황화수소(H₂S)는 인체에서 중요한 내인성 가스 전달 물질로 점점 더 많이 인식되고 있으며, 개념적으로 산화질소(NO) 및 일산화탄소(CO)와 유사합니다. 대학원 수준에서, 사람들은 이 물질이 갖는 다각적인 역할, 조절 메커니즘, 그리고 잠재적인 치료적 응용 가능성을 이해할 수 있습니다. 아래는 인간 생리학에서 H₂S의 효능과 기능적 중요성에 대한 포괄적인 설명입니다.
────────────────────────────
1. 내생적 생산과 대사 경로
- 생합성: H₂S는 주로 다음의 세 가지 효소에 의해 내생적으로 생산됩니다.
- 규정: H2S의 생산은 기질 가용성, 효소 발현 수준, 번역 후 변형에 의해 엄격하게 규제됩니다. 다양한 생리적 및 병리학적 자극이 이러한 효소를 조절하여 H2S 수준을 변화시킬 수 있습니다.
────────────────────────────
2. 생리적 기능과 메커니즘
- 혈관 확장 및 심혈관 조절:
- 세포 보호 및 항산화 스트레스:
- 항염 효과:
- 미토콘드리아 기능 및 에너지 대사:
- 신경 보호:
────────────────────────────
3. 치료적 잠재력과 임상적 시사점
- 심혈관 질환: 임상 연구에서 고혈압, 죽상 동맥 경화증, 허혈-재관류 손상 등의 보조 요법으로 H₂S 기증자(예: NaHS, GYY4137)의 사용을 평가하기 시작했습니다. 목표는 H₂S의 혈관 확장 및 항염 특성을 활용하여 심장 기능을 개선하고 경색 크기를 줄이는 것입니다.
- 신경 퇴행성 질환: 신경 보호 역할을 하는 H₂S는 알츠하이머병과 파킨슨병과 같은 질병의 치료 가능성에 대한 연구가 진행되고 있습니다. 연구의 초점은 독성 없이 유익한 효과를 얻기 위해 전달 방법과 투여 요법을 최적화하는 데 맞춰져 있습니다.
- 염증 및 대사 장애: H₂S의 항염증 및 항산화 특성은 류마티스 관절염과 당뇨병 관련 합병증과 같은 만성 염증이 특징인 질환에 대한 유망한 후보 물질입니다.
- 약물 전달 및 표적화: 연구자들은 H₂S의 지속적이고 통제된 방출을 달성하기 위해 서방성 H₂S 전달 물질과 나노입자 기반 전달 시스템을 개발하고 있습니다. 이를 통해 치료 효과를 향상시키면서 부작용을 최소화할 수 있습니다.
────────────────────────────
4. 도전과제와 미래 방향
- 용량 의존성 및 독성: H₂S의 생리적 농도 범위와 병적 농도 범위는 좁습니다. 낮은 농도에서는 H₂S가 보호 작용을 하지만, 높은 농도에서는 세포 독성을 일으키고 세포 호흡을 억제할 수 있습니다.
- 측정 및 바이오마커: H₂S는 휘발성과 빠른 대사 작용 때문에 생물학적 시스템에서 신뢰할 수 있는 정량화가 어렵습니다. H₂S 수준과 임상 결과 사이의 상관 관계를 더 잘 파악하기 위해서는 개선된 탐지 방법이 필요합니다.
- 임상 시험: 전임상 연구가 유망하지만, H₂S 기반 치료의 안전성과 효과를 입증하기 위해서는 대규모의 통제된 임상 시험이 필요합니다.
────────────────────────────
결론
황화수소(H₂S)는 인간 생리학에서 중요한 역할을 하며, 혈관 확장, 항산화, 항염증, 신경 보호 효과를 발휘하는 가스 전달 물질로 작용합니다. 내인성 생산과 엄격하게 조절되는 농도는 항상성 유지에 필수적입니다. H₂S가 다양한 질환, 특히 심혈관 질환과 신경 퇴행성 질환에 상당한 치료 효과를 보인다는 사실이 밝혀졌지만, 최적의 치료 기간을 정의하고, 전달 시스템을 개선하며, 임상 적용에서 장기적인 안전성을 확립하기 위해서는 추가 연구가 필요합니다.
H₂S에 대한 이러한 심층적인 이해는 새로운 치료법을 모색하고 건강과 질병에서 가스 전달 물질 매개 신호에 대한 지식을 심화할 수 있는 틀을 제공합니다.

