cells, including some extant great apes, as well as many other mammals and invertebrates18,19, and salmonid fish eggs20. Mutations in the CMAH gene cause humans to lose the ability to biosynthesize Neu5Gc, which is virtually absent in healthy individuals4. Notably, Neu5Gc accumulates in large amounts in the human body under certain pathological conditions, especially in some inflammatory and neoplastic diseases, and it may be involved in physiological responses in the human body21,22,23.
Exogenous dietary intake is considered the main source of Neu5Gc in the human body. Dietary sources rich in Neu5Gc include red meat (beef, pork, and lamb) and some milk products, it is largely absent in plants, microbes, poultry, and fish5. When healthy humans ingest foods containing Neu5Gc, sialidase produced by their intestinal flora can metabolize glycans and release free Neu5Gc from the food24. Some free Neu5Gc is absorbed and excreted in urine, and other exogenous Neu5Gc reaches lysosomes via the pinocytic/endocytic pathway. It is exported in free form to the cytosol for activation and transfer to glycoconjugates25. It is then incorporated into the surface or secreted glycans, glycoproteins, and glycolipids of cultured human cells. Current studies have proposed two possible mechanisms for the abnormal accumulation of Neu5Gc in the body. First, individual differences lead to variable susceptibility or resistance to some microbial pathogens, which then affects the function of some endogenous SA receptors in the immune system. Second, excessive dietary intake of Neu5Gc may also lead to disturbances in the SA synthesis pathway and excessive accumulation of Neu5Gc in the body, triggering antigenic reactions.
Neu5Gc
Neu5Gc는
일부 현존하는 대형 유인원을 포함한 비인간 영장류 세포,
그리고 연어과 어류 알20에 존재합니다.
CMAH 유전자의 변이는
인간이 Neu5Gc를 생합성하는 능력을 상실하게 하며,
이는 건강한 개인에서 거의 존재하지 않습니다4.
특히, Neu5Gc는
특정 병리적 조건 하에서 인간 몸에 대량으로 축적되며,
특히 일부 염증성 및 종양성 질환에서 관찰되며,
인간 몸의 생리적 반응에 관여할 수 있습니다21,22,23.
외인성 식이 섭취는
인간 몸의 Neu5Gc 주요 공급원으로 간주됩니다.
Neu5Gc가 풍부한 식이 원천에는 적
색 육류(소고기, 돼지고기, 양고기)와 일부 유제품이 포함되며,
식물, 미생물, 가금류, 어류에는 거의 존재하지 않습니다5.
건강한 인간이 Neu5Gc를 함유한 음식을 섭취할 때,
장내 미생물이 생성하는 시알리다아제는
글리칸을 대사하여 음식에서 자유로운 Neu5Gc를 방출합니다24.
일부 자유로운 Neu5Gc는
흡수되어 소변으로 배설되며,
다른 외인성 Neu5Gc는 피노시틱/엔도시틱 경로를 통해 리소좀으로 이동합니다.
이는 자유 형태로 세포질로 수출되어 활성화되고
글리코콘쥬게이트로 전달됩니다25.
이후 배양된 인간 세포의 표면이나 분비 글리칸, 글리코프로틴, 글리콜리피드에 통합됩니다.
현재 연구에서는
체내 Neu5Gc의 비정상적 축적에 대한 두 가지 가능한 메커니즘이 제안되었습니다.
첫째,
개인 차이로 인해 일부 미생물 병원체에 대한 감수성 또는 저항성이 달라지며,
이는 면역 체계 내 일부 내인성 SA 수용체의 기능에 영향을 미칩니다.
둘째,
Neu5Gc의 과도한 식이 섭취는
SA 합성 경로의 장애를 유발하고 체내 Neu5Gc의 과도한 축적을 초래해
항원 반응을 유발할 수 있습니다.
KDN
KDN, which was first identified in 1986 as an unknown deaminated SA in Salmo gairdneri (rainbow trout) egg polysialoglycoprotein, protects oligo(poly)sialyl chains from exo-sialidase26. Early studies suggested that KDN is prevalent in some lower vertebrates and bacterial capsular polysaccharides27. Studies based on some mammals have found that, although KDN has been detected in the spleen, kidney, heart, and muscle, its amount is <1% of the total SA content12.
KDN is a type of SA that is closely related to the occurrence and development of host diseases. KDN was identified in human fetal umbilical cord red blood cells and ovarian cancer cell samples and is overexpressed in ovarian tumor tissues27,28,29. Although direct evidence is lacking, these findings suggest that KDN synthesis is involved in blood cell formation during animal reproduction and embryonic development. In addition, overexpression of KDN in ovarian and prostate cancer cells suggests that it may be an oncoembryonic antigen in these tumors that may develop as an “early warning” signal for disease onset and/or a key marker of disease recurrence29.
Importantly, significant changes in KDN levels within the body are strongly influenced by dietary intake. Oral mannose intake results in significant intracellular KDN enrichment in the spleen, lungs, kidneys, and brain. These findings suggest that dietary mannose affects KDN metabolism in various organs and highlight the possibility that the targeted regulation of dietary SA content protects host health30.
KDN
KDN은 1986년 Salmo gairdneri (무지개 송어) 알 폴리시아고글리코프로틴에서 미지의 데아미네이트된 SA로 처음 식별되었으며, 올리고(폴리)시아릴 사슬을 엑소-시아리다아제로부터 보호합니다. 초기 연구는 KDN이 일부 저등 척추동물과 세균 캡슐 다당류에 널리 분포한다고 제안했습니다. 일부 포유류에 대한 연구에서는 KDN이 비장, 신장, 심장, 근육에서 검출되었지만, 그 양은 총 SA 함량의 1% 미만입니다12.
KDN은 호스트 질병의 발생과 발달과 밀접하게 관련된 SA의 한 유형입니다. KDN은 인간 태아 탯줄 적혈구와 난소 암 세포 샘플에서 검출되었으며, 난소 종양 조직에서 과발현됩니다27,28,29. 직접적인 증거는 부족하지만, 이러한 결과는 KDN 합성이 동물 생식 및 배아 발달 과정에서 혈액 세포 형성에 관여할 수 있음을 시사합니다. 또한 난소 및 전립선 암 세포에서의 KDN 과발현은 이 종양에서 질병 발병의 ‘조기 경고’ 신호로 작용하거나 질병 재발의 핵심 지표로 기능할 수 있는 '온코배아 항원'일 가능성을 제시합니다29.
