예를들어 신경전달물질의 신호를 증강시키는 항우울제를 투여했다. 그 효과가 2-4주 걸린다. 이유는?
남성호르몬(테스토스테론)이 분비되는 강력한 근력운동을 했다. 그 효과가 6개월이 걸린다. 그 이유는?
신경전달물질, 호르몬을 투여한 후 바로 효과가 나지 않고 시간차가 필요하다.
그 기전에 대한 탐구다.
그 메카니즘을 이해하기 위해서
autoreceptor and heteroreceptor의 이해가 필요하다.
An autoreceptor is a type of receptor located in the membranes of presynaptic nerve cells. It serves as part of a negative feedback loop in signal transduction. It is only sensitive to the neurotransmitters or hormones released by the neuron on which the autoreceptor sits. Similarly, a heteroreceptor is sensitive to neurotransmitters and hormones that are not released by the cell on which it sits. A given receptor can act as either an autoreceptor or a heteroreceptor, depending upon the type of transmitter released by the cell on which it is embedded.
자가수용체는 presynaptic 신경세포 막에 위치하는 수용체의 한 형태이다. 자가 수용체는 signal transduction에서 음성 되먹이기 루프의 일부로 작용한다. 그것은 오직 자가수용체 부위의 신경에 의해서 분비되는 신경전달물질이나 호르몬에 민감하다. 마찬가지로 타가수용체도 신경전달물질과 호르몬에 민감한데, 이것은 그 자체세포에서 분비하지 않는 호르몬과 신경전달물질에 민감하다. 주어진 수용체는 그것을 둘러싼 세포에 의해서 분비되는 신경전달물질의 형태에 의존하여 자가 수용체 또는 타가수용체에 작용할 수 있다.
참고) Heteroreceptor
A heteroreceptor is a receptor regulating the synthesis and/or the release of mediators other than its own ligand.[1]
Heteroreceptors are receptors that respond to neurotransmitters, neuromodulators, or neurohormones released from adjacent neurons or cells; they are opposite to autoreceptors, which are sensitive only to neurotransmitters or hormones released by the cell in whose wall they are embedded.[2]
타가수용체는 그 자신의 ligand보다는 합성 또는 매개체의 분비를 조절하는 수용체다. 타가수용체는 세포나 신경주위에서 분비되는 신경전달물질, neuromodulator, 신경호르몬에 반응는 수용체다. 타가수용체는 자가수용체와 반대되는 용어다.
Examples[edit]
- Norepinephrine can influence the release of acetylcholine from parasympathetic neurons by acting on α2 adrenergic (α2A, α2B, and α2C) heteroreceptors.[3]
- Acetylcholine can influence the release of norepinephrine from sympathetic neurons by acting on muscarinic-2 and muscarinic-4 heteroreceptors.
- CB1 negatively modulates the release of GABA[4][5] and glutamate,[6] playing a crucial role in maintaining a homeostasis between excitatory and inhibitory transmission.
- Glutamate released from excitatory neuron escapes synaptic cleft and preferentially effects mGluR III receptors on presynaptic terminal of interneurons. Glutamate spillover leads to inhibition of GABA release, modulating GABAergic transmission.[7][8]
Autoreceptors may be located in any part of the cell membrane: in the dendrites, the cell body, the axon, or the axon terminals.[1] Canonically, a presynaptic neuron releases a neurotransmitter across a synaptic cleft to be detected by the receptors on a postsynaptic neuron. Autoreceptors on the presynaptic neuron will also detect this neurotransmitter and often function to control internal cell processes, typically inhibiting further release or synthesis of the neurotransmitter. Thus, release of neurotransmitter is regulated by negative feedback. Autoreceptors are usually G protein-coupled receptors (rather than transmitter-gated ion channels) and act via a second messenger.[2]
자가수용체는 수상돌기, 신경세포체, 액손, axon terminal 세포 벽의 일부에 존재할 수 있다. 시냅스 전 신경세포는 postsynaptic 신경의 수용체에 의해 발견되는 synaptic cleft를 가로질러 신경전달물질을 분비한다. postsynaptic 신경세포의 자가수용체는 이러한 신경전달물질을 발견할 수 있고, 때로는 internal cell process 조절기능을 하는 .. 전형적으로는 신경전달물질의 합성이나 분비를 억제함으로서...그래서 신경전달물질의 분비는 음성되먹이기에 의해서 조절된다. 자가 수용체는 일반적으로 g protein-coupled 수용체(transmitter-gated ion channels)이고 second messenger를 통해 작용한다.