Biosci Rep
. 2022 Sep 14;42(9):BSR20221006. doi: 10.1042/BSR20221006
Reactive sulfur species and their significance in health and disease
Małgorzata Iciek 1,✉, Anna Bilska-Wilkosz 1, Michał Kozdrowicki 1, Magdalena Górny 1
- Author information
- Article notes
- Copyright and License information
PMCID: PMC9484011 PMID: 36039860
Abstract
Reactive sulfur species (RSS) have been recognized in the last two decades as very important molecules in redox regulation. They are involved in metabolic processes and, in this way, they are responsible for maintenance of health. This review summarizes current information about the essential biological RSS, including H2S, low molecular weight persulfides, protein persulfides as well as organic and inorganic polysulfides, their synthesis, catabolism and chemical reactivity. Moreover, the role of RSS disturbances in various pathologies including vascular diseases, chronic kidney diseases, diabetes mellitus Type 2, neurological diseases, obesity, chronic obstructive pulmonary disease and in the most current problem of COVID-19 is presented. The significance of RSS in aging is also mentioned. Finally, the possibilities of using the precursors of various forms of RSS for therapeutic purposes are discussed.
Abstract
반응성 황 종(RSS)은
지난 20년 동안 산화 환원 조절에서
매우 중요한 분자로 인식되어 왔습니다.
이들은 신진대사 과정에 관여하여
건강 유지에 중요한 역할을 합니다.
이 리뷰에서는
H2S,
저분자량 퍼설파이드,
단백질 퍼설파이드,
유기 및 무기 다황화물,
이들의 합성, 이화 작용 및 화학 반응성을 포함한
필수 생물학적 RSS에 대한 최신 정보를 요약합니다.
또한
혈관 질환, 만성 신장 질환, 제 2형 당뇨병, 신경 질환, 비만, 만성 폐쇄성 폐 질환 및
가장 최근의 문제인 COVID-19를 포함한
다양한 병리에서 RSS 장애의 역할이 제시됩니다.
노화에서
RSS의 중요성에 대해서도 언급합니다.
마지막으로
다양한 형태의 RSS 전구체를
치료 목적으로 사용할 수 있는 가능성에 대해 논의합니다.
Keywords: hydrogen sulfide, persulfides, polysulfides, protein persulfidation, reactive sulfur species
Introduction
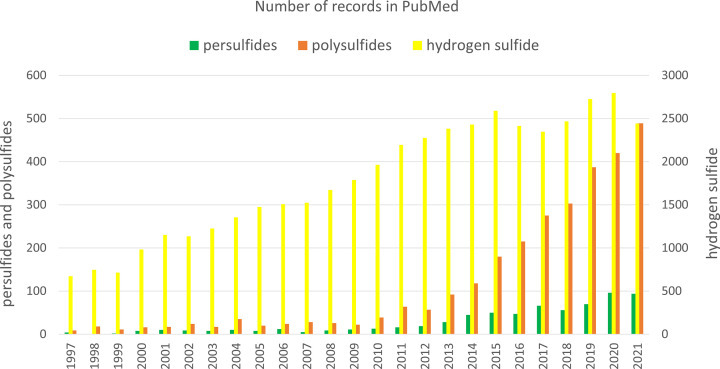
Sulfur is an essential element for living organisms and one of the most abundant elements on earth. Plants and numerous microorganisms absorb sulfur from the soil in the form of sulfate (SO42−), which is then reduced to S2− and incorporated into sulfur amino acids, methionine and cysteine [1]. In animals, sulfur is absorbed from the digestive tract in the form of sulfur amino acids, of which methionine is an essential amino acid, while cysteine can be produced endogenously from methionine; therefore, it is not an essential amino acid. Reactive sulfur species (RSS), including hydrogen sulfide (H2S), persulfides and polysulfides are synthetized in all living organisms mainly from cysteine and have been recognized in the last two decades as very important molecules in redox regulation. They participate in sulfur trafficking and are involved in metabolic processes and in this way, they are responsible for maintenance of health. The important role of RSS in the regulation of biological processes is reflected by the expanding interest of scientists and the ever-growing number of studies on individual biologically active forms of sulfur. Figure 1 shows the number of records in PubMed for the search terms: H2S, persulfides and polysulfides published over the last 25 years. Since 1997, there has been a steady increase in interest in all these RSS forms that proves that this topic is up-to-date and offers opportunities for new discoveries.