중요하게도, 체내 KDN 수치의 변화는 식이 섭취에 의해 크게 영향을 받습니다. 구강으로 섭취된 만노스는 비장, 폐, 신장, 뇌에서 세포 내 KDN 농도를 유의미하게 증가시킵니다. 이러한 결과는 식이 만노스가 다양한 장기에서 KDN 대사 과정에 영향을 미친다는 것을 시사하며, 식이 SA 함량의 표적 조절이 호스트 건강을 보호할 수 있다는 가능성을 강조합니다30.
Synthesis and metabolism of SA in human
SA is internally synthesized by the liver3. The UDP-N-acetylglucosamine 2-epimerase/N-acetylmannosamine kinase is a key enzyme affecting the hepatic synthesis of Neu5Ac31. However, owing to immature liver development and the low activity of UDP-N-acetylglucosamine 2-epimerase in newborn individuals, the amount of SA synthesized may not meet the demand for SA during early brain development32. Thus, exogenous SA is necessary, such as through maternal milk, to meet the needs of normal growth and development. Another major source of SA is placental transfer to the host offspring. Since fetal mice cannot produce SA, SA in pregnant mice may enter the fetus through the placenta to ensure fetal growth and development. Thus, the placenta can regulate the balance of SA content between the mother and fetus16.
Some of the SA in the body comes from exogenous intake, and the small intestine is the main site of SA uptake. After oral administration to rats, 90% of the administered Neu5Ac is absorbed from the intestine within 4 h. After intravenous administration, 90% of the administered Neu5Ac is excreted in the urine within 10 min33. Some Neu5Ac may reach the colon, where it can be catabolized by the microbial community and the host, thereby affecting the physiological state and microbiome structure of the gut34.
인간에서의 SA의 합성 및 대사
SA는
간에서 내부적으로 합성됩니다3.
UDP-N-아세틸글루코사민 2-에피메라제/N-아세틸만노사민 키나아제는
Neu5Ac의 간 합성에 영향을 미치는 핵심 효소입니다31.
그러나
신생아의 미성숙한 간 발달과 UDP-N-아세틸글루코사민 2-에피메라제의 낮은 활성으로 인해
SA의 합성량은 초기 뇌 발달 기간 동안의 SA 수요를 충족시키지 못할 수 있습니다32.
따라서
정상적인 성장과 발달을 위해 모유를 통해 공급되는 외인성 SA가 필요합니다.
SA의 또 다른 주요 공급원은
태반을 통한 태아로의 전달입니다.
태아 쥐는 SA를 생성할 수 없기 때문에 임신한 쥐의 SA는 태반을 통해 태아로 전달되어 태아의 성장과 발달을 보장합니다. 따라서 태반은 모체와 태아 사이의 SA 함량 균형을 조절할 수 있습니다16.
체내의 일부 SA는
외인성 섭취에서 유래하며,
소장은 SA 흡수 주요 부위입니다.
쥐에게 경구 투여 시 투여된 Neu5Ac의 90%가 4시간 이내에 장에서 흡수됩니다. 정맥 내 투여 시 투여된 Neu5Ac의 90%가 10분 이내에 소변으로 배설됩니다33. 일부 Neu5Ac는 대장에 도달하여 미생물 군집과 호스트에 의해 분해될 수 있으며, 이는 장의 생리적 상태와 미생물군 구조에 영향을 미칠 수 있습니다34.
Physiological function
Maintenance of brain health
In 1981, Morgan et al. injected radiolabeled Neu5Ac into 12 day-old pups and found that 80% of the Neu5Ac was incorporated into the synaptosome after 2 h35. The same phenomenon was observed in male domestic piglets injected with Neu5Ac through the jugular vein. After 120 min, Neu5Ac was significantly enriched in the brain and its content was higher than in the liver, pancreas, heart, and spleen36. These results indicate that exogenous SA can cross the blood-brain barrier and enter various tissues, with significant enrichment in brain tissue. Previous studies have shown that 70% of conjugated SA in the brain tissue is in forming gangliosides9. As an important component of brain gangliosides and sialic glycoproteins9, SA combines with nerve cell adhesion molecules (NCAMs) to form polySA-NCAM, which complexes play important roles in synaptic plasticity and neurodevelopment9,37. Therefore, exogenous intake of SA may contribute to physiological health, and SA may be involved in the process of neural development.
SA is frequently attached at the terminal of complex oligosaccharides in glycolipids, glycoproteins, and mucin. Sialylated human milk oligosaccharides are a key component of exogenous SA to infants, which may be a conditioned nutrient during rapid brain growth, promoting infant memory and intellectual development, and improving children’s intestinal absorption capacity and immunity38. Human milk containing SA in the form of sialyllactose influenced bound SA in the prefrontal cortex and the ratio of free SA to bound SA in the hippocampus in pigs39, which is closely associated with cognitive and memory development. However, incomplete de novo SA biosynthesis leads to SA deficiency, which reduces the pool of conjugated SA precursors. For example, the newborn rat brain absorbs more SA than the adult brain, and SA supplementation improves cognitive function in adult mice40. SA supplementation in the neonatal period through exogenous sources (such as milk powder) targeted to the brain to promote ganglioside and NCAM formation is conducive to promoting appropriate neurological, cognitive, and memory development and has a great role in promoting memory improvement and intellectual development in infants16. Therefore, SAs, especially Neu5Ac, have the potential to be a component of infant formulations.
