Open AccessReview
Sialic Acids in Health and Disease
by
Gerardo N. Guerrero-Flores
1,2

,
Fayth M. Butler
3,4,
Veronica L. Martinez Marignac
5,
Guangyu Zhang
3
1
Interdisciplinary Center for Research in Health and Behavioral Sciences, School of Medicine, Universidad Adventista del Plata, Libertador San Martín 3103, Argentina
2
Faculty of Medical Sciences, Universidad Nacional de Rosario (UNR), Rosario 3100, Argentina
3
Division of Biochemistry, Department of Basic Sciences, School of Medicine, Loma Linda University, Loma Linda, CA 92350, USA
4
Center for Nutrition, Healthy Lifestyles, and Disease Prevention, School of Public Health, Loma Linda University, Loma Linda, CA 92350, USA
5
Interdisciplinary Laboratory of Biology and Molecular Genetics (IBIOGEM-CONICET), Diamante 3105, Argentina
6
Department of Earth and Biological Sciences, School of Medicine, Loma Linda University, Loma Linda, CA 92350, USA
*
Author to whom correspondence should be addressed.
Biologics 2025, 5(2), 10; https://doi.org/10.3390/biologics5020010
Submission received: 19 December 2024 / Revised: 4 February 2025 / Accepted: 7 March 2025 / Published: 26 March 2025
Downloadkeyboard_arrow_down
Abstract
Vertebrate cell surfaces exhibit intricate arrangements of glycosaminoglycan polymers, which are primarily linked to lipids and proteins. Numerous soluble secreted proteins are also decorated with either individual sugar molecules or their polymers. The carbohydrate polymers commonly possess terminal nine-carbon sugars, known as sialic acids. Due to their widespread distribution and strategic positioning, sialic acids play a crucial role in mediating and regulating a wide range of physiologic processes and pathologic conditions. Human- or animal-based investigations predominantly concentrate on the effects of sialic acids during infections, inflammations, vascular disorders, or cancers. Further investigations encompass a variety of applications, including cell–cell interactions, signaling, host–pathogen interactions, and other biological functions associated with nutrition, metabolism, or genetic disorders. Nevertheless, future mechanistic investigations are needed to clarify the specific roles of sialic acids in these varied contexts, so that more effective interventions may be developed.
척추동물 세포 표면에는
주로 지질과 단백질에 결합된 글리코사미노글리칸 폴리머의 복잡한 배열이 존재합니다.
수많은 용해성 분비 단백질은
개별 당 분자나 그 중합체로 장식되어 있습니다.
탄수화물 중합체는
일반적으로 말단 9탄당인 시알산으로 알려져 있습니다.
시알산은 광범위한 분포와 전략적인 위치로 인해
다양한 생리적 과정과 병리적 상태의 매개 및 조절에 중요한 역할을 합니다.
인간 또는 동물 기반 연구는
주로 감염, 염증, 혈관 질환, 암 등에서의 시알산의 영향을 중심으로 진행되어 왔습니다.
추가 연구는
세포 간 상호작용, 신호전달, 호스트-병원체 상호작용, 영양, 대사, 유전적 장애와 관련된 생물학적 기능 등
다양한 응용 분야를 포함합니다.
그러나 이러한 다양한 맥락에서
시알산의 구체적인 역할을 명확히 하기 위해 미래의 메커니즘 연구가 필요하며,
이를 통해 더 효과적인 개입 방법이 개발될 수 있을 것입니다.
Keywords:
sialic acid; sialoglycoconjugate; sialidase; sialyltransferase; sialylation; desialylation
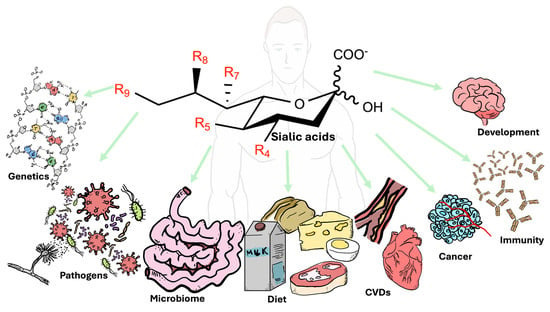
Graphical Abstract
1. Introduction
Cell surfaces are covered by a glycocalyx [1] composed of complex carbohydrate polymers (also known as glycans) anchored to cell membranes via glycoprotein or glycolipid linkages [2,3,4,5]. Biosynthetically derived from neuraminic acid [6,7], sialic acids represent a family of nine-carbon keto-aldonic acids (nonulosonic acids (NulOs)) [8]. These normally occupy the terminal positions of cell surface glycosaminoglycan moieties [9,10]. They are, however, also present freely in various biofluids or as modifiers of soluble secreted proteins [11,12,13]. Sialic acids are found in a wide range of animal phyla, including Platyhelminthes [14], Echinodermata [15], Cephalopoda [16], Crustacea [17], Cephalochordata [18], and Vertebrata [19]. In addition, sialic acids are also known to be associated with viruses, bacteria, protozoa, and fungi [20,21,22]. Consequently, it is not surprising that sialic acids are involved in a large variety of physiologic functions, such as scavenging of reactive oxygen species (ROS) [23], cell–cell interactions, receptor-binding, and cell signaling [9,13,21]. Further, sialic acids are also recognized to play a central role in some pathologic processes, like cardiovascular disorders and cancer [24,25,26].
1. 서론
세포 표면은 글리코칼릭스 [1]로 덮여 있으며,
이는 글리코프로틴 또는 글리콜리피드 결합을 통해
세포막에 고정된 복잡한 탄수화물 폴리머(글리칸으로도 알려져 있음)로 구성되어 있습니다 [2,3,4,5].
9탄소 케토-알도닉 산(비울루소닉 산(NulOs))으로 구성된 산물군입니다[8].
이들은 일반적으로
세포 표면 글리코사미노글리칸 단위의 말단 위치에 존재합니다[9,10].
그러나
다양한 생체액에 자유롭게 존재하거나
용해성 분비 단백질의 조절제로도 작용합니다[11,12,13].
시알산은
플라티헬민테스 [14], 에키노데르마타 [15], 세팔로포다 [16], 크루스타세아 [17], 세팔로코르다타 [18], 및 척추동물 [19]을 포함한
다양한 동물 문에서 발견됩니다.
또한
시알산은
바이러스, 세균, 원생동물, 곰팡이와 연관되어 있다는 것이 알려져 있습니다 [20,21,22].
따라서
시알산이 활성산소종(ROS) 제거 [23],
세포 간 상호작용,
수용체 결합,
다양한 생리적 기능에 관여한다는 것은 놀라운 일이 아닙니다.
Consequently, it is not surprising that sialic acids are involved in a large variety of physiologic functions,
such as scavenging of reactive oxygen species (ROS) [23],
cell–cell interactions,
receptor-binding, and
cell signaling
또한, 시알산은
심혈관 질환과 암과 같은 일부 병리적 과정에서도
중심적인 역할을 하는 것으로 알려져 있습니다 [24,25,26].
1.1. Analysis of Sialic Acids
Analytical approaches to characterizing sialic acids include separation techniques, such as chromatography [27] and capillary electrophoresis [28], and detection methods such as colorimetry [29] and spectrophotometry [30]. Colorimetry and capillary electrophoresis are relatively straightforward to operate, but tend to have lower sensitivity and specificity compared to other methods [31]. High-performance liquid chromatography (HPLC) is commonly used for the separation of sialic acids [31,32], and is generally combined with ultraviolet [33] or fluorescence detectors [27,34]. Currently, the study of sialic acids is greatly facilitated by the application of mass spectrometric techniques for specific detection and quantification [35,36,37,38,39].
Mass spectrometry can provide qualitative (structural) and quantitative (molecular mass or concentration) information about specific molecules of interest [40]. Mass spectrometers comprise four main components [41,42]: (1) an ion source, enabling ionization of sample molecules; (2) a mass analyzer, which separates ions according to their mass-to-charge ratio (m/z); (3) a detector, which measures the separated ion abundances in the form of electrical signals, and (4) a recording device, which transforms the detector signal into a format suitable for further analysis and processing. The process itself unfolds in its respective stages. (a) The vaporization and ionization of the sample [43,44]. Various approaches may be used for ionization in the gas phase, including electron ionization, chemical ionization, electrospray ionization (ESI), photoionization, inductively coupled plasma, and matrix-assisted laser desorption ionization [45]. (b) Acceleration of ions through an electric field to reach the same kinetic energy [46]. (c) Separation and sorting of ions according to their specific m/z ratios [40,47]. (d) Fragmentation of ions to generate fragments, the distribution of which is characteristic for each molecule. This is typically accomplished in tandem mass spectrometry. (e) Detection and recording of ions, based on the ion abundance and relative intensity [43,45].
Technical advantages of mass spectrometry include high sensitivity, specific selectivity, and small sample requirements [35,48]. Consequently, mass spectrometry has become the preferred approach for sialic acid research [49]. As a result of continuing advancements, specific sialic acids are now being studied in a variety of organisms and human tissues. Moreover, their potential roles in human health and disease are also being investigated.
1.1. 시알산의 분석
시알산 분석을 위한 분석적 접근법에는
크로마토그래피[27]와 카피라리 전기영동[28]과 같은 분리 기술,
그리고 분광광도법[29]과 분광광도계[30]와 같은 검출 방법이 포함됩니다.
분광광도법과 카피라리 전기영동은
상대적으로 조작이 간편하지만,
다른 방법에 비해 감도와 특이도가 낮은 경향이 있습니다[31].
고성능 액체 크로마토그래피(HPLC)는
일반적으로 자외선[33] 또는 형광 검출기[27,34]와 결합됩니다.
현재 시알산 연구는
특정 검출 및 정량화를 위한 질량 분석 기술의 적용으로 크게 촉진되고 있습니다[35,36,37,38,39].
질량 분석법은
관심 분자의 정성적(구조적) 및 정량적(분자량 또는 농도) 정보를 제공할 수 있습니다 [40].
질량 분석기는
(1) 이온화 소스, 시료 분자의 이온화를 가능하게 합니다;
(2) 질량 분석기, 이온을 질량 대 전하 비율(m/z)에 따라 분리합니다;
(3) 분리된 이온의 농도를 전기 신호 형태로 측정하는 검출기, 및
(4) 검출기 신호를 추가 분석 및 처리 가능한 형식으로 변환하는 기록 장치.
이 과정은 각각의 단계에서 진행됩니다.
(a) 시료의 증발 및 이온화 [43,44]. 가스 상에서 이온화에는 전자 이온화, 화학 이온화, 전기분사 이온화(ESI), 광이온화, 유도 결합 플라즈마, 매트릭스 보조 레이저 탈착 이온화 등이 사용될 수 있습니다 [45].
(b) 전기장 통해 이온을 가속시켜 동일한 운동 에너지를 갖도록 하는 단계 [46].
(c) 이온의 특정 m/z 비율에 따라 분리 및 분류 [40,47].
(d) 이온의 분열로 조각을 생성하며, 이 조각의 분포는 각 분자에 특유합니다.
이는 일반적으로 tandem mass spectrometry에서 수행됩니다. (
e) 이온의 검출 및 기록, 이온의 농도와 상대 강도에 기반합니다 [43,45].
질량 분석법의 기술적 장점에는
높은 감도, 특정 선택성, 작은 시료 요구량이 포함됩니다 [35,48].
따라서 질량 분석법은 시알산 연구에서 선호되는 접근 방법으로 자리 잡았습니다 [49].
지속적인 기술 발전으로 인해 특정 시알산은
다양한 생물체와 인간 조직에서 연구되고 있으며,
인간 건강과 질병에서의 잠재적 역할도 조사되고 있습니다.
1.2. Structures of Sialic Acids
Sialic acids constitute a family of more than 50 distinct molecules, 15 of which have been identified in human tissues [9,24]. The differences between sialic acids are based on particular N-linked or O-linked modifying groups, such as N-acetyl-, N-glycolyl, and O-acetyl, or, less frequently, the O-lactyl, O-methyl, and O-sulfate groups (Figure 1) [10,25,50]. The modifications confer distinct properties to the nine-carbon sugars, which support their specific physiologic functions [12]. Two major structural features include an often-modified amino group at position 5 and a carboxyl group at position 1. This carboxyl group, with its characteristic pKa of about 2.2, confers a negative charge on the sialic acid within a wide physiologic pH range [10,13]. Esterification of the hydroxyl groups at positions 7, 8, or 9 with acetic acid, in the form of O-acetylation, occurs less frequently [10]. Other known modifications include sulfation or methylation at position 8, or lactylation at position 9. Substitutions may occur at multiple sites, as illustrated by 7,8,9-tri-O-acetyl-N-acetyl- and N-glycolylneuraminic acid [8,9,51].
1.2. 시알산의 구조
시알산은 50개 이상의 서로 다른 분자로 구성된 가족으로,
이 중 15개가 인간 조직에서 식별되었습니다 [9,24].
시알산의 차이는
N-결합 또는 O-결합 수식 그룹에 따라 구분됩니다.
예를 들어 N-아세틸, N-글리콜릴, O-아세틸 그룹이 있으며,
덜 흔하게는 O-락틸, O-메틸, O-황산 그룹이 있습니다(그림 1) [10,25,50].
이러한 수정은
9탄당에 독특한 특성을 부여하여
특정 생리적 기능을 지원합니다 [12].
두 가지 주요 구조적 특징은
5위 위치의 자주 수식되는 아미노 그룹과 1위 위치의 카르복실 그룹입니다.
이 카르복실 그룹은 약 2.2의 특이적인 pKa를 갖춰
생리적 pH 범위 내에서 시알산에 음전하를 부여합니다 [10,13].
7, 8, 또는 9번 위치의 하이드록실 그룹이 아세트산과 에스테르화되는
O-아세틸화 형태는 덜 자주 발생합니다 [10].
기타 알려진 변형에는
8번 위치에서의 황산화 또는 메틸화,
9번 위치에서의 락틸화가 포함됩니다.
다중 위치에서의 치환이 발생할 수 있으며,
이는 7,8,9-트리-O-아세틸-N-아세틸- 및 N-글리콜릴뉴라민산 [8,9,51]에서 예시됩니다.