Examples[edit]
As an example, norepinephrine released from sympathetic neurons may interact with the alpha-2A and alpha-2C adrenoreceptors to inhibit further release of norepinephrine. Similarly, acetylcholine released from parasympathetic neurons may interact with M2 and M4 receptors to inhibit further release of acetylcholine. An atypical example is given by the β-adrenergic autoreceptor in the sympathetic peripheral nervous system, which acts to increase transmitter release.[1]
예를들어, 교감신경으로 부터 분비되는 노르에피네프린은 ...
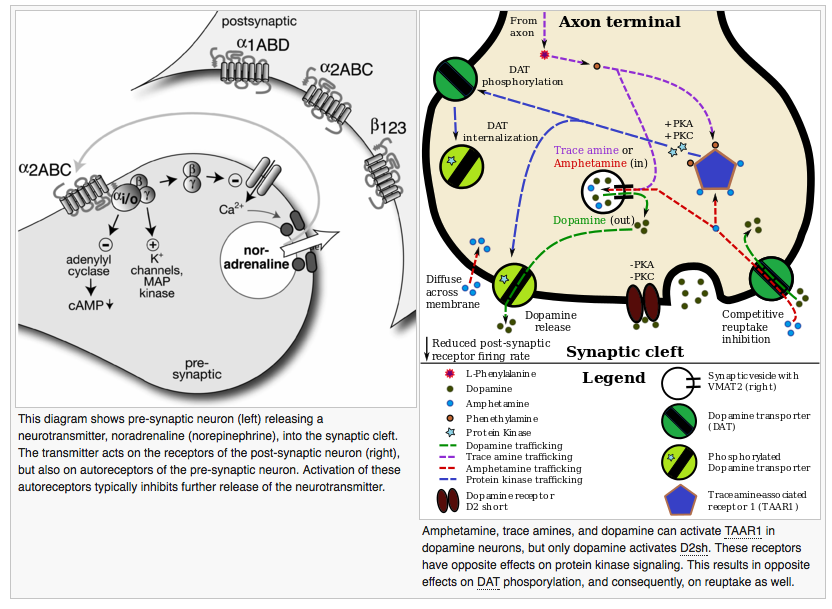
The D2sh autoreceptor interacts with the trace amine-assorted receptor 1 (TAAR1), a recently discovered GPCR, to regulate monoaminergic systems in the brain.[3] Active TAAR1 opposes the autoreceptor's activity by inactivating the dopamine transporter (DAT).[4] In their review of TAAR1 in monoaminergic systems, Xie and Miller proposed this schematic: synaptic dopamine binds to the dopamine autoreceptor, which activates the DAT. Dopamine enters the presynaptic cells and binds to TAAR1, which increases adenylyl cyclase activity. This eventually allows for the translation of trace amines in the cytoplasm and activation of cyclic nucleotide-gated ion channels, which further activate TAAR1 and dump dopamine into the synapse. Through a series of phosphorylation events related to PKA and PKC, active TAAR1 inactivates DAT, preventing uptake of dopamine from the synapse.[5] The presence of two presynaptic receptors with opposite abilities to regulate monoamine transporter function allows for regulation of the monoaminergic system.
Autoreceptor activity may also decrease paired-pulse facilitation (PPF).[citation needed] A feedback cell is activated by the (partially) depolarized post-synaptic neuron. The feedback cell releases a neurotransmitter to which the autoreceptor of the presynaptic neuron is receptive. The autoreceptor causes the inhibition of calcium channels (slowing calcium ion influx) and the opening of potassium channels (increasing potassium ion efflux) in the presynaptic membrane. These changes in ion concentration effectively diminish the amount of the original neurotransmitter released by the presynaptic terminal into the synaptic cleft. This causes a final depression on the activity of the postsynaptic neuron. Thus the feedback cycle is complete.