생리적 기능
뇌 건강 유지
1981년 Morgan 등 연구진은 12일령 쥐 새끼에게 방사성 표지된 Neu5Ac를 주사한 결과, 2시간 후 Neu5Ac의 80%가 시냅소체에 흡수되었다고 보고했습니다35. 동일한 현상은 경정맥을 통해 Neu5Ac를 주사한 수컷 가축 돼지 새끼에서도 관찰되었습니다. 120분 후 Neu5Ac는 뇌에서 유의미하게 풍부해졌으며, 그 함량은 간, 췌장, 심장, 비장보다 높았습니다36. 이 결과는 외인성 SA가 혈액-뇌 장벽을 통과해 다양한 조직으로 이동하며, 뇌 조직에서 유의미하게 풍부해진다는 것을 보여줍니다. 이전 연구에서 뇌 조직 내 결합된 SA의 70%가 형성 중인 글리코사미노글리칸에 존재한다는 것이 확인되었습니다9. 뇌 글리코사이드와 시알릭 글리코프로틴의 중요한 구성 요소인 SA는 신경 세포 접착 분자(NCAM)와 결합하여 polySA-NCAM 복합체를 형성하며, 이 복합체는 시냅스 가소성과 신경 발달에 중요한 역할을 합니다9,37. 따라서 외인성 SA 섭취는 생리적 건강에 기여할 수 있으며, SA는 신경 발달 과정에 관여할 수 있습니다.
SA는
글리콜리피드, 글리코프로틴, 뮤신 등의
복잡한 올리고사카라이드의 말단에 자주 결합됩니다.
시알화된 인간 모유 올리고사카라이드는
영아에게 공급되는 외인성 SA의 주요 구성 요소로,
뇌 성장의 급속한 기간 동안 조건부 영양소로 작용하여
영아의 기억력과 지적 발달을 촉진하며,
어린이의 장 흡수 능력과 면역력을 개선할 수 있습니다38.
인간 모유에 시알릴락토스 형태로 존재하는 SA는 돼지의 전전두엽 피질에 결합된 SA와 해마에서의 자유 SA 대 결합 SA 비율에 영향을 미쳤습니다39, 이는 인지 및 기억 발달과 밀접하게 연관되어 있습니다. 그러나 SA의 불완전한 신규 생합성은 SA 결핍을 초래하며, 이는 결합된 SA 전구체의 풀을 감소시킵니다. 예를 들어, 신생아 쥐의 뇌는 성인 뇌보다 더 많은 SA를 흡수하며, SA 보충은 성인 쥐의 인지 기능을 개선합니다40. 신생아기 동안 뇌를 대상으로 한 외인성 소스(예: 분유)를 통해 SA를 보충하여 글리코사미노글리칸과 NCAM 형성을 촉진하는 것은 적절한 신경학적, 인지적, 기억 발달을 촉진하며, 영아의 기억 개선과 지적 발달을 촉진하는 데 큰 역할을 합니다16.
따라서 SA,
특히 Neu5Ac는 영아용 제형의 구성 성분으로 잠재력을 가지고 있습니다.
Tumor growth signals
Abnormal sialylation of the surface of tumor cells and significant upregulation of sialotransferase activity are recognized cancer markers that are thought to be associated with aggressiveness and metastatic potential41,42,43, and abnormal SA levels have the potential to be used as a cancer biomarker44,45,46. There are several possible pathways whereby SA affects tumor growth. First, SA can be used as a recognizable receptor to mask other molecules; therefore, it can be used to mask the antigen determinant on the surface of malignant tumor cells, which can reduce their immunogenicity and increase their survival. Sialoglycans are recognized by CD33-associated Siglecs to negatively regulate the immune response, thus impairing immune surveillance47. Second, sialidase, which is an enzyme that catalyzes the removal of SA residues from glycoproteins and glycolipids, plays an important role in many biological processes by regulating cellular SA content48. For example, α2,3-sialyltransferase type I regulates ovarian cancer cell migration and peritoneal dissemination via epidermal growth factor receptor signaling49. On this basis, the use of α2, 3-sialylation inhibitors can significantly inhibit the migration and spread of tumor cells, indicating an important association between sialylation and tumor occurrence and development49. Third, SA is an important component of gangliosides, and in several types of cancer, the overexpression of gangliosides leads to the activation of cell signaling, increased cell proliferation and migration, and tumor growth50. The O-acetylation of SA residues is one of the main modifications of gangliosides and is mainly dependent on the activities of sialyl-O-acetyltransferase and sialyl-O-acetylesterase. Overexpression of O-acetylated gangliosides is a marker of some cancers51, and alterations may also affect tumor immunity and metastasis. Ganglioside synthesis is increased in human hepatocellular carcinoma stem-cell-like cells, and the inhibition of ganglioside synthesis can inhibit the proliferation and spheroid growth of hepatocellular carcinoma cells. This is partly due to the inhibition of chromosomal segregation and mitotic-progression-related regulatory genes52, suggesting targeted regulation of the synthesis of SA and its related products (such as gangliosides) is expected to be useful for treating tumors. Fig. 2
종양 성장 신호
종양 세포 표면의 비정상적인 시알릴화 및 시알로트랜스퍼레이즈 활성의 현저한 증가가 공격성과 전이 잠재력과 연관된 암 표지자로 인정받고 있습니다41,42,43, 그리고 비정상적인 SA 수준은 암 바이오마커로 활용될 잠재력을 가지고 있습니다44,45,46. SA가 종양 성장에 영향을 미치는 몇 가지 가능한 경로가 있습니다. 첫째, SA는 다른 분자를 가리는 인식 가능한 수용체로 사용될 수 있으며, 따라서 악성 종양 세포 표면의 항원 결정체를 가려 면역 원성을 감소시키고 생존율을 증가시킬 수 있습니다. 시알글리칸은 CD33과 연관된 Siglecs에 의해 인식되어 면역 반응을 음성 조절하여 면역 감시를 방해합니다47. 둘째, 글리코프로틴과 글리콜리피드에서 SA 잔기를 제거하는 효소인 시알리데이스는 세포 내 SA 함량을 조절함으로써 많은 생물학적 과정에 중요한 역할을 합니다48. 예를 들어, α2,3-시알릴트랜스퍼레이즈 유형 I은 상피 성장 인자 수용체 신호전달을 통해 난소 암 세포의 이동과 복막 확산을 조절합니다49. 이 기반에서 α2,3-시알릴화 억제제의 사용은 종양 세포의 이동과 확산을 크게 억제하며, 시알릴화와 종양 발생 및 발달 간의 중요한 연관성을 시사합니다49. 세 번째로, SA는 글리코지드의 중요한 구성 요소이며, 여러 유형의 암에서 글리코지드의 과발현은 세포 신호전달 활성화, 세포 증식 및 이동 증가, 종양 성장으로 이어집니다50. SA 잔기의 O-아세틸화는 글리코지드의 주요 변형 중 하나이며, 주로 시알릴-O-아세틸트랜스퍼레이즈와 시알릴-O-아세틸에스테라제의 활성에 의존합니다. O-아세틸화된 글리코지드의 과발현은 일부 암의 표지자입니다51, 그리고 변화는 종양 면역과 전이에도 영향을 미칠 수 있습니다. 인간 간세포암 줄기세포 유사 세포에서 글리코지드 합성이 증가하며, 글리코지드 합성을 억제하면 간세포암 세포의 증식 및 구형 성장도 억제됩니다. 이는 부분적으로 염색체 분리 및 분열 진행 관련 조절 유전자의 억제52 때문으로, SA 및 관련 제품(예: gangliosides)의 합성 조절을 표적으로 하는 것이 종양 치료에 유용할 것으로 예상됩니다. 그림 2
Fig. 2: Effects of dietary Neu5Gc on host health and disease.