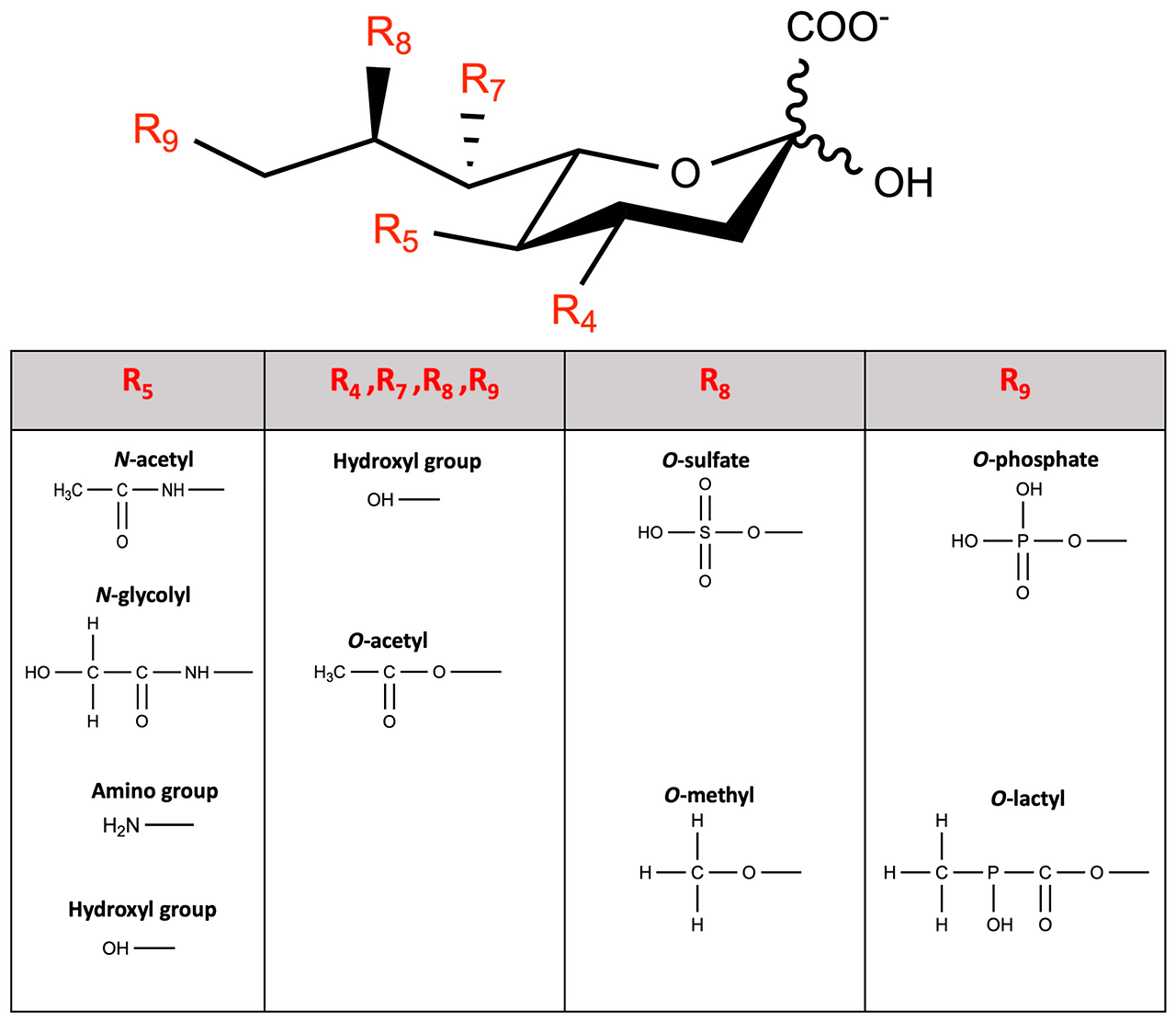
Figure 1. The molecular structure of sialic acid and its variants under various physiologic conditions. Sialic acids are characteristically carboxylate at group C1, attached to the C2 anomeric carbon. The wavy lines in C1 signify that the specific α or β anomeric configuration is not specified. The most common natural substituents (positions indicated in brackets) are a glycerol-like side chain (C7, 8, and 9) projecting out of the ring, and an acylated amino group at C5. Different groups with methyl, lactyl, acetyl, glycolyl, or sulfate can additionally diversify sialic acids. Prepared with ChemDraw, ver.: 23.1.2.7.
그림 1. 다양한 생리적 조건 하에서의 시알산 및 그 변이체의 분자 구조.
시알산은 C1 위치에 카르복실기가 결합되어 있으며, 이 카르복실기는 C2 아노메릭 탄소에 연결되어 있습니다.
C1 위치의 파선은 특정 α 또는 β 아노메릭 구조가 지정되지 않았음을 의미합니다.
가장 일반적인 자연적 치환기(괄호로 표시된 위치)는 고리에서 튀어나온 글리세롤 유사 측쇄(C7, 8, 9)와 C5에 위치한 아실화 아미노 그룹입니다.
메틸, 락틸, 아세틸, 글리콜릴, 또는 황산염 그룹이 추가로 시알산을 다양화할 수 있습니다.
ChemDraw, 버전 23.1.2.7로 작성되었습니다.
Widely distributed in a variety of human tissues and fluids [52], sialic acids exhibit particular functional roles associated with their specific structural features [10]. The most common member of this family is N-acetylneuraminic acid (Neu5Ac), with an acetylated amino group at position 5 (Figure 2) [7,50]. Neu5Ac can be converted to a less common N-glycolylneuraminic acid (Neu5Gc) by cytidine monophosphate-N-acetylneuraminic acid hydroxylase (Cmah), which hydroxylates the acetyl group [9,50,53]. Another sialic acid member, mainly found in its free form [10,54,55], and generally expressed at lower levels than Neu5Ac or Neu5Gc in mammalian tissues, is 2-keto-3-deoxy-D-glycero-D-galacto-nononic acid (KDN) [8,36,55,56]. More in-depth structural and stereochemical sialic acid analyses are presented elsewhere [6,10,25,57,58,59,60].
인간 조직과 체액의 다양한 부위에
널리 분포되어 있습니다 [52],
시알산은
특정 구조적 특징과 연관된 특별한 기능적 역할을 수행합니다 [10].
이 가족의 가장 일반적인 구성원은
5번 위치에 아세틸화된 아미노 그룹을 가진
N-아세틸뉴라민산(Neu5Ac)입니다 (그림 2) [7,50].
Neu5Ac는
시티딘 모노포스페이트-N-아세틸뉴라민산 하이드록시라제(Cmah)에 의해
아세틸 그룹을 하이드록실화하여
덜 일반적인 N-글리콜릴뉴라민산(Neu5Gc)으로 전환될 수 있습니다[9,50,53].
또 다른 시알산 계열 물질로,
포유류 조직에서 Neu5Ac 또는 Neu5Gc보다 낮은 수준으로 발현되는
2-케토-3-데옥시-D-글리세로-D-갈락토-노노닉 산(KDN)이 있습니다 [8,36,55,56].
시알산에 대한 구조적 및 입체화학적 분석은
다른 문헌에서 자세히 설명되어 있습니다 [6,10,25,57,58,59,60].
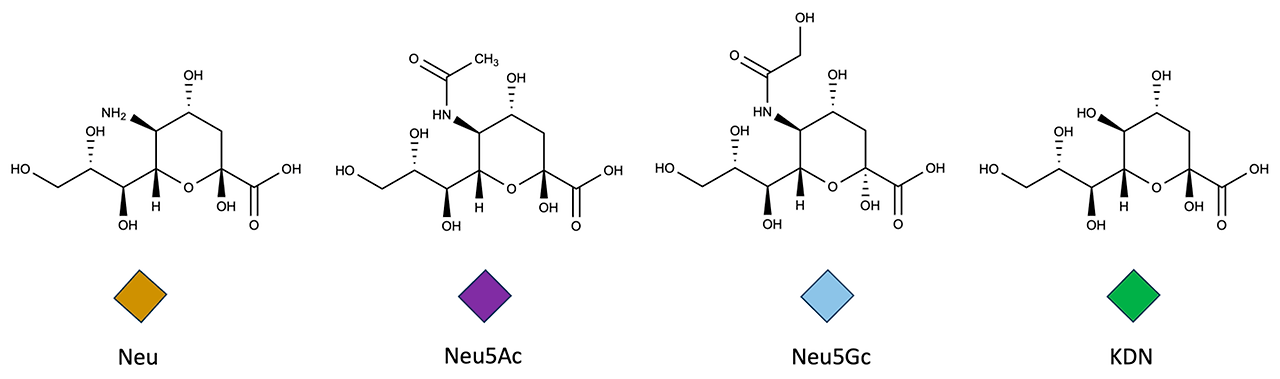
Figure 2. The structures of the most common sialic acids. The graphical representation of these glycans was developed according to the Symbol Nomenclature for Glycans (SNFG) guidelines. Prepared with ChemDraw, ver.: 23.1.2.7. Neu, neuraminic acid; Neu5Ac, N-acetylneuraminic acid; Neu5Gc, N-glycolylneuraminic acid (Neu5Gc); KDN, 2-keto-3-deoxy-D-glycero-D-galacto-nononic acid.
그림 2. 가장 일반적인 시알산(sialic acid)의 구조.
이 글리칸의 그래픽 표현은 글리칸 기호 명명법(Symbol Nomenclature for Glycans, SNFG) 지침에 따라 개발되었습니다. ChemDraw, 버전 23.1.2.7로 작성되었습니다. Neu, 뉴라민산; Neu5Ac, N-아세틸뉴라민산; Neu5Gc, N-글리콜릴뉴라민산 (Neu5Gc); KDN, 2-케토-3-데옥시-D-글리세로-D-갈락토-논오닉산.
1.3. Synthesis
In mammals, the synthesis of sialic acids involves more than 30 genes, encoding enzymes, and transporters allocated in various cellular compartments [61]. The synthesis of Neu5Ac begins in the cytosol with glucose, which enters the hexosamine biosynthetic pathway (HBP). This pathway phosphorylates glucose using ATP to produce the key metabolite uridine-5′-di-phospho-N-acetylglucosamine (UDP-GlcNAc) [60,62]. Then, the epimerase domain of UDP-N-acetylglucosamine 2-epimerase/N-acetylmannosamine kinase (GNE) mediates the formation of the uncharged monosaccharide N-acetylmannosamine (ManNAc) [63], with simultaneous cleavage of UDP [64,65,66]. ManNAc is phosphorylated by the kinase domain of GNE, leading to ManNAc-6P [67]. Sialic acid synthase (NANS) converts ManNAc-6P to Neu5Ac-9P in a condensation reaction with phosphoenol-pyruvate (PEP) [51], and subsequently, N-acetylneuraminic-acid-phosphatase (NANP) mediates the dephosphorylation to produce free Neu5Ac [68]. The free monosaccharide is activated in the nucleus by coupling with cytidine monophosphate (CMP), via the action of CMP-N-acetylneuraminate synthetase (CMAS), producing CMP-Neu5Ac [10,69]. Then, the CMP-Neu5Ac is added to lipid or protein to form glycans in the Golgi apparatus [70,71], or transferred to carbohydrate chains of nascent glycoconjugates by sialyltransferases (Figure 3) [10,13,24,72]. At this point, the bound Neu5Ac may be further altered via O-acetylation or O-methylation [73], before the glycoconjugate is transferred to the cell surface [72].
1.3. 합성
포유류에서 시알산의 합성은
30개 이상의 유전자,
효소 암호화 및 다양한 세포 구획에 분포된 수송체와 관련이 있습니다 [61].
Neu5Ac의 합성은
세포질에서 글루코스부터 시작되며,
이는 육당생합성 경로(HBP)로 들어갑니다.
이 경로는
ATP를 사용하여 글루코스를 인산화하여
핵심 대사산물인 우리딘-5′-디포스포-N-아세틸글루코사민(UDP-GlcNAc)을
그 다음, UDP-N-아세틸글루코사민 2-에피메라제/N-아세틸만노사민 키나제(GNE)의 에피메라제 도메인이
비전하 단당류 N-아세틸만노사민(ManNAc)의 형성을 매개합니다[63],
ManNAc는 GNE의 키나아제 도메인에 의해 인산화되어 ManNAc-6P로 전환됩니다 [67].
시알산 합성효소(NANS)는 ManNAc-6P를 포스포엔올피루베이트(PEP)와의 축합 반응을 통해 Neu5Ac-9P로 전환합니다 [51],
이후 N-아세틸뉴라민산 인산화효소(NANP)가 인산화 제거를 매개하여 자유로운 Neu5Ac를 생성합니다 [68].
자유로운 단당류는 핵 내에서 시티딘 모노포스페이트(CMP)와 결합하여 CMP-N-아세틸뉴라민산 합성효소(CMAS)의 작용으로 CMP-Neu5Ac를 생성합니다 [10,69].
이후 CMP-Neu5Ac는 골지체에서 지질 또는 단백질에 결합하여 글리칸을 형성하거나 [70,71], 시알릴전달효소(sialyltransferase)에 의해 신생 글리코콘쥬게이트의 탄수화물 사슬로 전달됩니다 (그림 3) [10,13,24,72].
이 단계에서 결합된 Neu5Ac는
O-아세틸화 또는 O-메틸화 [73]를 통해 추가로 변형될 수 있으며,
이후 글리코콘쥬게이트는 세포 표면으로 전달됩니다 [72].
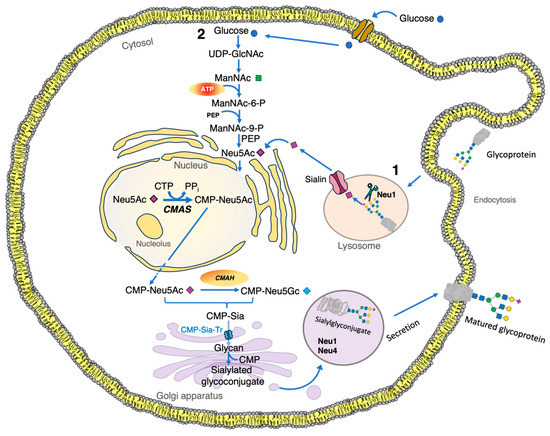
Figure 3. Sialic acid metabolism in eukaryote cells. (1) Sialic acid molecules found in the cytosol of eukaryotic cells are obtained from exogenous sources, such as glycolipids or glycoproteins. (2) Sialic acids can be produced from cytosolic UDP-GlcNAc molecules, biosynthesized from glucose in the hexosamine pathway. Finally, in the lysosome, after Neu1 hydrolyzes the terminal sialic acid residue from glycoconjugates, the sialylated glycoconjugate is transported by the sialin transporter encoded by the SLC17A5 gene. CMAH, cytidine monophospho-N-acetylneuraminic acid hydroxylase; CMAS, CMP-N-acetylneuraminate synthetase; CMP, cytidine-5′-monophosphate; CMP-Sia-Tr, CMP sialic acid transporter in the Golgi apparatus, encoded by the SLC35A1 gene; CTP, cytidine-5′-triphosphate; ManNAc, N-acetylmannosamine; ManNAc-6-P, N-acetylmannosamine 6-phosphate; ManNAc-9-P, N-acetylmannosamine 9-phosphate; Neu1, neuraminidase 1; Neu4, neuraminidase 4; PEP, phosphoenolpyruvate; PPi, pyrophosphate; Sia, sialic acid; Sialin, sialic acid transporter in the lysosome; UDP-GlcNac, uridine diphosphate N-acetylglucosamine.