소개
유황은
생명체에 필수적인 원소이자
지구상에서 가장 풍부한 원소 중 하나입니다.
식물과 수많은 미생물은
토양에서 황산염(SO42-)의 형태로 황을 흡수한 다음,
황산염이 S2-로 환원되어
황 아미노산, 메티오닌 및 시스테인으로 통합됩니다[1].
sulfate (SO42−)
sulfur amino acids, methionine and cysteine
동물에서 황은
소화관에서 황 아미노산의 형태로 흡수되며,
이 중 메티오닌은 필수 아미노산인 반면
시스테인은 메티오닌에서 내인성적으로 생산될 수 있으므로
필수 아미노산이 아닙니다.
황화수소(H2S), 퍼설파이드 및 폴리설파이드를 포함한
반응성 황 종(RSS)은
주로 시스테인으로부터 모든 생물체에서 합성되며
지난 20년 동안 산화 환원 조절에 매우 중요한 분자로 인식되어 왔습니다.
이들은 유황 거래에 참여하고 대사 과정에 관여하며
이러한 방식으로 건강 유지에 책임이 있습니다.
생물학적 과정의 조절에서 RSS의 중요한 역할은
과학자들의 관심이 확대되고
개별 생물학적 활성 형태의 황에 대한 연구가 계속 증가하고 있다는 사실에
반영되어 있습니다.
그림 1은 PubMed에서 검색어에 대한 기록 수를 보여줍니다: 지난 25년 동안 출판된 H2S, 퍼설파이드 및 폴리설파이드 검색어에 대한 기록 수를 보여줍니다. 1997년 이후 이러한 모든 RSS 형태에 대한 관심이 꾸준히 증가하여 이 주제가 최신 주제이며 새로운 발견의 기회를 제공한다는 것을 증명하고 있습니다.
Figure 1. The number of records in PubMed obtained by searching the database for RSS that included H2S (yellow), persulfides (green) and polysulfides (brown) published over the last 25 years.
There are many evidence that the disruption of RSS homeostasis leads to some pathologies. At the same time, it was also shown that the modulation of RSS levels in the cell by using their precursors could be a potential and promising therapeutic tool. Recently, the significance of RSS has also been investigated in relation to the infection caused by SARS-CoV-2 virus, and some potential donors or activators of endogenous RSS synthesis are tested as a preventive treatment in COVID-19.
RSS 항상성 파괴가 일부 병리를 유발한다는 많은 증거가 있습니다. 동시에, 전구체를 사용하여 세포의 RSS 수준을 조절하는 것이 잠재적이고 유망한 치료 도구가 될 수 있다는 사실도 밝혀졌습니다. 최근에는 SARS-CoV-2 바이러스로 인한 감염과 관련하여 RSS의 중요성도 조사되었으며, COVID-19의 예방 치료제로 내인성 RSS 합성의 일부 잠재적 기증자 또는 활성화제가 테스트되고 있습니다.
H2S and its properties
Hydrogen sulfide (H2S) is probably the best known form of RSS. The story of H2S as a physiological regulator started in the 1990s, when Kimura et al. documented that this small gaseous molecule was produced endogenously in the nervous system, where it fulfilled an important role [2]. After that many other studies have found that H2S is synthesized also in other animal tissues and its physiological role has been widely discussed [3–5]. This interest in the biological significance of H2S and research on its endogenous synthesis and catabolism was reflected in a large increase in the number of publications regarding this topic (Figure 1).
However, the beginning of discoveries related to endogenous H2S dates back 50 years earlier, when the transsulfuration pathway, involving the interconversion of cysteine and homocysteine, through the intermediate cystathionine, was discovered in animal liver and other tissues. During that study, the authors described the production of H2S, but they did not recognize the biological potential and significance of this gaseous mediator [6].
황화수소(H2S)는
아마도 RSS의 가장 잘 알려진 형태일 것입니다.
생리 조절 물질로서의 H2S에 대한 이야기는
1990년대에 시작되었는데,
Kimura 등이 이 작은 기체 분자가
신경계에서 내인성적으로 생성되어 중요한 역할을 수행한다는 사실을 문서화했습니다[2].
그 후 많은 다른 연구에서
H2S가 다른 동물 조직에서도 합성된다는 사실이 밝혀졌고
그 생리적 역할이 널리 논의되었습니다 [3-5].
H2S의 생물학적 중요성과 내인성 합성 및 이화 작용에 대한 연구에 대한 이러한 관심은
이 주제와 관련된 출판물의 수가 크게 증가한 데 반영되었습니다(그림 1).