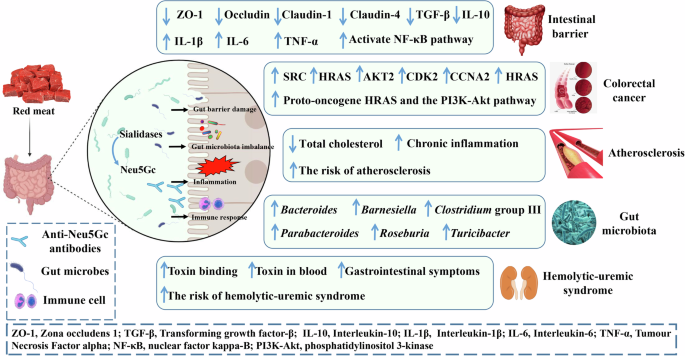
After dietary intake (such as red meat), Neu5Gc is released by sialidase produced by intestinal microbes, which further affects host health and disease. This mainly involves the following mechanisms (Fig. 2): (1) affecting intestinal barrier function, (2) regulating the structural composition of the intestinal flora, (3) inducing inflammation, and (4) inducing an immune response. The image is created by the author and there is no copyright or conflict of interest.
식이 섭취(예: 적색 육류) 후, 장내 미생물이 생성하는 시알리다아제에 의해 Neu5Gc가 방출되며, 이는 호스트의 건강과 질병에 추가적인 영향을 미칩니다. 이 과정은 주로 다음과 같은 메커니즘을 통해 이루어집니다(그림 2):
(1) 장 장벽 기능에 영향을 미치며,
(2) 장내 미생물 군집의 구조적 구성 조절,
(3) 염증 유발, 및
(4) 면역 반응 유발.
이 이미지는 저자가 제작한 것으로, 저작권 및 이해관계의 충돌이 없습니다.
Therefore, in addition to the body’s synthesis of SA, the intake of exogenous SA appears to be related to the occurrence and development of a variety of tumors. Many studies have demonstrated that excessive red meat intake may be associated with a higher risk of various digestive tract diseases, such as colon and liver cancers53,54. Long-term consumption of red meat by human-like Neu5GC-deficient mice significantly increases the incidence of cancer by five-fold, which may be attributed to the presence of Neu5Gc in red meat21. Neu5Gc in red meat can induce the production of anti-Neu5Gc antibodies. The response of the total anti-Neu5Gc antibody population to Neu5Gc antigen epitopes may be related to chronic inflammation and the occurrence of colorectal cancer55. This is caused by the interaction of metabolically accumulated dietary Neu5Gc with circulating anti-Neu5GC antibodies56. Importantly, CMAH is a key gene in the synthesis of Neu5Gc, and the recent use of CMAH gene deletion in mice has provided the possibility of further exploring the relationship between Neu5Gc, inflammation, and related tumors4. Systemic inflammatory responses occur when mice are fed a diet containing Neu5Gc, leading to a significantly higher incidence of cancer21. These experimental results may explain why diet and its specific components are closely related to susceptibility to certain diseases. Fig. 3
따라서,
신체 내 SA의 합성 외에도 외인성 SA의 섭취는
다양한 종양의 발생 및 진행과 관련이 있을 수 있습니다.
많은 연구에서 적색 육류의 과도한 섭취가
대장암 및 간암 등 다양한 소화기 질환의 위험 증가와 연관될 수 있음을 보여주었습니다53,54.
인간과 유사한 Neu5GC 결핍 마우스에서 적색 육류의 장기 섭취는 암 발생률을 5배 증가시키며,
이는 적색 육류에 존재하는 Neu5Gc 때문일 수 있습니다21.
적색 육류의 Neu5Gc는
anti-Neu5Gc 항체 생성을 유도할 수 있습니다.
Neu5Gc 항원 에피토프에 대한 전
체 anti-Neu5Gc 항체 군의 반응은 만성 염증과 대장암 발생과 관련될 수 있습니다55.
이는 대사적으로 축적된 식이 Neu5Gc와 순환하는
anti-Neu5GC 항체 간의 상호작용으로 인해 발생합니다56.
중요하게도, CMAH는 Neu5Gc 합성에 핵심적인 유전자이며, 최근 쥐에서 CMAH 유전자 삭제 기술을 적용한 연구는 Neu5Gc, 염증, 관련 종양 간의 관계를 더욱 탐구할 가능성을 제공했습니다4. 마우스에게 Neu5Gc를 함유한 식이를 공급할 경우 전신 염증 반응이 발생하며, 이는 암 발생률이 유의미하게 증가합니다21. 이러한 실험 결과는 식이와 그 특정 성분이 특정 질환의 발병 위험과 밀접하게 관련되어 있음을 설명할 수 있습니다. 그림 3
Fig. 3: SA in tumor immune evasion.