그림 3. 진핵세포에서의 시알산 대사.
(1) 진핵세포의 세포질에 존재하는 시알산 분자는 글리콜리피드나 글리코프로틴과 같은 외부에서 유래한 원천으로부터 얻어집니다.
(2) 시알산은 글루코스에서 헥소사민 경로를 통해 생합성된 세포질 내 UDP-GlcNAc 분자로부터 생성될 수 있습니다.
마지막으로, 리소좀에서 Neu1이 당단백질에서 말단 시알산 잔기를 가수분해한 후, 시알화 당단백질은 SLC17A5 유전자에 의해 암호화되는 시알린 수송체에 의해 수송됩니다.
CMAH, 시티딘 모노포스포-N-아세틸뉴라민산 하이드록실라제; CMAS, CMP-N-아세틸뉴라민산 합성효소; CMP, 시티딘-5'-모노포스페이트; CMP-Sia-Tr, 골지체에서 SLC35A1 유전자에 의해 암호화되는 CMP 시알산 수송체; CTP, 시티딘-5'-트리포스페이트; ManNAc, N-아세틸만노사민; ManNAc-6-P, N-아세틸만노사민 6-포스페이트; ManNAc-9-P, N-아세틸만노사민 9-인산; Neu1, 뉴라미니다제 1; Neu4, 뉴라미니다제 4; PEP, 포스포엔올피루베이트; PPi, 피로인산; Sia, 시알산; Sialin, 리소좀 내 시알산 운반체; UDP-GlcNac, 우리딘 디포스페이트 N-아세틸글루코사민.
The main precursor for KDN biosynthesis is mannose (Man). The synthetic pathway comprises a series of reactions, similar to those for the biosynthesis of Neu5Ac (Figure 4) [60,74,75,76]. (1) Man + ATP → Man-6-P + ADP; (2) Man-6-P + PEP → KDN-9-P + Pi; (3) KDN-9-P → KDN + Pi. Reaction 1, catalyzed by a hexokinase, represents the 6-O-phosphorylation of mannose to form D-mannose-6-phosphate (Man-6-P). Reaction 2, catalyzed by KDN-9-phosphate (KDN-9-P) synthetase, condenses Man-6-P and PEP to form KDN-9-P. Reaction 3, catalyzed by a phosphatase, involves dephosphorylation of KDN-9-P to yield free KDN [12,55,74].
KDN 생합성의 주요 전구체는 만노스(Man)입니다. 합성 경로는 Neu5Ac의 생합성과 유사한 일련의 반응으로 구성됩니다(그림 4) [60,74,75,76]. (1) Man + ATP → Man-6-P + ADP; (2) Man-6-P + PEP → KDN-9-P + Pi; (3) KDN-9-P → KDN + Pi. 반응 1은 헥소키나아제(hexokinase)에 의해 촉매되어 만노스의 6-O-인산화 반응으로 D-만노스-6-인산(Man-6-P)을 형성합니다. 반응 2는 KDN-9-인산(KDN-9-P) 합성효소에 의해 촉매되어 Man-6-P와 PEP를 축합시켜 KDN-9-P를 형성합니다. 반응 3은 인산분해효소에 의해 촉매되어 KDN-9-P의 인산분해로 자유로운 KDN을 생성합니다 [12,55,74].
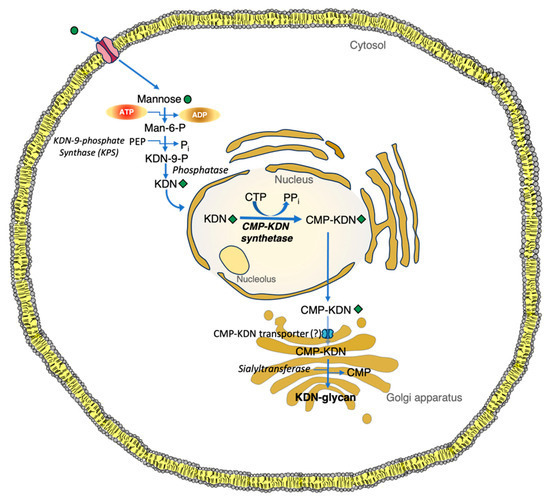
Figure 4. De novo pathways for the synthesis of KDN in animal cells. Man-6-P, a C2 epimer of glucose 6-phosphate, is a key precursor monosaccharide for KDN. The biosynthesis of KDN from Man-6-P follows comparable enzyme reaction steps to those of Neu5Ac from ManNAc-6-P. Notably, unlike other free monosaccharides, mannose is efficiently taken up by different cell types using a transporter that is unresponsive to glucose. CMP, cytidine-5′-monophosphate; CMP-KDN, cytidine 5′-(3-deoxy-D-glycero-D-galacto-2-nonulosonic phosphate); CMP-KDN-synthetase, CMP-3-deoxy-d-glycero-d-galacto-nonulosonic acid synthetase; CTP, cytidine triphosphate; KDN-9-P, KDN-9-phosphate; Man-6-P, mannose-6-phosphate; Pi, inorganic phosphate; PPi, pyrophosphate.
그림 4. 동물 세포에서의 KDN 합성을 위한 신규 경로.
글루코스 6-인산염의 C2 에피머인 Man-6-P는 KDN의 주요 전구체 단당류입니다. KDN의 Man-6-P로부터의 생합성은 ManNAc-6-P로부터 Neu5Ac가 합성되는 과정과 유사한 효소 반응 단계를 따릅니다. 특히, 다른 자유 단당류와 달리 만노스는 포도당에 반응하지 않는 운반체를 통해 다양한 세포 유형에 의해 효율적으로 흡수됩니다.
CMP, 시티딘-5′-모노인산; CMP-KDN, 시티딘 5′-(3-데옥시-D-글리세로-D-갈락토-2-노누로노산 인산); CMP-KDN-synthetase, CMP-3-deoxy-d-glycero-d-galacto-nonulosonic acid synthetase; CTP, cytidine triphosphate; KDN-9-P, KDN-9-phosphate; Man-6-P, mannose-6-phosphate; Pi, inorganic phosphate; PPi, pyrophosphate.
1.4. Functions
Sialic acids are involved in a variety of physiologically important functions [24,77], such as (a) antioxidant protection [78], (b) cell surface interactions [1], (c) infant bone development and skeletal growth [79], (d) immune recognition [80], (e) brain development [79,81], and (f) learning and memory [82].
In GNE myopathy patients and in a GNE mutant mouse model, muscles were hyposialylated and proteins were highly modified via S-nitrosylation, while oxidative stress-responsive genes were significantly upregulated [83]. In both cases, reactive oxygen species (ROS) production was elevated with cellular hyposialylation [83]. However, increasing overall sialylation through the intake of extrinsic sialic acid reduced ROS and protein S-nitrosylation. Additionally, the oral antioxidant N-acetylcysteine ameliorated muscle atrophy and weakness in GNE-mutant mice [83,84]. Together, this line of evidence demonstrates that overall sialylation is associated with increased antioxidant protection and improved reserve capacity to handle incidental ROS.
A biallelic mutation in NANS is associated with infantile-onset severe developmental delay and skeletal dysplasia [79,85]. The NANS gene is responsible for encoding the synthase for Neu5Ac [79,86]. Consequently, the immediate precursor substrate builds up, so that body fluids from affected individuals will have elevated levels of N-acetyl-D-mannosamine [79]. Knockdown of nansa, a zebrafish ortholog for the human NANS gene, results in abnormal skeletal development, while exogenously added sialic acid partially rescues this skeletal phenotype. Thus, NANS-mediated synthesis of sialic acid is required for early skeletal growth [79].
The glycocalyx is central to immune recognition and checkpoint inhibition, which enables pathogen detection and activation of immune defenses [80,87]. This carbohydrate coat serves as a cellular barrier that can either facilitate or obstruct immune cell interactions. Depending on the cell type and the composition of the glycosaminoglycan layer, the immune system may identify it as representing “self”. Alternatively, the sialic acid-rich layer may also potentially shield pathogens or cancerous cells from immune detection by masking their antigen-decorated surfaces with a carbohydrate-rich layer [88]. Ordinarily, for cancer cells to establish malignant lesions, they first need to evade recognition by immune cells [89,90]. They achieve this by tailoring their glycocalyx, which covers their neoantigens and other ligands, protecting them from acquired immune system cells. This alters the phagocytic efforts of myeloid cells and changes the normal immune responses [90]. This explains the upregulation of transmembrane mucins and other highly sialylated glycoproteins in cancer cells, as a way of establishing a protective glycocalyx [80,91,92]. Accordingly, in mouse models, tumor cells with the most efficient surface glycocalyx shielding have been shown to have the highest metastatic potential [93].
The human brain grows rapidly in early life and requires adequate nutrition [94,95,96]. Breastfeeding significantly improves the cognitive development of infants [94,97,98]. These benefits have been attributed to human milk oligosaccharides (HMOSs), some of which are linked with sialic acids [94,97,99]. Sialic acids are recognized as essential molecules for the proper development of gangliosides, and therefore, as critical for brain development and function [95]. Dietary supplementation with sialic acid or sialylated oligosaccharides is known to enhance intelligence and cognitive performance in early and later life [95,96,100]. Similarly, exogenous supplementation of sialic acid in animal models increases its concentration in the frontal cortex and improves learning performance [101]. As a building block for neurons, dietary 3′-sialyllactose is associated with improved language development in infants [94]. Conversely, some intellectual developmental disorders are associated with genetic defects affecting the synthesis of endogenous Neu5Ac [79].
1.4. 기능
시알산은
다양한 생리적으로 중요한 기능에 관여합니다 [24,77],
예를 들어
(a) 항산화 보호 [78],
(b) 세포 표면 상호작용 [1],
(c) 영아 골격 발달 및 성장 [79],
(d) 면역 인식 [80],
(f) 학습 및 기억 [82].
(a) antioxidant protection [78],
(b) cell surface interactions [1],
(c) infant bone development and skeletal growth [79],
(d) immune recognition [80],
(e) brain development [79,81], and
(f) learning and memory [82].
GNE 근육병 환자 및 GNE 돌연변이 마우스 모델에서
근육은 시알산화 수준이 낮았고,
단백질은 S-니트로실화 통해 고도로 변형되었으며,
산화 스트레스 반응 유전자 발현이 유의미하게 증가했습니다 [83].
두 경우 모두 세포 내 시알산화 감소와 함께
활성 산소 종 (ROS) 생산이 증가했습니다 [83].
그러나
외인성 시알산 섭취를 통해
전체 시알화 수준을 증가시키면
ROS와 단백질 S-니트로실화가 감소했습니다.
또한 경구 항산화제 N-아세틸시스테인은
GNE 돌연변이 마우스의 근육 위축과 약화를 완화시켰습니다 [83,84].
이러한 증거들은
전체 시알화가 항산화 보호 기능 강화와 우발적 ROS 대응을 위한
예비 용량 개선과 연관되어 있음을 보여줍니다.
NANS의 이중 대립 유전자는
영아기 발병의 심각한 발달 지연 및 골격 이형성증과 관련이 있습니다 [79,85].
NANS 유전자는
Neu5Ac의 신타제를 암호화하는 역할을 합니다 [79,86].
그 결과, 즉각적인 전구체 기질이 축적되어,
영향을 받은 개인의 체액에서 N-아세틸-D-만노사민의 수치가 상승합니다 [79].
인간 NANS 유전자의 zebrafish 동형인 nansa의 발현을 억제하면
골격 발달 이상이 발생하며,
외부에서 추가된 시알산은 이 골격 이상을 부분적으로 회복시킵니다.
따라서
NANS에 의한 시알산 합성은
초기 골격 성장에 필수적입니다 [79].
글리코칼릭스는
면역 인식과 체크포인트 억제에 중심적 역할을 하며,
병원체 감지와 면역 방어 활성화에 기여합니다 [80,87].
이 탄수화물 코트는
면역 세포 상호작용을 촉진하거나 방해할 수 있는 세포 장벽으로 기능합니다.
세포 유형과 글리코사미노글리칸 층의 구성에 따라 면역 체계는
이를 “자기”로 인식할 수 있습니다.
반면,
시알산이 풍부한 층은
항원 장식된 표면을 탄수화물 풍부한 층으로 가려
병원체나 암 세포를 면역 감지로부터 보호할 수 있습니다 [88].
일반적으로
암 세포가 악성 병변을 형성하려면
먼저 면역 세포의 인식을 회피해야 합니다 [89,90].
이들은
신항원 및 기타 리간드를 덮는 글리코칼릭스를 조정하여
획득 면역계 세포로부터 보호합니다.
이는 골수성 세포의 식작용 노력을 변화시키고 정상적인 면역 반응을 변형시킵니다 [90].
이는 암 세포에서 보호용 글리코칼릭스를 형성하기 위해
막을 관통하는 뮤신과 다른 고도로 시알화된 글리코프로틴의 발현이
이에 따라 마우스 모델에서 표면 글리코칼릭스 보호가
가장 효율적인 종양 세포가 가장 높은 전이 잠재력을 갖는 것으로 나타났습니다 [93].
인간 뇌는 생애 초기 단계에서 급속히 성장하며
모유 수유는
영아의 인지 발달을 크게 개선합니다 [94,97,98].
이러한 혜택은
인간 모유 올리고사카라이드(HMOSs)에 기인하며,
일부는 시알산과 연관되어 있습니다 [94,97,99].
시알산은
글리코사미노글리칸의 적절한 발달에 필수적인 분자로 인정되며,
따라서 뇌 발달과 기능에 필수적입니다 [95].