그러나
내인성 H2S와 관련된 발견의 시작은
50년 전 동물의 간 및 기타 조직에서
시스테인과 호모시스테인이 중간 시스타티오닌을 통해 상호 전환되는
트랜스황화 경로가 발견되었을 때로 거슬러 올라갑니다.
이 연구에서 저자들은
H2S의 생성에 대해 설명했지만,
이 가스 매개체의 생물학적 잠재력과 중요성을 인식하지 못했습니다[6].
H2S is the one of the three well-known gasotransmitters. The feature that distinguishes it from other gasotransmitters (carbon monoxide, CO and nitric oxide, NO) is its characteristic odor and ability to dissociate. As a weak acid, H2S dissociates in two stages to hydrosulfide anion (HS−) and sulfide anion (S2−) (reaction 1):
H2S는
잘 알려진 세 가지 가스 전달 물질 중 하나입니다.
다른 가스 전달 물질(일산화탄소, CO 및 산화질소, NO)과 구별되는 특징은
특징적인 냄새와 해리 능력입니다.
약산인 H2S는
두 단계로 해리되어
하이드로설파이드 음이온(HS-)과 황화물 음이온(S2-)으로 분해됩니다(반응 1):
| H2S⇄HS−⇄S2− | (1) |
Deprotonation of H2S to form HS− has a pKa≈7.0, while the second pKa value is 13.1. It means that under physiological pH in extracellular fluids (e.g. plasma), H2S exists predominantly as HS− (80%), concentration of S2− is negligible while H2S accounts for nearly 20%. Inside the cell, in the cytosol, where pH value is a little lower (≈7.0), approximately 50% of H2S exists as HS− anion. The proportion of the different ionized forms of H2S may vary depending on the prevailing pH in the specific cell organelles. In mitochondria, where the pH is above 7.8, the concentration of the HS− has been estimated to represent approximately 92%. On the other hand, in lysosomes, where the environment is acidic (pH 4.7), nearly 99% of H2S is present in the undissociated form and the HS− anion content is below 1% [7]. H2S, as a more hydrophobic molecule, freely penetrates the lipid bilayer of the cell membrane by simple diffusion. HS− anion is more hydrophilic, so it is transported by facilitated diffusion and requires special transporters, such as anion exchange protein AE1 present in erythrocytes [8]. It is assumed that the term hydrogen sulfide (H2S) is used to describe all forms of H2S existing in a pH-dependent equilibrium in the cellular environment. In this review, similarly, the term covers all forms of H2S.
HS-를 형성하기 위한 H2S의 탈프로토레이션은 pKa≈7.0이고, 두 번째 pKa 값은 13.1입니다.
이는
세포 외액(예: 혈장)의 생리적 pH에서
H2S는 주로 HS-(80%)로 존재하고,
S2-의 농도는 무시할 수 있는 반면
H2S는 거의 20%를 차지한다는 것을 의미합니다.
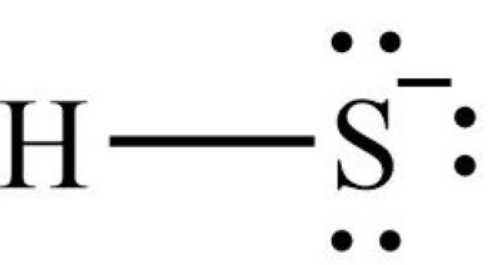
세포 내부,
pH 값이 약간 낮은(≈7.0) 세포질에서는
H2S의 약 50%가 HS- 음이온으로 존재합니다.
다른 이온화된 형태의 H2S의 비율은
특정 세포 소기관의 일반적인 pH에 따라 달라질 수 있습니다.
pH가 7.8 이상인 미토콘드리아에서는
HS-의 농도가 약 92%를 차지하는 것으로 추정됩니다.
반면,
환경이 산성(pH 4.7)인 리소좀에서는
거의 99%의 H2S가 해리되지 않은 형태로 존재하고
HS- 음이온 함량은 1% 미만입니다[7].
소수성이 강한 분자인 H2S는
단순한 확산을 통해 세포막의 지질 이중층을 자유롭게 투과합니다.
HS- 음이온은 더 친수성이기 때문에
촉진 확산에 의해 운반되며
적혈구에 존재하는 음이온 교환 단백질 AE1과 같은 특수 수송체가 필요합니다 [8].