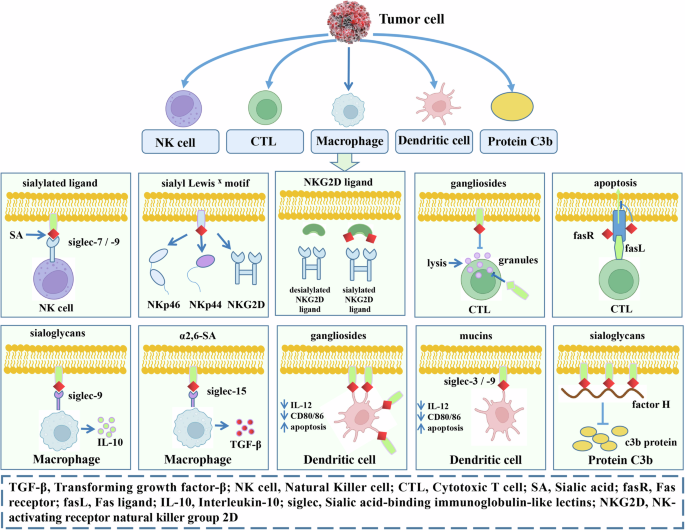
SA in cancer cells affects tumor immune evasion (Fig. 3). First, it protects them from being killed by natural killer (NK) cells. Siglecs expressed by NK cells bind to SA and inhibit its signal transduction. Sialyl Lewis X can be detected by activating the receptors NKp46, NKp44, and NKG2D, thereby inducing NK cell activation. NKG2D usually binds relative ligands, and sialylation of cancer cells reduces the NK-activating signals derived from tumor cells. Second, SA disables the main killing mechanism of cytotoxic T lymphocytes (CTLs). Hypersialylation of FasR desensitizes tumor cells to Fas-mediated cytotoxicity. CTLs expressing FasL can eliminate tumor cells that express FasR. Third, SA regulates the function of macrophages. Gangliosides influence the trafficking of lytic granules to immunological synapses and the free of lytic proteins. α2,6 SAs in some tumor cells stimulate the Siglec-15-dependent TGF-β production by monocytes nd macrophages. Expression of Siglec-9 in macrophages is capable of lowering TNF-α production and increasing IL-10 production. Fourth, SA inhibits the activation and function of dendritic cells. Tumor-bound or secreted salivary glycans regulate the activation and maturation of dendritic cells, thereby inhibiting the activation of antitumor T-cell responses. In addition, α2,6 SA-carrying mucin 2 can bind Siglec-3 and induce apoptosis of monocyte-derived dendritic cells. Fifth, a central regulatory protein factor H binds to sialoglycans on the surface of host cells, which inhibits the deposition and expansion of the complement-activating protein C3b on the cell surface and downstream activation of the complement replacement pathway. The image is created by the author and there is no copyright or conflict of interest.
In addition, KDN may act as an oncofetal antigen in these tumors, and its enhanced levels in tumor cells are closely related to human cancer pathogenesis29,57. In these studies, increased levels of KDH, along with a higher KDH: Neu5AC ratio, were considered “early warning” signals or predictive markers for detecting potentially malignant phenotypes in ovarian and other human cancers. Although relevant reports are limited at present, it is still proposed that the possible mechanism of high KDN levels in tumor cells involves hypoxia, which is a common feature of locally advanced tumors and highly invasive metastatic cancer cells. Hypoxic human cancer cultures can enhance the mRNA expression levels and enzymatic activity of Neu5Ac 9-phosphate synthetase (NPS) and phosphomannoisomerase. Elevated NPS levels promote KDN synthesis58. Based on these results, targeting SA and its related products may be an effective strategy for alleviating tumor-related diseases. For example, the use of sialidase inhibitors, such as 2, 3-didehydro-2-deoxyneuramic acid in combination with epidermal growth factor receptor inhibitors, can significantly inhibit the malignant biological behavior of tumor cells49.
Antiviral activity
SA, usually as terminal monosaccharides on different cell surfaces and as secreted glycoconjugates, can interact with bacteria and viruses to affect their function. SAs are involved in critical interactions with many different viruses (as well as other pathogens) at different stages of their infection and transmission cycles59. The first step in the viral life cycle is for virus particles to attach to the cell surface and then proliferate in the target cell. SA plays an important role in regulating virus-receptor interactions and appears to be associated with the increased virulence and infectivity of viruses60. Frierson et al. explored the role of SA binding in the pathogenesis of neuroviruses using animal models60. The results showed that SA-bound reoviruses (wild-type) showed stronger infectivity than the serotype 3 reovirus (mutant), which does not bind SA. In addition, the virus replicated in primary cultures of cortical neurons in an SA-dependent manner. Notably, neuraminidase treatment significantly reduces the infectivity and replication capacity of wild-type viruses in mice. The goblet cells and mucus are considered the main innate defense mechanism of the body61. SAs on the mucosal and cell surfaces can bind and trap viruses, preventing them from entering target tissues and removing bound virions through active processes mediated by mucociliary transport62. For example, the antiviral activity of ovomucin is closely related to its SA component, and free SA strongly promotes the binding of ovomucin to hemagglutinin (HA), thereby enhancing the interaction63. In addition to masking effects and regulating receptor interactions, SA o-acetylation of glycolipids has been shown to regulate immune cell development and activation. SA bound to the virus is involved in the maturation and activation of immune cells. O-acetylation of SA at the c9 site on B cells is necessary for the appropriate development and activation of these cells, and the 9-O-acetylation state of SA regulates the function of CD22, a Siglec that acts as a BCR signaling inhibitor in vivo64.
The acetylated form of SAs is prevalent in mammalian cells. Viruses encode SA-modifying enzymes (sialidases or esterases) that allow them to control viral replication and release with high specificity59. SAs are typically present at the endpoints of N- and O-glycoproteins, which perform important recognition responses to environmental factors, such as other cellular factors and/or pathogens. Viral sialidases have traditionally been defined as neuraminidase (NA) because they degrade Neu5Ac59. SA is the target of specific recognition by many viruses. It can mediate viral binding and cell infection, and can also act as a decoy receptor to bind virions and block viral infections59. Therefore, reducing SA levels on the cell surface by neuraminidase treatment has been shown to inhibit the entry of Middle East respiratory syndrome coronavirus into Calu-3 human airway cells. This provides direct evidence that virus-SA interactions may contribute to virion attachment65. Further studies on viral-SA interactions are required to prevent zoonotic transmission. Host-derived SAs are closely associated with the virulence of certain viruses. SA plays a distinct role in the viral infection of host cells, which may be attributed to different modifications occurring in different viruses at different stages of their infection and transmission cycles59. Given the high variability of SA in individual animal cells and tissues, determining the functional diversity and pathways of action resulting from different virus-SA carbohydrate combinations is critical for understanding the complex effects of SA on viral systems59.