시알산 또는 시알화 올리고사카라이드의 식이 보충은
초기 및 후기 생애에서 지능과 인지 성능을 향상시키는 것으로 알려져 있습니다 [95,96,100].
동물 모델에서 시알산 외인성 보충은
전두엽 피질의 시알산 농도를 증가시키고 학습 성능을 개선합니다 [101].
신경 세포의 구성 요소로,
식이 3′-시알릴락토스는 영아의 언어 발달 개선과 연관되어 있습니다 [94].
반면, 일부 지적 발달 장애는
내인성 Neu5Ac 합성에 영향을 미치는 유전적 결함과 연관되어 있습니다 [79].
1.5. Genetic Disorders
1.5.1. UDP-GlcNAc 2-Epimerase/ManNAc Kinase (GNE) Myopathy
GNE myopathy is a hereditary myogenic disorder that results in muscle weakness due to a defect in the muscle itself. The GNE gene provides instructions for expression of the bifunctional GNE enzyme, which plays a key role in the synthesis of Neu5Ac [83,84,102]. The N-terminal epimerase domain of GNE uses UDP-GlcNAc as a substrate and epimerizes it into ManNAc by the epimerase domain, which is then subsequently phosphorylated by the kinase domain of the GNE enzyme [63,83].
Usually, GNE myopathy patients present with muscle symptoms between the ages of 15 and 35 [103]. The pathophysiology of the disease is not entirely understood, but hyposialylation of muscle glycans is thought to play an important role [84,102]. The typical presentation of bilateral foot drop in early adulthood is a consequence of weakness in the anterior tibialis muscles [102]. Over the subsequent decades, the disease gradually progresses, gradually affecting skeletal muscles throughout the body [102]. The quadriceps muscles tend to be relatively spared until the advanced stages of the disease [84,102,103].
Currently, a phase 2 clinical trial with ManNAc supplementation as a therapeutic intervention is ongoing for patients diagnosed with GNE myopathy [63,104]. Preliminary evidence of its long-term safety and biochemical efficacy, consistent with the intended mechanism of action, is reported in this cohort of patients [63]. Additionally, ManNAc supplementation restores the intracellular biosynthesis of Neu5Ac in affected subjects. Surprisingly, such an improvement is observed even in patients with homozygous GNE mutations in the kinase domain [104].
1.5. 유전적 장애
1.5.1. UDP-GlcNAc 2-에피메라제/ManNAc 키나제 (GNE) 근육병
GNE 근육병은
근육 자체의 결함으로 인해 근육 약화를 유발하는 유전성 근육 질환입니다.
GNE 유전자는
Neu5Ac 합성에 핵심 역할을 하는
이중 기능성 GNE 효소의 발현을 조절합니다 [83,84,102].
GNE의 N-말단 에피메라제 도메인은
UDP-GlcNAc를 기질로 사용하여 에피메라제 도메인을 통해 ManNAc로 에피메라제화한 후,
GNE 효소의 키나제 도메인에 의해 인산화됩니다 [63,83].
일반적으로
GNE 근병증 환자는
15세에서 35세 사이에 근육 증상이 나타납니다 [103].
이 질환의 병리 생리학은 완전히 밝혀지지 않았지만,
근육 당의 저시알릴화(hyposialylation)가
중요한 역할을 하는 것으로 생각됩니다 [84,102].
성인 초기 양측 발 마비가 나타나는 전형적인 증상은
이후 수십 년에 걸쳐 질환은 점차 진행되어
신체 전체의 골격근에 영향을 미칩니다 [102].
대퇴사두근은 질환의 진행 단계까지
상대적으로 덜 영향을 받습니다 [84,102,103].
현재 GNE 근육병 환자를 대상으로
ManNAc 보충제를 치료제로 사용하는 제2상 임상 시험이 진행 중입니다 [63,104].
이 환자 집단에서 보고된 초기 증거는 의도된 작용 기전과 일치하는 장기 안전성과 생화학적 효능을 보여줍니다 [63].
또한 ManNAc 보충은 영향을 받은 대상에서 Neu5Ac의 세포 내 생합성을 회복시킵니다. 놀
랍게도, 키나아제 도메인에 동형접합 GNE 돌연변이를 가진 환자에서도 이러한 개선이 관찰됩니다 [104].
1.5.2. Free Sialic Acid Storage Disorder (FSASD)
Lysosomes are cytoplasmic organelles that contain a variety of hydrolases [105]. A genetic deficiency in one of these hydrolases can result in the accumulation of material to be degraded by the lysosome [106]. Sometimes, such lysosomal storage can be caused by the deficiency of an activator protein [107,108]. Alternatively, accumulation of lysosomal material can also occur due to a transporter deficiency, as seen in the Salla disease [105,109].
Damage to the transport mechanism of free Neu5Ac through the lysosomal membrane leads to lysosomal accumulation of Neu5Ac [110,111]. Lysosomal free sialic acid storage disorders constitute a spectrum of uncommon, autosomal recessive, neurodegenerative, and multisystemic disorders caused by defective sialic acid lysosomal membrane exporter Sialin (Figure 3) [112,113,114].
The biallelic pathogenic gene variants of SLC17A5, which encode Sialin, produce three forms of free sialic acid storage disorders: (a) Salla disease, (b) intermediate severe Salla disease, and (c) infantile free sialic acid storage disease [109,114]. Salla disease and infantile free sialic acid storage disease exemplify the mildest and the most severe manifestations of the FSASD phenotypic spectrum, respectively [109]. At six months of age, infants with Salla disease typically manifest hypotonia, ataxia, developmental delay, and, in most cases, hypomyelination [105,115]. This is followed by a gradual neurological decline in an otherwise near-normal lifespan [115]. In contrast, infantile free sialic acid storage disease is characterized by nonimmune hydrops fetalis (24%), hepatosplenomegaly, failure to thrive, severe developmental delay, cardiomegaly, club feet, increasingly coarse facial features, neurological deterioration, and premature death [112]. The recently described intermediate severe Salla disease represents individuals with an intermediate phenotype [115]. Patients usually present within the initial six months of life with severe hypotonia and developmental delay, gradually progressing to ataxia, spasticity, and epilepsy [109].
1.5.2. 자유 시알산 저장 장애 (FSASD)
리소좀은
다양한 히드롤라제를 포함하는 세포질 소기관입니다 [105].
이러한 히드롤라제 중 하나에 유전적 결함이 발생하면
리소좀에 의해 분해되어야 할 물질이 축적됩니다 [106].
때로는 이러한 리소좀 저장 장애가
활성화 단백질 결핍으로 인해 발생할 수 있습니다 [107,108].
대안적으로,
리소좀 물질의 축적은 운반체 결핍으로 인해 발생할 수 있으며,
이는 Salla 질환에서 관찰됩니다 [105,109].
리소좀 막을 통해 자유 Neu5Ac의 운반 메커니즘 손상은
Neu5Ac의 리소좀 축적을 초래합니다 [110,111].
리소좀 내 자유 시알산 저장 장애는
시알산 리소좀 막 수출 단백질 시알린(그림 3)의 결함으로 인해 발생하는 드물고,
상염색체 열성, 신경퇴행성, 다계통성 장애의 스펙트럼을 구성합니다 [112,113,114].
Sialin을 암호화하는 SLC17A5의 생물학적 유전적 변이형은 세 가지 형태의 자유 시알산 저장 장애를 유발합니다:
(a) Salla 질병, (b) 중등도 Salla 질병, (c) 영아 자유 시알산 저장 질병 [109,114].
Salla 질환과 영아형 자유 시알산 저장 장애는 FSASD 표현형 스펙트럼의 가장 경증과 가장 중증 사례를 각각 대표합니다 [109]. Salla 질환을 가진 영아는 생후 6개월경에 근육 긴장 저하, 운동 실조, 발달 지연, 대부분의 경우 신경절 세포 미성숙을 나타냅니다 [105,115]. 이는 otherwise near-normal lifespan 동안 점진적인 신경학적 악화로 이어집니다 [115]. 반면, 유아기 자유 시알산 저장 질환은 비면역성 태아 수종 (24%), 간비대, 성장 부진, 심각한 발달 지연, 심장 비대, 발굽 발, 점차적으로 거친 얼굴 특징, 신경학적 악화, 조기 사망으로 특징지어집니다 [112]. 최근에 보고된 중간 중증 Salla 질환은 중간 표현형을 보이는 개인을 나타냅니다 [115]. 환자들은 생후 6개월 이내에 심각한 근육 이완과 발달 지연을 보이며, 점차 운동 실조, 경련, 간질로 진행됩니다 [109].
2. Diet
In addition to their endogenous production, sialic acids can also be obtained from dietary sources, absorbed by the intestine, and transported via circulation to tissues [104,116,117]. Most of the common sialic acids are obtained through dietary sources [118], either from red meat, in the form of lamb, pork, or beef, or from a variety of milk products [119,120]. Exogenous Neu5Ac can be picked up by cells via fluid pinocytosis and a lysosomal transporter [121]. In contrast to Neu5Ac, Neu5Gc cannot be synthesized by human tissues from endogenous precursors [122]. Instead, it is metabolically incorporated in the tissues of subjects who consume red meat [123,124,125]. Consequently, low levels of surface expression of diet-derived Neu5Gc occur on endothelial and some epithelial cells [53,126]. Once sialic acids are internalized by cells into the cytosol [116], they are integrated with endogenous glycoconjugates, of which a relatively small fraction is retained in the body, while the rest is metabolized and excreted in urine [126,127] or feces [104].
Unlike animals, plants are known to lack sialic acids [110,128,129]. However, they are rich sources of precursors for the synthesis of sialic acids [7,60,75,129]. In fish, O-acetylated Neu5Ac occurs frequently on N-glycans, while a smaller number of Neu5Acα2-8Neu5Ac structures are also observed [130]. Neu5Gc is seen very rarely in certain marine sources (fish eggs or echinoderms) [116,131,132] and is apparently not synthesized by birds or reptiles [133,134].
The precise metabolic fates and the relative contributions of sialic acids from endogenous and dietary sources in humans remain unknown, making the inferences from current models difficult [126]. Implications of dietary uptake and the utilization of Neu5Ac and Neu5Gc deserve further investigation.
2. 식이
내인성 생산 외에도
시알산은 식이 소스에서 얻어지며, 장에서 흡수되어 순환계를 통해 조직으로 운반됩니다 [104,116,117].
일반적인 시알산은
주로 식이 소스에서 얻어집니다 [118],
적색 육류(양고기, 돼지고기, 소고기)나
외인성 Neu5Ac는
세포에 의해 체액성 핀오시토시스 및 리소좀 운반체를 통해 흡수됩니다 [121].
Neu5Ac와 달리 Neu5Gc는
인간 조직에서 내인성 전구체로부터 합성되지 않습니다 [122].
대신, 적색 육류를 섭취하는 대상의 조직에서
따라서 식이 유래 Neu5Gc의 표면 발현 수준은
내피 세포와 일부 상피 세포에서 낮게 나타납니다 [53,126].
시알산이 세포 내 사이토솔로 내재화되면 [116],
내인성 글리코콘쥬게이트와 통합됩니다.
이 중 상대적으로 작은 부분은 체내에 유지되며,
나머지는 대사되어 소변 [126,127] 또는 대변 [104]을 통해 배설됩니다.
동물과 달리 식물은
그러나 시알산 합성의 전구체로 풍부한 원천입니다 [7,60,75,129].
어류에서는 N-글리칸에 O-아세틸화 Neu5Ac가 자주 관찰되며,
Neu5Acα2-8Neu5Ac 구조는 상대적으로 적은 수로 관찰됩니다 [130].
Neu5Gc는 특정 해양 자원(어류 알이나 척삭동물)에서 매우 드물게 관찰되며 [116,131,132],
조류나 파충류에서는 합성되지 않는 것으로 알려져 있습니다 [133,134].
인간에서 내인성 및 식이성 시알산산의 대사 경로와 상대적 기여도는
아직 명확히 밝혀지지 않아 현재 모델로부터의 추론이 어렵습니다 [126].
Neu5Ac 및 Neu5Gc의 식이 섭취 및 이용에 대한 함의는
추가 연구가 필요합니다.
Microbiome and Sialic Acids
Plant polysaccharides include long-chain glycans, as components of plant-based fiber, which animals generally cannot digest [135]. Fiber, however, is an essential source of carbon and energy that supports the maintenance and functioning of gut microbiota [136]. These gut bacteria, which are able to utilize various types of plant glycans [136], are categorized into seven predominant divisions: Actinobacteria, Bacteroidetes, Cyanobacteria, Firmicutes, Fusobacteria, Proteobacteria, and Verrucomicrobia. Of these, Bacteroidetes and Firmicutes represent more than 90% of the microbiome [137]. Gut bacteria are believed to be essential to human health by (a) conferring additional energy-harvesting capacity, (b) providing niche exclusion of pathogens, (c) producing vitamins and glycans, and (d) carrying out molecular communication activities, all of which form an integral part of human physiologic processes [136,138,139]. Thus, the quantity and quality of plant glycans consumed daily by the human host can have an impact on health [136]. Both pre-clinical and clinical studies indicate that different types of dietary plant glycans and their metabolic products contribute beneficial metabolic effects to the host [140]. For example, the fermentation of cellulose, hemicellulose, resistant starch, pectin, lignin, or various oligosaccharides can lead to the production of short-chain fatty acids (SCFAs) like acetic, propionic, or butyric acids, which, in turn, help to regulate glucose and lipid metabolism [140,141]. The release of these fatty acids can occur during digestion in the upper gastrointestinal tract but may also continue in the colon [142].
Studies of the microbiome composition in adults are mainly focused on the resulting health impacts, while the effects of dietary sialic acids in this context are largely overlooked [143]. Given the considerable dietary variation between different people, the effects of dietary sialic acids on the adult gut microbiome are often difficult to tease apart. Consequently, cohort studies focusing on particular dietary patterns, like the Adventist Health Study-2 [144] or the EPIC-OXFORD study [145], are potentially helpful in examining the impact of dietary sialic acids, like Neu5Ac or Neu5Gc, on the gut microbiome. Such information about the relationship between dietary sialic acids and the microbiome may reveal additional insights into several non-communicable diseases, like type 2 diabetes [146,147], cardiovascular disorders [148,149], or cancer [12,150,151,152].