황화수소(H2S)라는 용어는
세포 환경에서 pH 의존적 평형 상태에 존재하는 모든 형태의 H2S를 설명하는 데
사용된다고 가정합니다.
이 리뷰에서도 마찬가지로 이 용어는
모든 형태의 H2S를 포괄합니다.
Sources of endogenous H2S
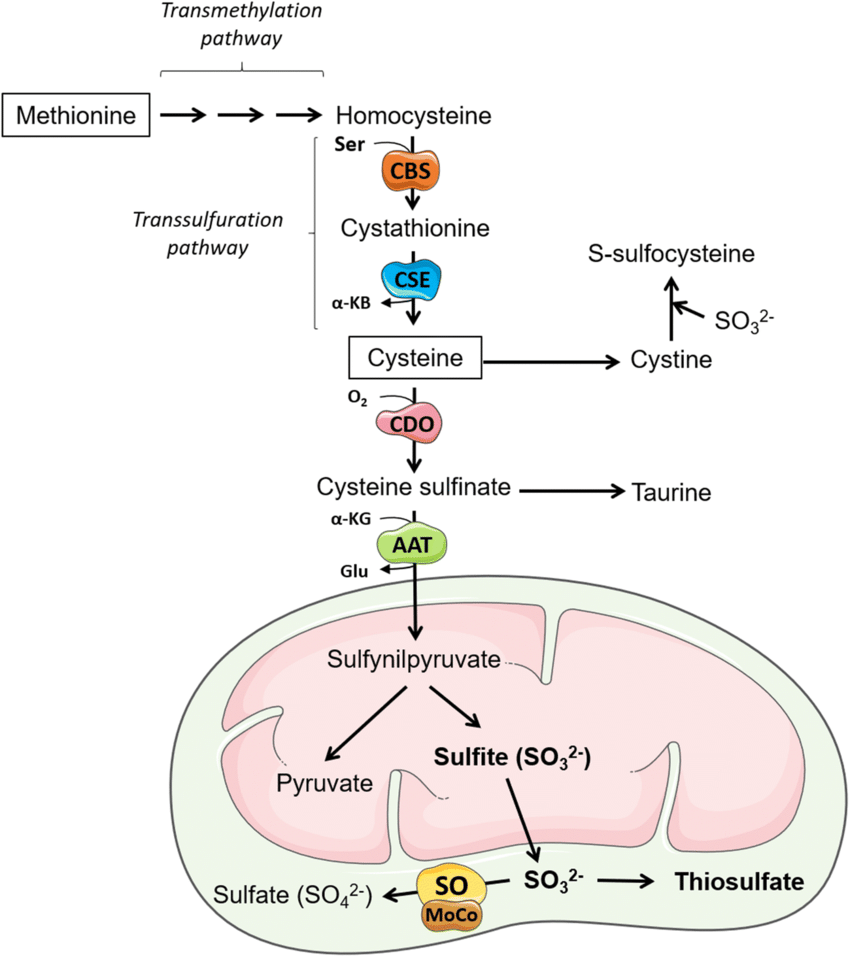
H2S is produced endogenously in mammalian cells mainly from L-cysteine (Cys-SH) and this process is catalyzed by three enzymes: cystathionine β-synthase (CBS), cystathionine γ-lyase (also known as cystathionase, CSE) and 3-mercaptopyruvate sulfurtransferase (MST). CBS and CSE are pyridoxal phosphate (PLP) dependent and are localized in the cytosol whereas MST is PLP independent and resides in mitochondria. CBS and CSE participate in the transsulfuration pathway that converts L-homocysteine (derived from methionine) into Cys-SH (Figure 2). Moreover, these two enzymes are responsible for H2S generation in many additional reactions (Figure 3).
내인성 H2S의 공급원
H2S는 포유류 세포에서
주로 L-시스테인(Cys-SH)에서 내인성적으로 생성되며
이 과정은
시스타티오닌 β-신타제(CBS),
시스타티오닌 γ-리아제(시스타티오나제, CSE라고도 함) 및
3-mercaptopyruvate sulfurtransferase(MST)의 3가지 효소에 의해
촉매 작용을 거칩니다.
CBS와 CSE는
피리독살 인산염(PLP)에 의존적이며 세포질에 국한되어 있는 반면,
MST는
PLP에 독립적이며 미토콘드리아에 존재합니다.
CBS와 CSE는
L-호모시스테인(메티오닌에서 유래)을 Cys-SH로 전환하는 트랜스설퍼레이션 경로에 참여합니다(그림 2).