Marker of inflammatory diseases
Numerous studies have demonstrated that elevated total SA levels are associated with the pathogenesis of various inflammation-related diseases, such as atherosclerosis and chronic heart failure66,67,68. Gruszewska et al. showed that chronic viral hepatitis can affect the serum total SA concentration, which may be a useful marker for distinguishing chronic hepatitis B from hepatitis C69. Therefore, serum total SA content has been considered a marker of inflammation in patients with various inflammatory diseases. Further experiments have provided direct evidence of the relationship between SA and inflammation. Dietary Neu5Gc supplementation may promote inflammation, hepatocellular carcinoma, and hemolytic uremic syndrome23. However, Neu5AC appears to inhibit high-fat diet (HFD)-induced inflammatory responses70. In addition, intravenous administration of SA exerts a similar inhibitory effect on inflammation, significantly reducing lipopolysaccharide-induced endotoxin levels, oxidative stress, alanine aminotransferase and aspartate aminotransferase levels, and associated inflammatory responses. Moreover, intravenous SA administration reverses histopathological changes in the liver71. These results highlight the potential role of SA in modulating inflammatory responses.
SA affects inflammatory diseases in several ways (Fig. 4). First, it acts as an energy source to promote the growth of pathogenic bacteria. SA derivatives located at the terminus of host glycans in the intestinal mucosa are sources of carbon and nitrogen for symbiotic and pathogenic bacteria, supporting their growth via the SA catabolic pathway. SA can also serve as a cell wall precursor that masks bacteria from host immune surveillance, further enhancing the viability and virulence of pathogenic bacteria72,73. For example, sialidase activity in the gut mediates the release of SA from intestinal tissues, thereby promoting the growth of Escherichia coli during inflammation. Excessive proliferation of E. coli may stimulate the production of pro-inflammatory cytokines by intestinal dendritic cells, thus exacerbating the inflammatory response74. Similarly, excess SA levels, induced by abnormal SA metabolism in TP53-mutated zebrafish, can be ingested by Aeromonas species, which promotes their excessive growth. Aeromonas species colonize the intestine and induce intestinal inflammation in TP53-mutated zebrafish75. These results indicate that SA may induce inflammation through the accumulation of pathogenic bacteria in a specific environment. Notably, the sialidase inhibitors oseltamivir and Philippin A are effective at blocking the increased inflammatory response caused by dysbiosis and prevent the overgrowth of sialic-utilizing pathogens by reducing the availability of free SA75. Therefore, the manipulation of SA metabolism may provide an effective treatment strategy for gut microecological disorders and inflammatory diseases caused by related pathogens. Second, SA affects host immunity. Immunoglobulin G (IgG) exhibits distinct effects on regulating the immune response (pro-inflammatory and anti-inflammatory), which may be attributed to certain residues in the sugar portion attached to the constant fragment of IgG76. SA can reduce the immune response of glioblastoma cells by increasing the expression levels of anti-inflammatory factors, such as IL-4 and IL-1077. These results indicate that SA may be directly and/or indirectly involved in regulating the immune response, which in turn affects the body’s inflammatory response. In addition, exogenous SA, as an antigen, can produce an immune reaction in the body. Since Neu5Gc is not synthesized in humans, exogenous Neu5Gc can react with circulating anti-Neu5GC antibodies78. The resulting antigen-antibody interaction may explain the association between a Neu5GC-rich diet and susceptibility to inflammation and related diseases.
Fig. 4: SA in inflammation and metabolism.
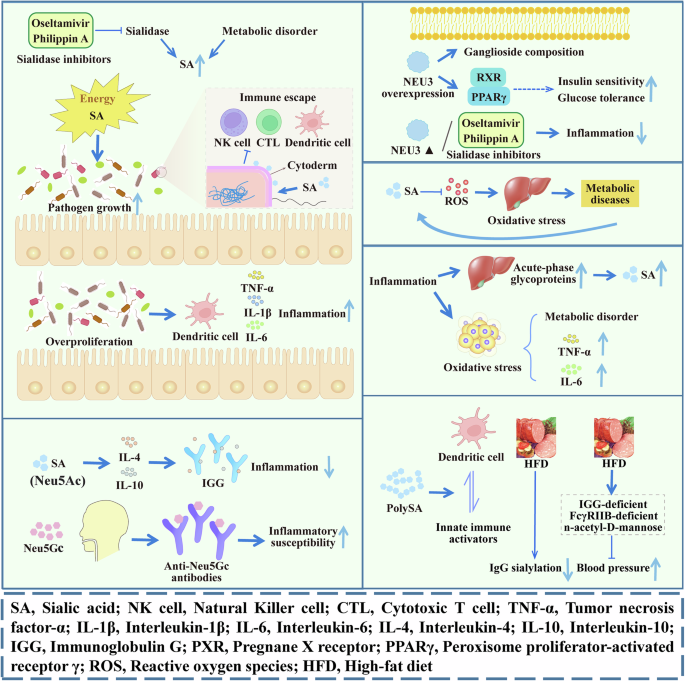
SA regulates inflammation by promoting the growth and proliferation of pathogens and regulating the immune response. SA and its related proteins regulate body metabolism through peroxidation stress, inflammation, and immunity. SA Sialic acid, NK cell Natural Killer cell, CTL Cytotoxic T cell, TNF-α Tumor necrosis factor-α, IL-1β Interleukin-1β; IL-6 Interleukin-6, IL-4 Interleukin-4, IL-10 Interleukin-10, IGG Immunoglobulin G, PXR Pregnane X receptor, PPARγ Peroxisome proliferator-activated receptor γ, ROS Reactive oxygen species, HFD High-fat diet. The image is created by the author and there is no copyright or conflict of interest.
Effects on metabolic diseases
SA and its related metabolic enzymes are important indicators of the pathophysiology of metabolic diseases. Understanding the synthesis and catabolic patterns of SA can deepen our knowledge of the pathophysiology of metabolic diseases. The liver is an important site for synthesizing and glycosylation of glycoproteins and glycolipids. The physiological state of the liver largely influences changes in the sialylation patterns of glycoproteins and glycolipids. Liver disease may affect the serum concentrations of certain carbohydrate derivatives, especially SA, attached to the ends of oligosaccharide chains69. The release of SA from the glycolipid terminal residues of cell membranes may be due to cell membrane rupture and/or lipid oxidation, and the released SA enters the bloodstream. Increased SA levels reflect changes in the structural integrity of glycolipids in cell membranes and are associated with pathological states79. On this basis, a large amount of pre-clinical and clinical trial data supports the use of serum SA content as an important indicator of metabolic diseases80,81,82.