Bacteria can release free sialic acids or use them as nutrient sources [153,154]. Gut microbiome symbionts, such as Ruminococcus gnavus, can scavenge Neu5Ac from mucins and convert it to 2,7-anhydro-Neu5Ac [1]. However, catabolism of 2,7-anhydro-Neu5Ac is not restricted to R. gnavus. Escherichia coli can also transport and catabolize sialic acids such as Neu5Ac, Neu5Gc, or KDN to satisfy its carbon and nitrogen needs. These sialic acids are transported via the transporter NanT and catabolized using the sialic acid aldolase NanA [155]. Glycosylhydrolases cleave the glycoketosidic linkages of sialic acid O-acceptor substrates through an exohydrolytic reaction [156]. In addition, bacterial sialidases can release Neu5Gc from red meat [153,157], helping to reduce the inflammatory effects of red meat consumption [143].
미생물군집과 시알산
식물 다당류는
식물 기반 섬유의 구성 성분으로,
동물은 일반적으로 소화할 수 없는 장쇄 글리칸을 포함합니다 [135].
그러나 섬유는
장 미생물군집의 유지 및 기능에 필수적인
탄소 및 에너지 공급원입니다 [136].
이 장내 세균은
다양한 유형의 식물 글리칸을 활용할 수 있으며 [136],
7개의 주요 분류군으로 구분됩니다:
Actinobacteria, Bacteroidetes, Cyanobacteria, Firmicutes, Fusobacteria, Proteobacteria, 및 Verrucomicrobia. 이 중 Bacteroidetes와 Firmicutes는
미생물군의 90% 이상을 차지합니다 [137].
장 내 세균은
(a) 추가적인 에너지 획득 능력을 부여하고,
(b) 병원체 배제 기능을 제공하며,
(c) 비타민과 글리칸을 생산하며,
(d) 분자 간 통신 활동을 수행함으로써 인간 생리 과정의 필수적인 부분을 형성합니다 [136,138,139].
따라서
인간 호스트가 매일 섭취하는 식물 글리칸의 양과 품질은
건강에 영향을 미칠 수 있습니다 [136].
전임상 및 임상 연구는
다양한 유형의 식이 식물 글리칸과 그 대사 산물이 호스트에게 유익한 대사 효과를 기여한다는 것을 보여줍니다 [140].
예를 들어,
셀룰로오스, 헤미셀룰로오스, 저항성 전분, 펙틴, 리그닌 또는 다양한 올리고사카라이드의 발효는
아세트산, 프로피온산, 부티르산과 같은 단쇄 지방산(SCFAs)의 생산으로 이어질 수 있으며,
이는 다시 포도당 및 지질 대사 조절에 도움을 줍니다 [140,141].
이러한 지방산의 방출은
상부 소화관에서 소화 과정에서 발생할 수 있지만
대장에서 계속될 수도 있습니다 [142].
성인의 미생물군집 구성에 대한 연구는 주로 건강 영향에 초점을 맞추고 있으며, 이 맥락에서 식이 시알산산의 영향은 대부분 간과되고 있습니다 [143]. 개인 간 식이 다양성이 크다는 점을 고려할 때, 식이 시알산산이 성인 장 미생물군집에 미치는 영향을 명확히 구분하는 것은 종종 어렵습니다. 따라서 특정 식이 패턴에 초점을 맞춘 코호트 연구, 예를 들어 Adventist Health Study-2 [144] 또는 EPIC-OXFORD 연구 [145]는 식이 시알산(예: Neu5Ac 또는 Neu5Gc)이 장 미생물군집에 미치는 영향을 조사하는 데 유용할 수 있습니다. 식이 시알산과 미생물군집 간의 관계에 대한 이러한 정보는 제2형 당뇨병 [146,147], 심혈관 질환 [148,149], 또는 암 [12,150,151,152]과 같은 여러 비전염성 질환에 대한 추가적인 통찰을 제공할 수 있습니다.
세균은 자유로운 시알산을 방출하거나 영양소원으로 사용할 수 있습니다 [153,154]. 장 미생물군집의 공생균인 Ruminococcus gnavus는 뮤신에서 Neu5Ac를 흡수하여 2,7-anhydro-Neu5Ac로 전환할 수 있습니다 [1]. 그러나 2,7-anhydro-Neu5Ac의 분해는 R. gnavus에 한정되지 않습니다. Escherichia coli는 Neu5Ac, Neu5Gc, 또는 KDN과 같은 시알산을 운반하고 분해하여 탄소와 질소 요구를 충족시킬 수 있습니다. 이러한 시알산은 NanT 운반체를 통해 운반되며, 시알산 알도라제 NanA를 사용하여 분해됩니다 [155]. 글리코실하이드로라제는 시알산 O-수용체 기질의 글리코케토시드 결합을 외수분해 반응을 통해 분해합니다 [156]. 또한 세균성 시알리다제는 적색 육류에서 Neu5Gc를 방출하여 적색 육류 섭취의 염증 효과를 감소시키는 데 기여합니다 [153,157] [143].
3. Interactions with Pathogens
Sialic acids act as receptors for various pathogens that produce infectious diseases [158]. Some pathogens can synthesize or appropriate sialic acids from their hosts and incorporate them into their own glycoconjugates and derivatives [159]. For example, bacteria produce adhesins or toxins that interact with sialic acids on host cells [157], viruses can use sialic acids to attach to and enter cells [160], and protozoa can use sialic acids to elude host immunity [161,162,163].
3. 병원체와의 상호작용
시알산은 감염성 질환을 유발하는 다양한 병원체의 수용체로 작용합니다 [158]. 일부 병원체는 호스트로부터 시알산을 합성하거나 획득하여 자신의 글리코콘쥬게이트 및 유도체에 통합합니다 [159]. 예를 들어, 세균은 호스트 세포의 시알산과 상호작용하는 접착인자나 독소를 생성합니다 [157], 바이러스는 세포에 부착하고 침투하기 위해 시알산을 활용합니다 [160], 원생동물은 호스트 면역계를 회피하기 위해 시알산을 사용합니다 [161,162,163].
3.1. Sialic Acids and Bacteria
Some bacterial species can produce Neu5Ac and corresponding glycoconjugates. Others, in contrast, are merely able to use sialic acids as carbon sources. This capability to use sialic acids appears to be correlated with bacterial virulence in a range of infections, particularly in the profusely sialylated environment of the gut [143]. Bacteria obtain sialic acids in two ways: either via de novo synthesis, or from scavenging pathways using specific sialidases to cleave sialic acids from glycoconjugates [155,164]. As in eukaryotes, the de novo synthesis pathway in bacteria begins with the phosphorylation of glucose by a hexokinase [165].
In E. coli, in the NeuC pathway, glucose undergoes conversion into glucose-6-phosphate through the action of a hexokinase [166,167,168,169,170]. This is followed by the conversion of glucose-6-phosphate to fructose-6-phosphate by glucose-6-phosphate isomerase (PGI). Four reactions take place sequentially, facilitated by three enzymes: glucosamine-6-phosphate synthase (glmS), phosphoglucosamine mutase (glmM), and the bi-functional enzyme glucosamine-1-phosphate acetyltransferase/N-acetylglucosamine-1-phosphate uridyltransferase (glmU). The result of these reaction steps is the formation of UDP-GlcNAc [166,171]. The conversion of UDP-GlcNAc to ManNAc is catalyzed by UDP-GlcNAc 2-epimerase [172] and NeuC, which is responsible for the biosynthesis of the precursor of N-acetyl-D-mannosamine, followed by NeuB-catalyzed conversion of N-acetyl-D-mannosamine with phosphoenolpyruvate to the product Neu5Ac (Figure 5) [168,171,172,173,174,175,176]. Alternatively, a shortened synthetic pathway can utilize sialyl precursors scavenged from animal hosts to produce Neu5Ac [177], particularly during cell wall synthesis [165].
3.1. 시알산과 세균
일부 세균 종은 Neu5Ac와 대응하는 글리코콘쥬게이트를 생성할 수 있습니다. 반면 다른 종은 시알산을 단순히 탄소원으로만 사용할 수 있습니다. 시알산을 활용하는 이 능력은 다양한 감염에서 세균의 독성과 관련이 있으며, 특히 장 내 시알산이 풍부한 환경에서 두드러집니다 [143]. 박테리아는 시알산을 두 가지 방법으로 획득합니다: 신규 합성 또는 특정 시알리다아제를 사용하여 글리코콘쥬게이트에서 시알산을 분해하는 회수 경로를 통해 [155,164]. 진핵생물과 마찬가지로 박테리아의 신규 합성 경로는 글루코오스의 인산화 과정으로 시작되며, 이는 헥소키나아제에 의해 촉진됩니다 [165].
E. coli의 NeuC 경로에서 글루코스는 헥소키나아제의 작용으로 글루코스-6-인산으로 전환됩니다 [166,167,168,169,170]. 이어 글루코스-6-인산은 글루코스-6-인산 이소메라제 (PGI)에 의해 프럭토스-6-인산으로 전환됩니다. 이 네 가지 반응은 세 가지 효소에 의해 순차적으로 촉진됩니다: 글루코사민-6-인산 합성효소(glmS), 포스포글루코사민 뮤타제(glmM), 그리고 이중 기능 효소인 글루코사민-1-인산 아세틸트랜스퍼레이스/N-아세틸글루코사민-1-인산 우리딜트랜스퍼레이스(glmU). 이 반응 단계의 결과는 UDP-GlcNAc의 형성입니다 [166,171]. UDP-GlcNAc에서 ManNAc로의 전환은 UDP-GlcNAc 2-에피메라제 [172]와 NeuC에 의해 촉매되며, 이는 N-아세틸-D-만노사민의 전구체 생합성에 관여합니다. 이어 NeuB에 의해 N-아세틸-D-만노사민과 포스포엔올피루베이트가 반응하여 최종 제품인 Neu5Ac로 전환됩니다 (그림 5) [168,171,172,173,174,175,176]. 대안으로, 동물 호스트에서 회수된 시알릴 전구체를 활용하는 단축된 합성 경로를 통해 Neu5Ac를 생산할 수 있습니다 [177], 특히 세포벽 합성 시에 특히 그렇습니다 [165].
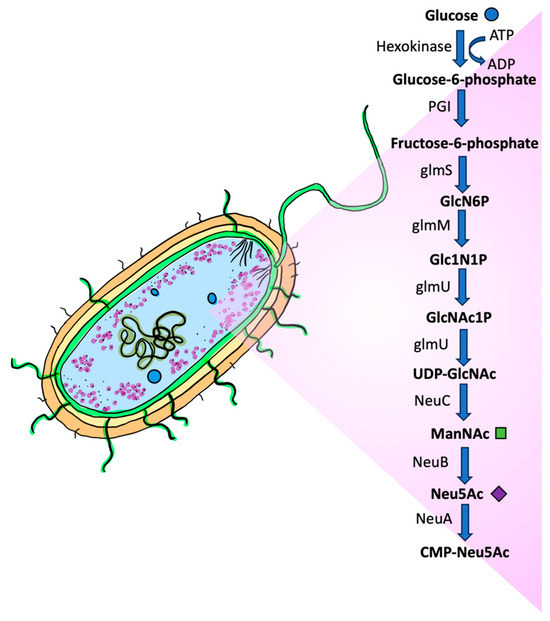
Figure 5. Biosynthesis of N-acetylneuraminic acid in prokaryotes. In the bacterium E. coli, glucose is metabolized through a series of enzymatic reactions. The process begins when glucose enters the cell and is phosphorylated by hexokinase to form glucose-6-phosphate. The next step involves an isomerization reaction catalyzed by the enzyme glucose-6-phosphate isomerase (PGI), which converts glucose-6-phosphate into fructose-6-phosphate by a rearrangement. The pathway proceeds through several more steps, including the cleavage of fructose-6-phosphate, followed by a series of four enzyme-catalyzed reactions to generate a nucleotide sugar called UDP-N-acetylglucosamine (UDP-GlcNAc). Then, UDP-GlcNAc is converted to ManNAc, catalyzed by NeuC, a hydrolyzing UDP-GlcNAc epimerase. Neu5Ac is produced via a condensation reaction of ManNAc with phosphoenol pyruvate (PEP), which is catalyzed by NeuB (sialic acid synthase). The Neu5Ac is primed by NeuA (Sialic acid O-acetylesterase) and CTP to form CMP-Neu5Ac. CMP: cytidine 5′-monophosphate. CTP: cytidine triphosphate; CMP-Neu5Ac: cytidine-5′-monophospho-N-acetylneuraminic acid; UDP-GlcNAc: uridine diphosphate N-acetylglucosamine.
Some bacteria may be able to synthesize additional structurally related NulOs. Such bacterial NulOs include legionaminic acid [157], pseudaminic acid [178,179], fusaminic acid [180], and acinetaminic acid [181]. In this context, the naming convention typically follows a name derivative of the microbial species first reported as its producer [178]. For example, legionaminic acid was first observed in Legionella pneumophila, known for the respiratory infection Legionnaires disease [182]. Similarly, acinetaminic acid was first identified in the multi-drug-resistant pathogen Acinetobacter baumannii [154,183].
3.1.1. Porphyromonas gingivalis
Periodontitis is one of the most prevalent oral diseases [184], characterized by gradual damage of the periodontal ligament and surrounding tissues, which can potentially include alveolar bone, with ultimate tooth loss [185]. These conditions are characteristically associated with poor brushing and flossing habits, allowing polymicrobial colonization on the surfaces of teeth, in the form of a sticky biofilm [186]. Porphyromonas gingivalis serves as a keystone pathogen in the microbial community, responsible for the occurrence and progression of periodontitis [187,188,189].
The survival of P. gingivalis next to the oral mucosa depends on its dynamic interface and its cell membrane [190], which facilitates the release of wastes and the acquisition of nutrients. Once attached to the oral cavity, P. gingivalis utilizes the host’s amino acids and other carbon-containing molecules as energy sources and as building materials for its functions and growth [191,192]. Interestingly, this Gram-negative pathogen, although long described as asaccharolytic, can utilize sugars such as glucose for the biosynthesis of other intracellular macromolecules [193]. The cell membrane of P. gingivalis also contributes toward effective tissue colonization [190], by releasing vesicles with an arsenal of virulence factors, including lipopolysaccharides (LPS), fimbriae, gingipains, and others [194,195]. The host’s immune system responds to these by ramping up the production and release of inflammatory cytokines such as tumor necrosis factor (TNF)-α and interleukin (IL)-1β [196]. The interplay between the destructive microorganisms and the pro-inflammatory immune responses causes periodontal tissue degradation [190,197].