또한 이 두 효소는 여러 추가 반응에서 H2S 생성을 담당합니다(그림 3).
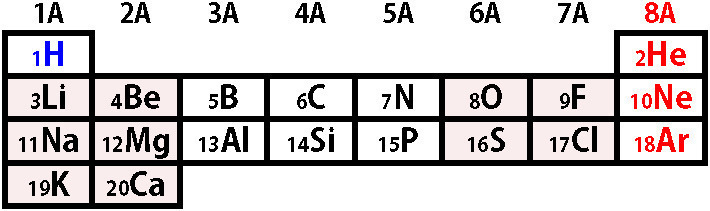
Figure 2. Biosynthetic pathway leading to H2S production from L- and D-cysteine.
L-cysteine is synthesized endogenously from methionine. It is converted to L-homocysteine that reacts with L-serine forming cystathionine in the reaction catalyzed by CBS. Cystathionine is then cleaved by CSE to L-cysteine and L-homoserine. L-cysteine is a substrate for reactions leading to H2S generation presented in Figure 3. L-cysteine can be also a substrate for CAT, which results in the formation of 3-MP. The latter compound is a substrate for MST that temporarily creates a persulfide form and then H2S is released under the influence of reducing agents: Trxred or DHLA. The pathway of H2S synthesis from D-cysteine exists mainly in peroxisomes, where DAO converts it to 3-MP.
L-시스테인은 메티오닌에서 내인성적으로 합성됩니다. 이는 CBS에 의해 촉매되는 반응에서 L-세린과 반응하여 시스타티오닌을 형성하는 L-호모시스테인으로 전환됩니다. 그런 다음 시스타티오닌은 CSE에 의해 L-시스테인 및 L-호모시스테인으로 분해됩니다. L-시스테인은 그림 3에 제시된 H2S 생성으로 이어지는 반응의 기질입니다. L-시스테인은 또한 3-MP를 형성하는 CAT의 기질이 될 수 있습니다. 후자의 화합물은 일시적으로 퍼설파이드 형태를 생성한 다음 환원제의 영향으로 H2S가 방출되는 MST의 기질입니다: Trxred 또는 DHLA. D-시스테인에서 H2S가 합성되는 경로는 주로 퍼옥시좀에 존재하며, DAO가 이를 3-MP로 전환합니다.
Figure 3. Specific reactions catalyzed by CBS and CSE responsible for H2S generation.
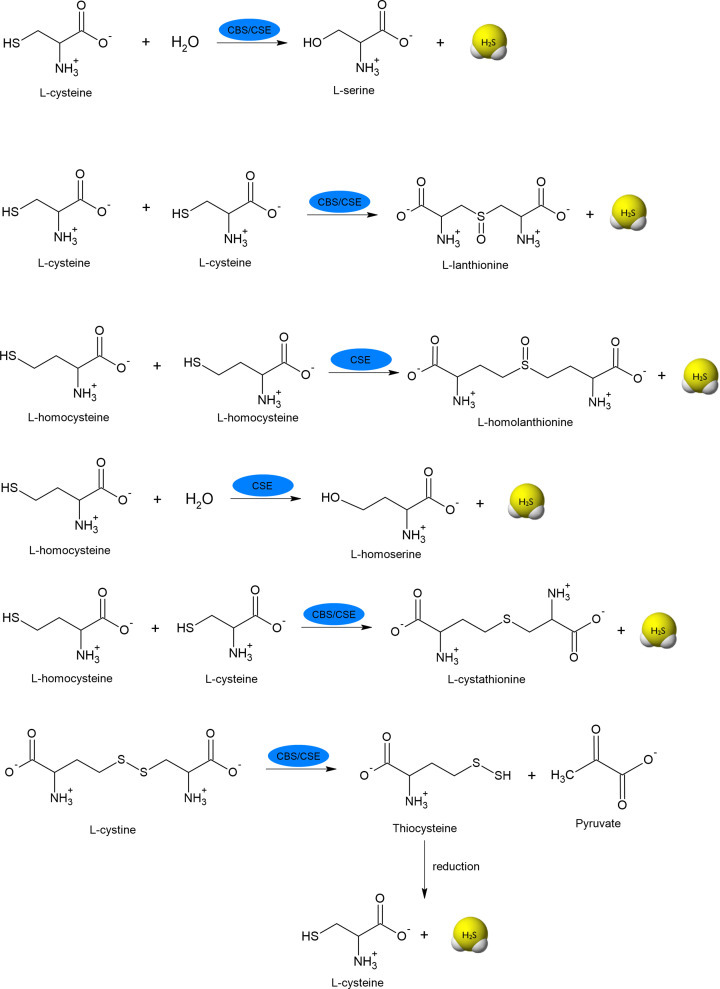
Cys-SH can be transformed to 3-mercaptopyruvate (3-MP) in reaction catalyzed by PLP-dependent cysteine aminotransferase (CAT). The product of this reaction, 3-MP, is a substrate for MST, which takes on a sulfur atom forming intermediate MST-SSH and then H2S is released under the influence of reducing agents, e.g. thioredoxin (Trxred) or dihydrolipoic acid (DHLA) (Figure 2) [9,10].