Several possible mechanisms underlying the influence of SA on metabolic diseases have been proposed (Fig. 4), including the role of sialidase, inhibition of oxidative stress, influence of metabolism-related inflammatory responses, and immune regulation. SA is important for the function of insulin receptors83. During insulin resistance, hyperglycemia, and type 2 diabetes, increased SA synthesis in the liver and kidney results in the release of more SA into the bloodstream during cell renewal84. Correspondingly, altered levels of SA and its related enzymes can lead to altered insulin signaling85,86. For example, impaired insulin sensitivity in the skeletal muscle and liver is associated with decreased NEU3 protein abundance87, and NEU3 overexpression in the liver improves insulin sensitivity and glucose tolerance by altering ganglioside composition and peroxisome proliferator-activated receptor gamma signaling85. Mice lacking NEU3 have reduced HFD-induced adipose tissue accumulation and liver inflammation compared to normal mice, and the injection of sialidase inhibitors can achieve similar effects88. These data suggest that sialidase regulates fat and liver tissue inflammation and that sialidase inhibitors are potential therapies for inflammation caused by an HFD.
Oxidative stress is one possible mechanism through which serum SA levels influence metabolic disease. While SA is located mainly at the outermost end of the glycan chain of all cell types, released SA acts as an active oxygen scavenger89 to reduce oxidative stress. Increasing evidence supports the central role of inflammatory factors in the pathogenesis of metabolic diseases. An HFD leads to excessive lipid storage in adipocytes, the production of stress signals, and the disturbance of metabolic function, which induces inflammatory responses90. IgG sialylation is reduced in HFD-fed mice, and transferring it to IGG-deficient mice results in increased blood pressure, while no hypertensive response occurred in mice lacking FcγRIIB. Supplementation with the SA precursor n-acetyl-D-mannose inhibits obesity-related hypertension induced by an HFD in mice91, suggesting that SA influences metabolic diseases by modulating immunity.
Regulation of the gut microbiota
SA may be utilized by sialidases encoded by intestinal microbial genes, thus regulating the structure of the intestinal flora. Sialidase cleaves these terminal residues from the sialic-binding substrate that can be used as a metabolic substrate for intestinal bacteria adapted to the mucosal environment. Increased sialidase activity in the gut mediates the release of SA from intestinal tissues, which is then utilized by intestinal microbes. Some native microbes such as Bifidobacterium, a symbiotic organism in the gut, can degrade milk gangliosides and release SA, thus playing a probiotic role in the gut92.
SA intake also has a regulatory effect on intestinal microbes. Under homeostatic conditions in the gut, SA binds to mucus. A small amount of free SA is present in the intestinal lumen, which is then degraded by symbiotic bacteria that can degrade polysaccharides. In Cmah−/− mice, which cannot synthesize Neu5Gc, a Neu5Gc-rich diet induces significant changes in the gut microbiota, enriching Clostridiales and Bacteroidales24. Ma et al. found that oral SA administration increased the relative weight of the liver and decreased serum aspartate aminotransferase activity34. It also regulated the relative abundances of the intestinal flora. However, in some special cases (such as an imbalance of intestinal flora caused by antibiotics), the abundance of symbiotic bacteria that degrade polysaccharides decreases and this promotes the production of free SA by increasing sialidase activity, resulting in the proliferation and increased virulence of a large number of opportunistic bacteria in the gut (such as E. coli). This further promotes mucosal layer degradation and intestinal barrier breakdown93. These results not only demonstrate the close relationship between SA, the gut microbiota, and host health but also emphasize that the effects of SA are influenced by the ecological environment of the gut.
The diversity of glycans in complex multicellular organisms is driven by endogenous and exogenous evolutionary selection. Interactions with cells within the body may affect host health. SAs, such as Neu5Ac and Neu5Gc, can be transported to the Golgi, where they can be used as donors of newly synthesized glycoproteins and glycolipids to be delivered to the cell surface as part of the glycosylation of the cell surface of gut microbes94. These glycoconjugates play important roles in a wide range of cell-cell and cell-molecule recognition processes95,96. SA can serve as a surface decoration or attachment site for human pathogens, enabling microbial sialylation to evade the immune system97,98,99. The influence of SA on microorganisms is manifested through its involvement in formatting bacterial surface capsules. Some studies have shown that Neu5Ac can be incorporated as a terminal non-reducing sugar into lipooligosaccharides, which are the main components of the outer membrane of Haemophilus influenzae, and may play important roles in microbial virulence and pathogenicity100.
Based on the findings of previous studies, the acquisition of SA by bacteria depends on several main pathways, including de novo biosynthesis and environmental sources73,97. Neu5Ac aldolase (NanA) and NeuAc synthetase (NeuB) are important enzymes involved in Neu5Ac synthesis. NeuB then catalyzes the condensation of ManNAc 2 with phosphoenolpyruvate to form SA101. NanA catalyzes the production of Neu5Ac from ManNAc and pyruvate102. These results suggest that the ability of microorganisms to synthesize SA de novo is due to specific genes possessed by the microorganisms themselves, and also the influence of SA on microbial physiological functions, such as colonization ability103. Bacteria have evolved multiple ways to obtain SA from the environment73,97. Many bacteria produce sialidases, which remove SA residues from complex glycan structures. Normally, SA levels in the gastrointestinal tract are extremely low. However, the free SA content in the environment may be increased by intestinal dysbiosis or mucosal inflammation, due to increased expression levels of associated sialidases. For example, the human pathogen H. influenzae uses lipopolysaccharide sialylation to evade the host’s innate immune response. However, H. influenzae lacks the gene for the de novo synthesis of SA; therefore, it uses exogenous SA to modify lipopolysaccharides and relies on their respective transporters to protect lipopolysaccharide sialation100,104,105,106.