In addition, P. gingivalis can produce and release sialidases, which can also serve as virulence factors [156]. The infection may be sustained by the action of sialidases and sialopeptidases (the O-sialoglycoprotease hydrolyzes the sialic acid O-acceptor substrate through an endohydrolytic reaction [156]), cleaving the sialic acids from the host glycoproteins [156] and contributing to disease progression [189]. In a similar manner, P. gingivalis virulence may be further enhanced if the sialidases also regulate the activity of gingipains (cysteine proteases) [156,198]. Conversely, the inhibition of P. gingivalis sialidases may be strategically helpful in treating periodontitis [189].
3.1.2. Streptococcus pneumoniae
Streptococcus pneumoniae, commonly known as pneumococcus, is a Gram-positive extracellular opportunistic pathogen adapted to the human upper respiratory tract [199]. This bacterium is especially predominant in pre-school-age children and those attending daycare centers [199,200]. S. pneumoniae is responsible for a spectrum of diseases, ranging from mild respiratory tract infections, such as sinusitis or otitis media, to severe conditions, like pneumonia, septicemia, or meningitis [181,182,184]. Pneumococcal infections pose a significant threat to global health. Globally, deaths attributed to pneumococcal disease among children under the age of five have been estimated to reach around 1.8 to 2.0 million, causing about 11% of all deaths in this age group [200,201,202,203].
The primary source of S. pneumoniae spread is mediated by transmission from the host nasopharyngeal airway [203]. Upon entering the human nasopharynx, S. pneumoniae encounters mucus, which is rich in glycoconjugates with terminal sialic acid residues [202]. The respiratory tract is coated with mucin glycoproteins, which are rich in GlcNAc, N-acetylgalactosamine (GalNAc), galactose, and Neu5Ac [202,204]. These sugars are degraded by the glycan-specific metabolic machinery of S. pneumoniae [204], a process that includes the removal of terminal sialic acids, which also serve as carbon sources for the bacteria [204,205].
S. pneumoniae expresses up to three sialidases, including SpNanA, SpNanB, and/or SpNanC [201]. SpNanA, along with SpNanB, provides a source of sialic acids to the pathogen, and helps in host infection, nutrition, colonization, and biofilm formation [201,206,207]. SpNanC serves as a regulator of hydrolytic sialidases, such as SpNanA, catalyzing the production of the sialidase inhibitor Neu5Ac2en [201,208]. SpNanC is thought of as a potential marker of pneumococcal haemolytic uraemic syndrome in children [201], since its activity is linked to the development of this severe complication [201,209,210].
Continuing studies of these sialidases and their structure–function relationships have the potential to yield novel approaches to bacterial infections [188,192,193,194,195]. The development of selective inhibitors against sialidases opens up new perspectives for the treatment of S. pneumoniae [201].
3.1.3. Haemophilus influenzae
Haemophilus influenzae is a human pathogen that is adapted to its host [211,212]. It is classified into typeable strains, which produce a polysaccharide capsule (serotypes a to f), and nontypeable, noncapsulated strains of H. influenzae (NTHi) [211,213]. Healthy humans may asymptomatically carry H. influenzae in the upper respiratory tract [214]. Typable H. influenzae strains are associated with invasive forms of infection, such as sepsis or meningitis [215]. In contrast, NTHi strains are more often responsible for airway-associated inflammation, such as sinusitis, conjunctivitis, pharyngitis, otitis media, and pneumonia, in addition to possible bacteremia and meningitis [216]. Invasive NTHi, representing 43–91% of invasive isolates, is increasing in a number of countries, including Canada, the Netherlands, Portugal, Slovenia, and Sweden [217]. NTHi tends to cause severe invasive disease in neonates and in children, with significant comorbidities [216,218,219]. Approximately 10% of children from 2 to 4 years of age [220], and 17% of children in their first year, succumb to fatal NTHi infections [219,220,221]. The increased incidence of invasive NTHi is due to several factors, including higher vulnerability of some patients [217,220].
H. influenzae relies on its capacity to resist host defenses, including the complement system [212,222]. While evading both innate and adaptive immunity, H. influenzae causes recurrent and persistent infections [222]. Several factors contribute to the survival of NTHi [211,220], including the sialylation of the released lipooligosaccharide from outer membrane fragments during bacterial growth [215,220,223]. This modification protects specific epitopes of the lipooligosaccharide from bactericidal IgM in circulation [222,223]. Sialic acids, incorporated as the terminal residues on lipooligosaccharides, enable NTHi to evade antibacterial defenses, to persist within mucosal tissues, and to form a biofilm. Neither the underlying mechanisms involved in these processes, nor the role of sialic acid scavenged by NTHi during infection, are well understood yet [222,223]. However, since NTHi strains defective in sialic acid utilization are no longer virulent in animal models for otitis media, the sialic acid utilization pathway is a promising therapeutic target [223,224].
3.1.4. Pseudomonas aeruginosa
Pseudomonas aeruginosa is a free-living, motile, and aerobic Gram-negative bacterium [225,226]. It is an opportunistic infectious pathogen, generally associated with nosocomial infections, severe burns, chronic cystic fibrosis, cancer, AIDS, transplantation, and other immunosuppressed conditions [226].
P. aeruginosa lacks the de novo sialic acid synthetic pathway. However, it can take up monosaccharides from exogenous sources, including the glycolyl sialic acid derivative, Neu5Gc [227,228]. When sialic acids are incorporated by P. aeruginosa, the deposition of the complement C3b on the surface of the bacterial cell is reduced [228]. Furthermore, sialylated P. aeruginosa has an affinity for human CD33 receptors, including the immune cell surface receptors Siglecs 7 and 9, as a characteristic shared with other sialylated bacteria [227,229,230,231]. The sialylated P. aeruginosa also suppresses macrophage antimicrobial responses and inhibits phagosome maturation, thereby enabling its persistent viability and intracellular replication within macrophages [227]. In conclusion, acquisition of sialic acids by P. aeruginosa within the host contributes toward bacterial pathogenicity and further host infection by reducing complement deposition and by mimicking Siglec-dependent recognition.
3.1.5. Clostridium perfringens
Clostridium perfringens is the primary pathogen in intestinal and histotoxic infections, producing conditions such as gas gangrene and clostridial myonecrosis [232,233]. C. perfringens is also frequently associated with cases of food poisoning [234]. C. perfringens is known to be able to utilize sialic acids as carbon sources [232,235]. In addition to a variety of toxins [233], this Gram-positive anaerobic bacterium also produces a number of sialidases. Of these, NanH has a cytoplasmic location during the culture log-growth phase. In contrast, NanJ and NanI are secreted exosialidases [232,233,236]. Most C. perfringens strains produce all three sialidases. However, some C. perfringens strains produce only one or two of them. For strains that produce all three, NanI is typically responsible for 70% of total exosialidase activity [236]. When present, NanI enhances the colonization of C. perfringens in the intestinal tract [237] and increases the cytotoxic activity and interaction of several toxins with host cells [232].
The role of sialidases in histotoxic infections remains unclear [233]. Sialidases could enhance C. perfringens adherence to host cells through both nonspecific and specific mechanisms [233]. The nonspecific mechanisms include disruptions of physiologic host cell interactions. Terminal sialic acids promote endothelial barrier integrity and cellular interactions in epithelial monolayers [238]. Sialidases mediate the removal of terminal sialic groups, leading to barrier disruption and increasing access for C. perfringens [238,239]. Therefore, nonspecific effects of secreted NanI on both surface charges and epithelial barrier integrity could help to increase toxin binding and C. perfringens colonization [238].
The specific mechanisms of sialidase-mediated enhancement of C. perfringens infectivity include modifications to host cell surface receptors. Some strains, such as C. perfringens CN3718, display significantly enhanced adhesion to specific mammalian cells, including Caco-2 and HT-29 intestinal cell lines [240]. Similarly, toxins involved in C. perfringens intestinal infections bind to and affect only certain cells. These observations suggest that NanI sialidase modifies host cell surface adhesins and toxin receptors and trims adjacent molecules to enhance the accessibility of the adhesin or toxin receptors [233].
C. perfringens infections pose significant challenges, in spite of antibiotic treatments, due to the persistence of previously synthesized toxins [233,241]. Similarly, altered surface antigens in certain C. perfringens strains make it difficult to prepare comprehensively effective vaccines or neutralizing antibodies [234]. As a result, development of effective sialidase inhibitors represents novel treatment options for C. perfringens infections.
3.1.6. Neisseria gonorrhoeae
Gonorrhea poses a significant global health challenge, due to escalating infection rates, the emergence of antibiotic resistance, and evasion of defensive immune responses [242,243,244]. The exact mechanisms of N. gonorrhoeae-mediated disruptions of host immune responses are not well understood. It is known, however, that N. gonorrhoeae sialyltransferase attaches sialic acids, scavenged from the host, to its own abundant surface lipooligosaccharide [243]. The length and composition of these glycolipids are regulated by genes encoding the lipooligosaccharide glycosyltransferases [245,246]. The expression of these genes constitutes a primary mechanism of bacterial adaptation to challenging environments by altering their surface oligosaccharide composition [233,235]. N. gonorrhoeae, isolated from urethral infection, predominantly produces lipooligosaccharides, which can be sialylated by sialyltransferases for optimal genital tract infection [247,248]. In this context, CMP-sialic acid from the host can be used by N. gonorrhoeae sialyltransferases to attach sialic acid to its own acceptor hydroxyl groups [243]. Many aspects of the N. gonorrhoeae virulence mechanism, and its association with sialic acids, remain to be worked out.
3.2. Sialic Acids and Viral Interactions
The initial step in the viral life cycle is the attachment of virus particles to the cell surface [249]. This adherence is facilitated by the interaction of the virus with a specific receptor [250]. Receptor molecules are integral components of the cell membrane. The receptor determinant, which serves as the binding site for the virus, may consist of either a protein epitope or the carbohydrate molecule of a glycoprotein or glycolipid [160]. Soluble proteins in body fluids and mucus on respiratory and enteric epithelia may also contain carbohydrates and interfere with virus binding to the cell surface [251]. Attachment is attained by the virus binding to a cell surface receptor, usually via electrostatic interactions. Sometimes, co-receptors are also involved, which might promote post-attachment events in the entry process. Early contact between the viral attachment protein and a receptor is weak and reversible. However, as multiple viral attachment proteins interact with multiple receptor molecules, the binding becomes stronger and irreversible [252]. Thus, cells with a higher density or a higher number of receptors are more readily infected [253].
3.2.1. Influenza Virus
As members of Orthomyxoviridae, influenza viruses contain segmented, single-stranded, negative-sense RNAs packaged as ribonucleoproteins in enveloped virions [254,255]. Based on the antigenic diversity of the virion proteins, such as the matrix protein and the nucleocapsid protein, influenza viruses are further classified into three genera: A, B, and C [256,257,258]. Additional classification is based on the haemagglutinin and sialidase glycoproteins expressed on the viral envelope of influenza A and B viruses [258]. Avian influenza A viruses exhibit a strong affinity for α2,3-linked sialic acid as a receptor [258]. As a result, avian viruses tend to bind to Neu5Ac when linked to galactose by an α2,3 bond, while human viruses bind largely to Neu5Ac, linked to galactose via an α2,6 bond [259,260,261].
Usually characterized by annual seasonal epidemics, sporadic pandemic outbreaks involve influenza A virus strains of zoonotic origin [262]. The World Health Organization estimates that annual epidemics of influenza result in approximately 1 billion infections, 3–5 million cases of severe illness, and 300,000–500,000 fatalities [263]. Approximately 10% of the global population is affected by influenza annually, resulting in approximately half a million fatalities annually [264].
Influenza viruses enter cells via endocytosis, by both clathrin-independent and clathrin-dependent pathways [265]. The viral phospholipid bilayer, originating from the host membrane, includes hemagglutinin, which is expressed in the form of spikes on the virion surface [266,267]. Through the clathrin-dependent endocytosis pathway, hemagglutinin binds to sialic acids of host cell surface glycoproteins or glycolipids and mediates virus–host membrane fusion [266,268,269,270]. However, alternative endocytic pathways, such as macropinocytosis, may also be employed [265,271,272].
Furthermore, sialidases are also inserted into the viral phospholipid bilayer as spikes [266,267]. These sialidases are of crucial importance during viral invasion of the ciliated epithelium in human airways [273], specifically, in the removal of decoy receptors on mucins, cilia, and cellular glycocalyx [266,274]. Moreover, sialidase contributes to the pathogenicity of influenza through several additional mechanisms, all which stem from its ability to cleave the α-ketosidic linkage between the terminal sialic acid and the adjacent sugar residue [275]. Notably, influenza sialidase facilitates the release of sialic acid, which can serve as a carbon source for bacteria [273], explaining the frequent superinfection by bacteria following influenza viral infection [276,277].
It is anticipated that influenza vaccines will be introduced in the near future that simultaneously target both the viral hemagglutinin and the viral sialidase [278].
3.2.2. SARS-CoV-2
In response to the rapid global spread of Coronavirus disease 2019 (COVID-19), the World Health Organization (WHO) declared a public health emergency of international interest on 30 January 2020 [279]. This infectious disease is caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and has resulted in many deaths worldwide. By October 31, 2020, over 45 million confirmed COVID-19 cases and 1.2 million deaths had been reported globally [280]. While all coronaviruses infect the respiratory tract, SARS-CoV-2 also damages other organs, like the heart, gastrointestinal tract, liver, kidneys, and central nervous system. This can potentially lead to multi-organ failure [281,282].
The SARS-CoV-2 virus particle is a spherical, enveloped RNA virus containing a single-stranded genome. The viral envelope has an average diameter between 65 and 97 nanometers [281]. Like other coronaviruses, SARS-CoV-2 contains spike S protein trimers protruding from its envelope. The S protein is made up of 1273 amino acids comprising two subunits, S1 and S2. Binding of the S1 subunit to host cell receptors mediates viral entry and infection. The S1 subunit contains two distinct domains: an N-terminal domain and a C-terminal domain [283,284]. The glycan-binding domain located in the N-terminal domain interacts with glycolipids and glycoproteins on the host cell surface [285]. The C-terminal domain contains the receptor-binding domain that attaches to the ACE2 receptor, the primary entry point for SARS-CoV-2 and other coronaviruses [286].