In peroxisomes, there is another possibility of H2S synthesis, namely from D-cysteine. In the first step, FMN-dependent D-amino acid oxidase (DAO), occurring in these organelles, converts D-cysteine to 3-MP, which then is a substrate for the MST [11]. The reactions catalyzed by CAT, MST and DAO are presented in Figure 4. Moreover, information about enzymes involved in H2S synthesis and their substrates are summarized in Table 1.
Cys-SH는 PLP 의존성 시스테인 아미노트랜스퍼라제(CAT)에 의해 촉매되는 반응에서 3-머캅토피루브산(3-MP)으로 변형될 수 있습니다. 이 반응의 결과물인 3-MP는 MST의 기질로, 황 원자를 흡수하여 중간체 MST-SSH를 형성한 다음 티오레독신(Trxred)이나 디하이드로리포익산(DHLA)과 같은 환원제의 영향으로 H2S가 방출됩니다(그림 2) [9,10].
퍼옥시좀에서는 D-시스테인으로부터 H2S가 합성될 수 있는 또 다른 가능성이 있습니다. 첫 번째 단계에서, 이 소기관에서 발생하는 FMN 의존성 D-아미노산 산화효소(DAO)는 D-시스테인을 3-MP로 전환하는데, 이것은 MST의 기질입니다 [11]. CAT, MST, DAO에 의해 촉매되는 반응은 그림 4에 나와 있습니다. 또한, H2S 합성에 관여하는 효소와 그 기질에 대한 정보는 표 1에 요약되어 있습니다.
Figure 4. Reactions catalyzed by CAT, MST and DAO responsible for H2S generation.
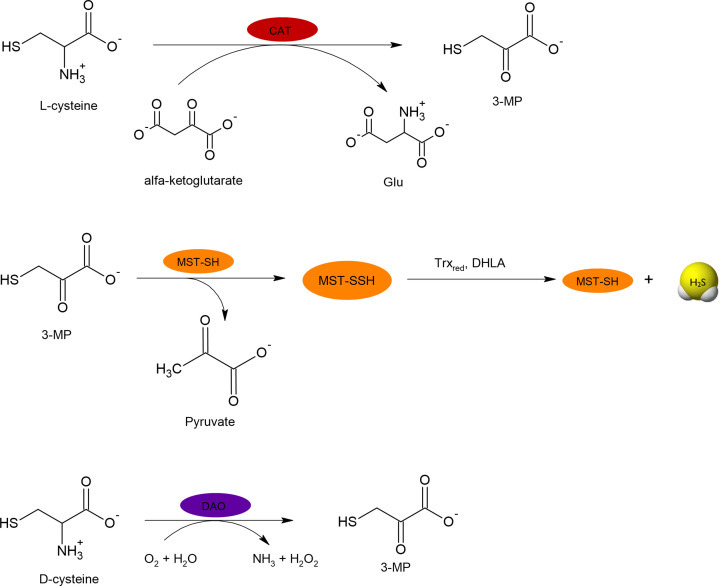
Table 1. Enzymes involved in synthesis of various RSS.
Enzyme involved in RSS synthesisKind of RSS formed by the enzymeSubstrate for RSS synthesis
| CBS* | H2S, persulfides | L-cysteine, L-homocysteine, L-cystine |
| CSE* | H2S, persulfides | L-cysteine, L-homocysteine, L-cystine |
| MST | polysulfides | 3-MP |
| CAT | H2S | L-cysteine |
| DAO | H2S | D-cysteine |
| SQR | persulfides | GSH, CoA-SH |
| GR | persulfides | GSSSG |
| CARS | persulfides, polysulfides | L-cysteine |
*
Pirydoxal phosphate dependent enzyme.