Besides, host-derived SA influences the viability and virulence of pathogenic microorganisms under certain circumstances. SA can be released by sialidases and then taken up by certain bacteria and used to decorate their surfaces or by microorganisms as a source of nutrients. For example, the results of in vitro and in vivo experiments have shown that free SA induces the expression of neuraminidase genes and toxicity regulators, and enhances the colonization and invasion capacity of Streptococcus pneumoniae in the nasopharynx107. This result suggests that SA is a signaling factor that may lead to fundamental changes in the physiological characteristics of bacteria, thereby enhancing their ability to attach to surfaces and/or survive in biofilm environments. Therefore, it is possible to target host SA, and the virus-binding region to resist viral infection.
Other functions
Serum SA levels are elevated in patients with hypothyroidism, and there is a correlation between SA levels and atherosclerotic indicators108. A significant association has been observed between elevated plasma Neu5Ac levels and an increased risk of poor clinical outcomes in patients with heart failure109. Total serum SA levels in renal dialysis patients are closely correlated with C-reactive protein levels110. There is a correlation between serum SA levels, high-sensitivity C-reactive protein levels, and blood pressure in patients with prehypertension111. Based on the findings of the abovementioned studies, SA may be a superior marker to C-reactive protein for assessing the occurrence and development of many diseases; however, direct evidence is still lacking. Presently, serum SA levels of patients with different diseases are mostly determined for common types of SA (such as total SA or Neu5Ac), but few studies have explored the relationship between the levels of other types of SA and diseases. Importantly, different SAs play different roles in various physiological environments.
Many questions remain regarding the link between Neu5Gc and host health and disease. Accumulating evidence suggests that excess Neu5Gc intake is closely associated with the occurrence of tumors and other diseases. Paradoxically, the absence of Neu5Gc has adverse health effects. Loss of Neu5Gc synthesis in CMAH-deficient mice is associated with delayed wound healing and age-dependent hearing loss4. These results suggest that adequate Neu5Gc levels play an important role in maintaining physiological health, whereas excessive intake of Neu5Gc may pose health risks.
Dietary SA supplementation, safety evaluation, and application prospects
Neu5Ac mainly exerts protective effects on brain health and metabolic diseases. Further experiments have reinforced the great potential of Neu5Ac, but not Neu5Gc or KDN, in developing functional products. Thus, it is important to determine whether long-term high-dose SA intake has negative effects on the body. Notably, the existing research mainly focuses on animal experiments, and few studies have examined its effects on humans. For example, Choi et al. conducted a subchronic dietary toxicity study of Neu5Ac in Sprague-Dawley rats before the intrauterine stage to assess the in vivo genotoxicity and mutagenicity of Neu5Ac112. The results showed that when the maternal diet contained 2% Neu5Ac, no maternal toxicity or adverse effects on offspring development were observed. In vitro, genotoxicity and mutagenicity tests have also supported the notion that SA is non-genotoxic. These results support the use of Neu5Ac in infant formulas and related foods. Besides, red-meat/Neu5Gc-mediated increased risk for atherosclerosis, which can be mitigated by a diet rich in Neu5Ac113 in the human-like Cmah-null background. However, unlike humans, who cannot synthesize Neu5Gc, Sprague-Dawley rats can synthesize SA, and both Neu5Ac and Neu5Gc in the organs are significantly increased shortly after birth, indicating that individual experimental animals do not fully mimic the physiological characteristics of the human body114. The safety evaluation of SA needs to be further clarified in clinical studies. Therefore, further clinical studies are warranted to evaluate the safety of Neu5Ac as a food additive. However, there are currently no safety evaluation results for Neu5Gc or KDN.
Considering the great potential of SA in maintaining host health, it has been used in drug development. For example, Neu5Ac has been used to synthesize several antiviral drugs, such as zanamivir, which has been recognized as effective in treating and preventing infections, it can be used to prevent and treat the infections of both influenza types A and B115, leading to increased global demand for Neu5Ac. Notably, SA content has also been used as an important indicator of pathology and as a target for intervention notably in cardiovascular disease, diabetes, and malignancy116, given the close association of SA content with individuals in different diseases. A 32 month cohort study found that serum total SA was independently associated with hs-CRP, lipoprotein, intima-media wall thickness, and wall-to-lumen ratio in hemodialysis patients. Increased serum total SA concentrations were associated with more CV events66. To sum up, SA has shown a relatively huge application prospect in the field of food and medicine.
The European Commission approved Neu5Ac for use as a food additive in the market at the prescribed amounts. Notably, in food such as edible bird’s nest, egg yolk peptide, CGMP, etc., SA mainly contains conjugate structure rather than monomer. Approximately 14%–33% of human milk oligosaccharides (HMOs) are cross-linked with Neu5Ac. For example, 3′-sialyllactose (3′-SL) and 6′-sialyllactose (6′-SL), two simplest sialylated HMOs, incorporate Neu5Ac residue with galactose molecules of lactose via α2,6- and α2,3-linkages, respectively117.
Future prospects
Different SAs show variable effects on host health, and previous studies have mainly focused on Neu5Ac, Neu5Gc, and KDN. Based on previous reports, Neu5Ac has great application prospects owing to its significant probiotic functions. However, some issues remain unaddressed. First, although a large number of preclinical studies have demonstrated the probiotic roles of Neu5Ac, there is a lack of sufficient clinical and safety evaluation data to support this viewpoint. Therefore, it is necessary to strengthen data regarding the application and safety of Neu5Ac in larger clinical samples. In addition, attention should be paid to the addition ratio of Neu5Ac in food (such as infant formula), so that the content and proportion of milk powder are more similar to the composition of breast milk. Second, the requirements for and types of SA vary at different growth stages. Therefore, a safe dosage of Neu5Ac for different individuals and the appropriate age groups should be determined. An excess and/or lack of SA can have adverse effects on host development. Third, the function of different types of SA is affected by the external environment, such as the physiological state of the host (health or disease), which may cause changes in the way SA is absorbed, metabolized, and transported by the body, resulting in different physiological effects. Further analysis is needed to determine the pathways by which different types of SA are absorbed and transported to healthy and diseased tissues to develop appropriate treatments that target SA regulation.
Notably, at present, multi-omics (especially glycomics) and other technologies can be used to quantitatively detect changes in the content of different SAs and their related synthetic precursors. Combined with data from human physiological and biochemical index databases, artificial intelligence, and other technologies to assess the regulation of SA content in the host, a more accurate SA supplement type and dose can be formulated for the test population.
References