Some coronaviruses are known to bind sialic acids, and SARS-CoV infection might also involve sialic acid interactions [287,288]. Computational predictions, modeling studies, and microscopic analyses indicate that SARS-CoV-2 may bind to sialic acids [289,290]. This binding capacity may explain the extremely high transmissibility of SARS-CoV-2, compared with other human coronaviruses [287,291]. Although gold nanoparticles conjugated with sialic acids have high avidity for the SARS-CoV-2 S1 protein (containing both the N-terminal domain and receptor-binding domain), their binding to model sialylated molecules appears to be relatively weak [292,293]. While numerous reports and reviews [294,295] discuss the potential role of sialic acids as co-receptors for SARS-CoV-2, the specific sialyl structure that is targeted by the spike protein remains unclear [296]. The receptor-binding domain of the SARS-CoV-2 spike protein is known to bind oligosaccharides containing sialic acids. Nonetheless, the preferential interactions seem to be with lipids that enable the virus to enter host cells [293]. Additionally, direct binding between sialic acid-containing oligosaccharides (α2,3 and α2,6 sialyl N-acetyllactosamine) and the N-terminal domain of the SARS-CoV-2 spike glycoprotein has been verified through saturation transfer difference nuclear magnetic resonance spectroscopy [297]. However, the relevance of this interaction for cellular entry still needs to be confirmed. More work is needed for an improved understanding of the infection mechanism and spread of SARS-CoV-2.
3.3. Sialic Acids and Fungi
Fungi are effective degraders of dead organic matter [298] and may impact the human ecosystem [20,299]. Occasionally, fungi may become pathogenic, leading to the development of disease, exploiting the host living tissue for survival [300]. Before that can happen, however, fungal cells must somehow withstand the human immune system defenses. Hence, fungal infections tend to occur in immunocompromised individuals. Despite this, fungi with polysaccharide capsules tend to be more infectious.
3.3.1. Cryptococcus neoformans
Cryptococcus neoformans is used as a model organism illustrating how sialic acids might contribute to the evasion of the human immune response [20,301]. This pathogenic fungus has a true polysaccharide capsule on the outside of its cell wall [302], serving as its major virulence factor [301,303]. Protection from the immune system is aided by sialic acids, and their 9-O-acetylated derivatives, which are a part of the fungal capsular polysaccharides [20,304]. These sialic acids appear to reduce the likelihood of phagocytosis by alveolar macrophages during the initial stages of infection, as the basidiospores are inhaled and gain entry to alveolar spaces [302,305].
3.3.2. Histoplasma capsulatum
Infection with Histoplasma capsulatum results in substantial global morbidity and mortality [306]. As a dimorphic ascomycete, this fungus grows in its hyphal form in the soil and bird and bat droppings [307]. Upon entry into the host, in the form of microconidia or hyphal fragments, H. capsulatum travels through the respiratory system inside phagocytes until it reaches the alveoli [308]. There, the yeast, inside or outside of host phagocytes, is transformed into the pathogenic phase, producing histoplasmosis [296,297]. While invading the host respiratory system, the yeast must evade immune-mediated and intracellular defenses [306]. Meanwhile, these yeast cells need to survive or evade the hostile anti-microbial environment within phagocytes [309,310], including potential exposure to reactive oxygen, such as hydrogen peroxide, and reactive nitrogen species [309,311]. Yeast cells actively inhibit phago-lysosomal fusion, thereby preventing exposure to the acidic hydrolytic enzymes of the lysosomes [308,312]. Moreover, H. capsulatum impedes the accumulation of vacuolar ATPase, which is ordinarily needed for proton accumulation in phagosomes, allowing the fungus to alkalinize the phagosomal pH to 6.5 [309,311,312]. If a suitable environment for growth and reproduction is located, the yeast may also disseminate and develop granulomas [313]. Subsequently, the pathogen is disseminated to the lymph nodes, and then to various organs, by phagocytes as its carriers [306,308,314].
Interactions between carbohydrates and lectins are considered to be pivotal to the recognition of target particles by phagocytes [311,315]. The expression of lectins by pathogenic microorganisms has been correlated to the organisms’ attachment and invasion of host tissues [311,316,317]. H. capsulatum expresses a 50 kDa lectin that recognizes sialic acid residues on laminin, a key protein of the basement membrane, potentially mediating surface interactions [318,319,320].
The identification and isolation of sialic acid-specific lectins from pathogenic fungi, particularly airborne species that cause severe infections, is highly desirable. It represents an opportunity to intervene at the initial stages of infection, as the microconidia interact with lung epithelial cells. If this initial stage is dependent on specific lectin structures, then several options for diagnosis and therapy could become available [320].
3.3.3. Candida albicans
Candida albicans commonly colonizes the skin, the oropharyngeal cavity, the gastrointestinal system, and the vaginal tract [321]. Infections by Candida species are widespread and are increasing in frequency [321,322,323], particularly during infancy or in old age [324,325]. A diverse array of conditions, encompassing environmental, local, and systemic factors, as well as hereditary predispositions, may alter Candida’s normally dormant state [321,325,326]. The signs and symptoms may correspond with a range of morbidities, from localized superficial mucocutaneous disorders to invasive diseases involving multiple organ systems [309,315]. The transition in pathophysiology from the onset through the progression of infection is also influenced by the particular virulence traits that ultimately lead to the development of candidiasis [321,327].
Three types of monosaccharides form carbohydrate chains, or glycans, within the Candida cell wall: d-glucose (Glc), N-acetyl-d-glucosamine (GlcNAc), and d-mannose (Man) [328]. Several reports document sialic acids as constituents of the Candida cell wall [20,304,329]. Sialic acids contribute to the negative charge of fungal cells and have a role in their specific interaction with the host tissue, including a potential role in pathogenicity [329]. Although several microorganisms infecting animals or humans produce sialidases for adhesion or invasion, C. albicans does not, implying that fungal sialidase is likely of no relevance in cutaneous or mucosal candidiasis [330]. Consequently, this implies that sialic acids are likely acquired by C. albicans from the host environment via sialyltransferases. Further investigations are needed into the role of surface sialylation of C. albicans, particularly as it relates to the infectivity of this organism.
3.4. Sialic Acids and Parasites
Parasites are significant contributors to human disease worldwide, particularly in less-developed nations. The clinical severity and outcomes of parasitic diseases are often strongly affected by the host’s immune status [331]. For example, helminths tend to elicit a protective helper T-lymphocyte type 2 host response, with production of IgE, eosinophilia, and mastocytosis [332], whereas protozoa induce a humoral and/or cellular immune response that is not usually associated with eosinophilia [333]. To overcome the immune defenses, parasites employ a variety of mechanisms. These may include (a) becoming “invisible” to the immune system by invading immune-privileged organs or cells, such as the central nervous system, eyes, or blood cells, or (b) decorating surface glycoconjugates with sialic acids, which may be directly synthesized or appropriated from the host [334]. Consequently, the T-cells and antibodies of the vertebrate’s immune system either cannot access the parasite, or do not easily recognize the parasite’s specific antigenic surfaces [335] for effective detection, attack, and subsequent elimination. Such evasive mechanisms are crucial for the parasite’s survival inside the host [159].
3.4.1. Trypanosoma cruzi
Trypanosoma cruzi is a protozoan parasite that causes Chagas disease [336]. In Latin America, nearly 30% of chronically infected people with Chagas disease manifest an intense systemic response that is frequently associated with damage to essential organs, such as the heart or the gastrointestinal tract. This suggests that a wide range of host–parasite interactions affect the course of the infection [163,337].
The protozoan parasite is carried by a triatomine insect [338]. After the infected insect has a blood meal, T. cruzi metacyclic trypomastigotes are discharged via the insect’s feces or urine. Then, the trypomastigotes can infect the mammalian host by penetrating the mucosa, or via discontinuous regions in the epithelium. Once inside the host, the protozoan parasite rapidly infects a variety of nucleated mammalian cells. The attachment to and invasion of host cells, and the formation of the parasitophorous vacuoles with T. cruzi, involves a collection of polymorphic glycosylphosphatidylinositol-anchored surface proteins, including mucins and trans-sialidases [339]. Using its arsenal, the parasite destabilizes both the host’s innate and adaptive immunity [340,341]. Though unable to synthesize sialic acids de novo, T. cruzi uses a unique trans-sialidase enzyme that cleaves terminal sialic acid residues from host cell surface glycoconjugates and transfers them onto parasite surface mucins, generating a protective/adhesive covering [342]. The sialylation of parasite-derived mucins has inhibitory effects on CD4 T cells [341], inducing suppression of their responses. Additionally, T. cruzi sheds trans-sialidase into circulation, as a mechanism of modifying the host’s surface sialic acid signature, exploiting the signaling and functional properties of mammalian host target cells [22]. After lysosomes merge with parasitophorous vacuoles, a complex network of antioxidant enzymes, including superoxide dismutase, shield the microorganism from reactive oxygen and nitrogen species [343]. The acidification associated with the lysosomal contents activates certain critical steps that allow the parasite to escape from the phagosome to the cytoplasm, where it differentiates into the replicative amastigote form. Following numerous duplications and rupture of the host cell membrane, the amastigotes differentiate into infective trypomastigotes that are capable of infecting adjacent cells and being transported by them to distant areas via circulation [343,344]. Based on screening studies, ciprofloxacin binds trans-sialidases, which are essential for T. cruzi survival [345]. This suggests that the antiparasitic effects of quinolones are, at least in part, caused by trans-sialidase inhibition [346].
3.4.2. Plasmodium spp.
Malaria can be caused by several Plasmodium species [347]. According to the most recent World Malaria Report, in 2023, there were 263 million cases, a slight increase from 252 million cases the year before. In 2023, malaria-related deaths were estimated to reach 597,000 [348]. The escalation in malaria cases is primarily attributed to (a) the developing resistance of the parasite to antimalarial medications, and (b) the rapid population growth in the most severely affected regions [347,349,350].
Infection is initiated during a blood meal by an infected Anopheles mosquito, when, along with anticoagulant saliva, 10–100 sporozoites are injected into the host circulation [351,352]. The parasites invade hepatocytes in the liver [353], where they undergo their first replication cycle, producing thousands of merozoites. These merozoites are released into the bloodstream [354], where they infect erythrocytes. The parasite encases itself within a parasitophorous vacuolar membrane and commences remodeling the host cell [355]. These processes entail both internal and external alterations that facilitate the parasite’s survival and proliferation within the red blood cell [356]. During the blood stage, the parasites replicate asexually, maturing into schizonts that undergo multiple rounds of nuclear division to produce merozoites [354,357]. Since red blood cells are not targeted by cytotoxic cells, the parasite is protected from the immune system [354,358]. While digesting host hemoglobin and replicating within the red blood cell, the parasite undergoes expansion and modifies the red blood cell membrane [162,354,358]. Red blood cells infected by P. falciparum become more rigid, whereas those infected by P. vivax become more flexible [352]. At this stage, a fraction of parasites differentiate into sexually competent gametocytes, which can then be transmitted via the mosquito vector [359].
The malaria pathogen has a relatively high selectivity for its respective host [360]. Of the five Plasmodium species that infect humans, P. falciparum and P. vivax are by far the most common and most lethal [361]. Both rely on sialic acids for the establishment of infection, the invasion of red blood cells, and the evasion of the immune system [354,357,362,363]. Reticulocyte invasion by P. vivax, however, is primarily dependent on the reticulocyte Duffy antigen/receptor for chemokines (DARC) [364]. P. falciparum, on the other hand, employs a more direct and critical dependence on erythrocyte sialic acid residues, mediated in part by two distinct sialic acid-binding protein ligands, expressed on the parasite surface and used during red blood cell invasion. These are (a) cysteine-rich protective antigen (PfCyRPA) [365], and (b) antigen 175 (EBA-175) [366,367]. The merozoite surface receptor EBA175 binds to the sialic acid residues of erythrocyte glycophorin A [162,362]. The binding activity of EBA175 is primarily restricted to Region II of the protein. Consequently, this domain is thought to present a potential target for the development of a malaria vaccine [368]. The expression of different forms of EBA and Rh proteins in field parasite isolates, including EBA175, EBA140, EBA181, Rh1, and Rh2, makes such efforts more challenging. Therefore, targeting EBA175 alone by a vaccine against P. falciparum is likely to be insufficient. Developing a multivalent vaccine, incorporating several key blood-stage proteins, should be more promising [368].
4. Inflammatory Processes
Several pathological conditions are characteristically associated with structural and functional changes in sialic acid expression [77]. Such alterations in sialylated glycans can readily be detected in histological sections using plant lectins or antiglycan antibodies [369]. Increased levels of both total and free sialic acid forms in almost any severe condition or inflammatory process are consistent with their potential role as markers of inflammatory disorders [49,56,370,371]. While the rationale supporting the prognostic value of sialic acids in pathological conditions is unclear, some potential explanations have been proposed. These include (a) activation of the hepatocyte-based synthesis and secretion of acute-phase proteins, including several sialoglycoproteins, (b) impaired membrane integrity due to cell damage, and (c) upregulated activities of sialidases and sialyltransferases [371]. These circumstances could lead to measurable changes in sialic acid levels or sialylation patterns, correlating with disease progression.
4.1. Cardiovascular Disease
Cardiovascular disease (CVD), with its behavioral, environmental, cardiometabolic, and social risk factors, represents the leading cause of global morbidity and premature mortality in Western developed countries [372]. Some of the risk factors that could be modified to help prevent or reduce CVD include a lack of adequate physical activity, a sedentary lifestyle, hyperlipidemia, obesity, hypertension, diabetes, and tobacco smoking [373].
Arterial walls comprise several cell types and a specialized extracellular matrix, all of which provide adjustable vascular elasticity and strength [374]. Vascular luminal surfaces are lined with a layer of endothelial cells, which themselves are covered with a dense, heavily sialylated glycocalyx [375]. These sialylated glycoconjugates participate in the regulation of cell–cell interactions for (a) cellular junctions between endothelial cells, and (b) contact between endothelial and circulating blood cells. They also play a role as vascular receptors that modulate the binding of soluble ligands and their signaling [376]. Additionally, sialic acids provide net negative surface charges to vascular endothelial cells and low-density lipoprotein (LDL) receptors. Thus, sialylation, or the covalent addition of sialic acids to the termini of glycoconjugates, and desialylation, or the removal of sialic acid from glycoconjugates, represent a pivotal part of sialic acid metabolism in a variety of physiological and pathological processes [89,377].
Since sialic acids coat all cell surfaces, many circulatory molecules and most molecular complexes, their alteration could have varied effects, potentially including a role in the pathogenesis of CVD [26,148,378]. Desialylation, for example, is recognized as one of the most important steps in the pro-atherogenic pathological cascade [379]. The deposition of cholesterol in cultured human aortic intimal cells is significantly increased when LDLs are treated with sialidase before addition to the culture [379]. The diminished sialic acid content of LDLs leads to increased formation of aggregates, which are readily taken up by smooth muscle cells. Moreover, some free sialic acids, such as the nonhuman Neu5Gc, could also be potential risk factors for CVD [380], or serve as biomarkers for it, as does Neu5Ac [381,382,383]. Neu5Gc accumulates on endothelial surfaces and in atherosclerotic plaques, where anti-Neu5Gc antibodies can trigger endothelial activation and induction of vascular inflammation [384]. Despite such in vitro studies, however, the clinical correlation between serum sialic acids, anti-Neu5Gc antibodies, and their relationship to CVD remains unclear (Table 1).
Comparing the content of sialic acids in the adipose tissue among individuals following habitually vegetarian or non-vegetarian dietary patterns, significantly higher levels of Neu5Ac were found in vegans and lacto-ovo-vegetarians relative to non-vegetarians [39]. These findings could be comparable with other studies reporting intrinsically protective cardiometabolic effects of Neu5Ac or ManNAc supplementation [380,385,386]. Despite this, the beneficial effects of dietary Neu5Ac in vegetarian subjects have not yet been established. However, the positive effects are frequently inferred, given the low risk of cardiovascular disease mortality among these individuals [387].
An excessive accumulation of abdominal adipose tissue is strongly associated with a prognosis of atherosclerosis [388]. Interestingly, it seems that diet could also indirectly affect sialic acids through alterations in BMI or adiposity. For example, KDN levels are lower with increasing BMI, independent of diet, even when comparing individuals following habitually vegetarian or non-vegetarian dietary patterns [39]. These findings are consistent with previous reports of lower sialic acids in obese individuals [386], suggesting that obesity is associated with a higher inflammatory tone. Moreover, in contrast to non-vegetarians, vegetarians have been reported to have a lower BMI and smaller waist circumference [389], which are risk factors for metabolic syndrome, diabetes [390], and cardiovascular disease [387,391]. Therefore, the effects of diet on sialic acids could be related, at least in part, to adiposity. Either way, a plant-based diet promotes a lower BMI and an anti-inflammatory state, both of which appear to be associated with higher sialic acids in adipose tissue. The sialic acids, in turn, could help to regulate or prevent the development of cardiovascular and other chronic diseases. Nevertheless, more detailed epidemiological and clinical studies are necessary to better elucidate the relationships between diet, BMI, and sialic acid levels.
4.2. Cancer
The immune system can be directed in various ways to identify and attack tumors [392]. Sialic acids shield host surfaces by resisting inappropriate immune reactions against self-antigens. This effectively suppresses the immune response. In this context, glycans, including terminal sialic acids, represent self-associated molecular patterns (SAMPs), which are recognized by self-pattern recognition receptors [393]. As a result, sialic acids play a pivotal role in modulating immune reactivity by serving as a self-recognition signal in several ways, including (a) via complement interactions, (b) via alteration of antibody-mediated clearance of pathogens, or (c) via sialic acid-binding immunoglobulin-like lectins (Siglecs) [394]. Consequently, strategies that disrupt sialic acid-mediated immune evasion, such as targeting Siglec interactions or modulating sialoglycan expression, hold promise for enhancing immune recognition and improving cancer therapies.
Table 1. Sialic acids and cardiometabolic diseases.
Lectins represent a family of immunomodulatory receptors located on the surfaces of granulocytes, monocytes, antigen-presenting cells, and natural killer cells. Commonly, Siglecs contain an immunoreceptor tyrosine-based inhibitory motif (ITIM) within the cytoplasmic domain [152,399,400]. A subset of these, Siglecs 14–16, do not possess an ITIM, but instead contain within their transmembrane region a lysine residue, which facilitates interactions with immunoreceptor tyrosine-based activatory motif (ITAM)-containing adaptor proteins [401].
The role of the immune system is unclear during tumor formation and progression, or in the development of resistance to and eradication of emerging neoplasias, late-stage tumors, and micrometastases [402]. The theory of immune surveillance holds that cells and tissues are constantly scanned by the immune system, and that such ever-alert immune surveillance is responsible for recognizing and eliminating most early cancer cells and nascent tumors [403]. The detection of abnormal cells may occur when aberrant surface expression of the sialylated glycans SLeA, SLeX, STn, or GM2 takes place (Figure 6) [91,399,404,405,406]. This can be a result of upregulated activity of sialyltransferases, leading to various surface sialic acids (Table 2) [92,407]. However, once Siglecs are bound to sialylated glycans on cancer cells, immunosuppressive signaling may also be promoted [408,409,410]. For instance, natural killer (NK) cell-mediated tumor cell death is inhibited by interactions between NK-expressed Siglec-7 or Siglec-9, and sialylated glycans, which serve as Siglec ligands on tumor cells [268].
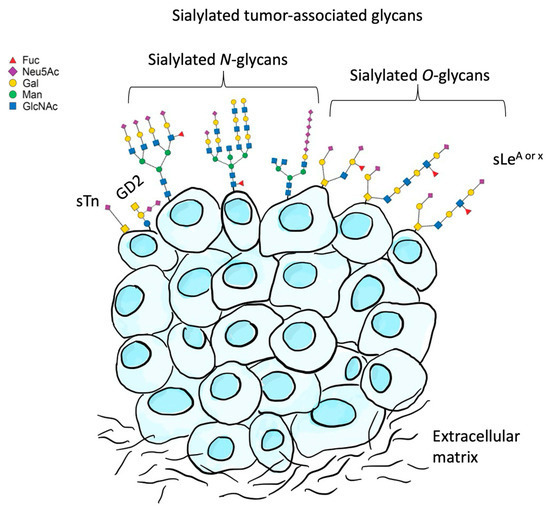
Figure 6. Sialylated glycans in cancer. Tumor cells have increased levels of sialylated glycans on their cell surface, including sialyl LewisA (SLeA), sialyl LewisX (SLeX), and sialyl-Tn (sTn). GD2, di-sialoganglioside.
Table 2. Sialic acid concentrations in human cancer, in different tumor cells.
Through a process of immunoediting [403], tumor cells are selected that mimic the glycosylation patterns of healthy cells using “self”-surface-sialylated glycan signals, as part of evasive strategies [416,417]. This mechanism might represent a potential immune checkpoint for cancer, since immune cells make aberrant sialoglycans prime ligands for Siglecs [418,419]. Thus, it is feasible to target the sialoglycan–Siglec glyco-immune checkpoint by employing Siglec-blocking antibodies. An alternative approach could be to reduce the ligand density by targeting sialoglycans [420]. It has been demonstrated, for example, that when the glyco-profile of a cancer cell is altered by hypersialylation (an increment in sialic acid residues of up to 40–60%) [416], occurring either by the upregulation of sialyltransferases, the downregulation of neuraminidases, or a combination of both, the tumor becomes more aggressive, leading to metastasis and chemoresistance [6,416]. This suggests that downregulation of the surface sialylation of cancer cells may render them less aggressive and more accessible to the immune system.
Colorectal Cancer
Colorectal cancer (CRC) is among the most common cancers and is associated with one of the highest worldwide mortality rates [421]. It is the third most commonly diagnosed cancer among men, and the second most common cancer among women [422]. Numerous studies report associations between dietary factors and total cancer risk [387,423,424,425,426,427]. Diet and nutrition are estimated to explain as much as 30–50% of the worldwide incidence of CRC [428]. Dietary approaches to cancer prevention are of high interest [387], because of their impact as risk modifiers within the multistep process of colorectal tumorigenesis [428]. In this context, vegetarian diets are associated with an overall lower incidence of colorectal cancer [387]. Conversely, consumption of red and processed meats is associated with an increased risk of colon and rectal cancers [429,430,431,432].
Multiple mechanisms explain the potential cancer-promoting effects associated with the consumption of red or processed meats. These include (a) DNA damage due to toxic and mutagenic effects of N-nitroso compounds produced during high-temperature grilling [433], (b) high dietary intake of salt and saturated fats [434], (c) pro-oxidant effects of heme and iron [435], (d) production of trimethylamine (TMA) by the gut microbiome, which is subsequently oxidized by hepatic enzymes to form trimethylamine N-oxide (TMAO) [150,436], (e) altered human gut microbiome composition [153], and (f) increased levels of contaminating cancer-causing viruses [437].
The hypothesis of xenosialitis as an aspect of chronic inflammation is being increasingly discussed [50]. The human body synthetizes its own sialic acids, in addition to exogenously obtained dietary sugars [55,72]. Such dietary sources can include Neu5Gc, especially from foods like red meat, milk, or dairy products [120,438,439]. It has been suggested that Neu5Gc could be a potential aggravating risk factor for carcinoma [436]. The proposed likely mechanism is based on mouse model studies [122,131,432]. The two principal contributors are (a) the fact that humans have a mutation in the gene CMAH, encoding cytidine monophosphate-N-acetylneuraminic acid hydroxylase (Cmah), which converts Neu5Ac to Neu5Gc [122,125,131,440], and (b) the fact that humans can metabolize dietary Neu5Gc and incorporate it during the formation of native human glycans in the place of the usually employed Neu5Ac [131,441]. Thus, multiple instances of potentially immunogenic Neu5Gc-containing glycan neoantigens can be expressed on the colon’s epithelial surfaces [131]. As a result, two different mechanisms may be activated: (a) the canonical NF-κB signaling pathway, which regulates the transcription of inflammatory factors, and (b) the in situ formation of antigen–antibody immunocomplexes [122]. Both processes promote systemic inflammation and subsequent tissue damage [442]. Consequently, each of these could explain the link between a red meat-rich diet and colon carcinoma.
This xenosialitis hypothesis was tested, with some mixed results. Since polyclonal rabbit anti-T cell IgG (ATG) is known to elicit an immune response in humans toward Neu5Gc [443], allograft recipients treated with animal-derived IgG were tested. Yet, in one study, no evidence was found that exposure to higher anti-Neu5Gc levels is associated with a higher incidence of colon carcinoma [53], possibly due to low cohort numbers. In contrast, in another study of 71 subjects, a positive association between total anti-Neu5Gc and CRC risk was reported [150]. However, a significant association between anti-Neu5Gc IgG and dietary red meat intake has not yet been demonstrated [150]. Interestingly, a different study reported a clear link between the levels and repertoire of serum anti-Neu5Gc IgG and the Neu5Gc from dietary red meat and dairy consumption [131]. Further, adequately powered investigations are needed to elucidate the links between diet, Neu5Gc, anti-Neu5Gc, and the risks of chronic diseases.
5. Conclusions
Sialic acids are versatile nine-carbon carbohydrates that play essential roles in human health and disease. Given their abundance on cell surfaces and secreted proteins, they mediate critical processes, such as facilitating cell–cell interactions, signaling mechanisms, immune responses, and developmental processes. Pathogens frequently exploit sialic acids to mimic host surfaces and evade immune responses. Conversely, changes in host sialylation patterns are known to be associated with inflammation, cardiovascular diseases, and cancers. Advances in analytical techniques, particularly mass spectrometry, have expanded understanding of sialic acid biology. Further investigations are needed to elucidate the diverse roles of sialic acids in physiologic and pathologic processes, in order to enable targeted therapeutic interventions.
Author Contributions
Conceptualization, D.S.B., G.N.G.-F. and F.J.P.; methodology, D.S.B. and G.N.G.-F.; writing original draft and preparation, D.S.B., G.N.G.-F. and F.J.P.; writing review and editing, D.S.B., G.N.G.-F., F.J.P., F.M.B., G.Z. and V.L.M.M.; visualization, G.N.G.-F. and D.S.B.; supervision, D.S.B. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by: (a) Grant for Research and School Partnerships (GRASP), Loma Linda University (#2170258), California, USA; (b) Doctoral scholarship to GNGF, Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Ministry of Science and Technology, Argentina (RESOL-2018-2704-APN-DIR#CONICET); (c) Research grant, School of Medicine, Adventist University of River Plate, Entre Rios, Argentina.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| ADP | adenosine diphosphate |
| ATP | adenosine triphosphate |
| CMAS | CMP-N-acetylneuraminate synthetase |
| Cmah | cytidine monophosphate-N-acetylneuraminic acid hydroxylase; CMP |
| CMP | cytidine monophosphate |
| CTP | cytidine triphosphate |
| GalNAc | N-acetylgalactosamine |
| GlcNAc | N-acetylglucosamine |
| GNE | UDP-GlcNAc 2-epimerase/ManNAc kinase |
| GD2 | disialoganglioside |
| GM2 | ganglioside-mono sialic acid 2 |
| GlmM | phosphoglucosamine mutase |
| GlmS | glucosamine-6-phosphate synthase |
| GlmU | glucosamine-1-phosphate acetyltransferase/N-acetylglucosamine-1-phosphate uridyltransferase |
| HBP | hexosamine biosynthesis pathway |
| ITAM | immunoreceptor tyrosine-based activatory motif |
| ITIM | immunoreceptor tyrosine-based inhibitory motif |
| KDN | 2-keto-3-deoxy-D-glycero-D-galacto-nononic acid |
| LC-MS/MS | liquid chromatography with tandem mass spectrometry |
| Man | mannose |
| ManNAc | N-acetylmannosamine |
| MS | mass spectrometry |
| NANP | N-acetylneuraminic acid phosphatase |
| NANS | N-acetylneuraminic acid 9-phosphate synthase |
| Neu | neuraminic acid |
| Neu5Ac | N-acetylneuraminic acid |
| Neu5Gc | N-glycolylneuraminic acid |
| NeuA | sialic acid synthase A |
| NeuB | sialic acid synthase B |
| NeuC | sialic acid synthase C |
| NulO | nonulosonic acid |
| PEP | phosphoenolpyruvate |
| PGI | glucose-6-phosphate isomerase |
| ROS | reactive oxygen species |
| SLeA | sialyl-Lewis A |
| SLeX | sialyl-Lewis X |
| STn | sialyl-Tn |
| SCFAs | short-chain fatty acids |
| UDP | uridine diphosphate |
| SARS-CoV-2 | severe-acute-respiratory-syndrome-related coronavirus 2. |
References
- Tarbell, J.M.; Cancel, L.M. The glycocalyx and its significance in human medicine. J. Intern. Med. 2016, 280, 97–113. [Google Scholar] [CrossRef]