The main enzymes involved in endogenous H2S synthesis (CBS, CSE and MST) are expressed in many tissues. The highest expression of all three enzymes has been documented in the liver. CBS is mainly expressed in the brain, liver, kidney and pancreas. CSE has been found predominantly in the liver, kidney, thoracic aorta, ileum, portal vein, uterus, pancreatic islets and in the placenta. A modest expression and activity of CSE has been shown in the brain. MST, apart from the liver, is expressed also in the brain, colon, kidney, heart, lung and pancreas [12]. It can be concluded that CBS together with MST play a dominant role in the nervous system, while CSE with MST in the cardiovascular system [13–15].
CBS in its active form consists of four 63-kDa subunits, each of which contains three domains: PLP-binding domain, heme binding domain (N-terminal) and regulatory domain (C-terminal) [16]. The activity of CBS is influenced by many factors, both endogenous and exogenous [17]. The allosteric activator of CBS S-adenosylmethionine (SAM) can increase its activity [16,18]. Moreover, SAM was shown to modulate binding of other gasotransmiters NO and CO to the heme, and in this way it can also regulate enzymatic activity of CBS [19]. The activity of CBS is also modulated by S-glutathionylation [20], phosphorylation [21] and sumoylation [22]. Two sites of ubiquitination have been found in the CBS structure [23], and it can be suspected that also this common modification of proteins plays a role in the regulation of CBS activity.
CSE, just like CBS, is a tetramer but the molecular weight of its monomer is 45 kDa [12]. The regulation of CSE is not very well understood, yet. An in vitro study suggests sumoylation as a post-translational modification of CSE [24]. Expression of CSE is regulated by phosphorylation [25] and promotor methylation [26]. The increased expression of CSE was observed in some pathological conditions, i.e. in inflammation [27], oxidative stress [28,29] and in malnutrition [30].
In the case of MST regulation, the influence of reducing compounds such as Trxred or DHLA is an important aspect [10]. Moreover, the MST/CAT pathway is inhibited by Ca2+ ions that limit H2S synthesis through CAT inhibition [31].
Another important source of H2S derives from microbiome in the human and animal gut. Sulfate-reducing bacteria (SRB) living in the oceans in the Archean have also been found in the mammalian gut, where they have a suitable anaerobic environment. SRB are ubiquitous members of the mammalian colon and include several types of bacteria with dominant genera Desulfovibrio. SRB can reduce sulfate to H2S non-enzymatically. Some species of SRB (namely Desulfovibrio piger) are also able to use sulfated glycans as a substrate [32]. It has been documented that a sulfate-rich diet results in increased H2S production in the colon of mice [33]. Besides SRB, several anaerobic bacterial strains living in the digestive tracts, including Escherichia coli, Salmonella enterica, Clostridia and Enterobacter aerogenes can metabolize cysteine to H2S, pyruvate and ammonia in reaction catalyzed by cysteine desulfhydrase [34,35]. Another possibility of H2S production by most of the above-mentioned and some other bacteria (Klebsiella, Bacillus, Staphylococcus, Corynebacterium and Rhodococcus) consist in the reduction of sulfite by sulfite reductase [36,37].
Some authors suggest that H2S production by the gut microbiota exceeds its endogenous formation from cysteine [38]; however, other researchers indicate an equal share of both sources [39]. It has been reported that the total sulfide concentration in the luminal content of the large intestine is in the range of millimolar concentration in mammals; however, less than 8% of the total sulfide exists in the free form due to a large binding capacity of feces [40,41]. Interestingly, studies performed with germ-free and conventional mice showed that the presence of the microbiota resulted in a higher H2S concentration in the plasma (0.03 nmol/mg protein vs. 0.009 nmol/mg protein) as well as in the intestinal tract [41]. Moreover, in the plasma of germ-free mice, a significant decrease (0.005 nmol/mg vs. 0.03 nmol/mg protein) in the level of bound sulfane sulfur, considered to be a way of H2S storage, was observed [42]. On the other hand, in most organs of germ-free animals, an increased cysteine level and a decreased activity of CSE were found [43]. These observations strongly support an important role of the microbiota in H2S production in the mammalian body as well as suggest an impact of the gut microflora on bioavailability of cysteine and activity of CSE. Flannigan et al. examined colonic H2S synthesis derived from bacteria and colonic tissue in healthy and colitic mice [39]. They found that approximately one half of the H2S in feces was derived from colonic cells. When the tissue is injured or during an inflammation, the production of H2S by colonic cells is markedly increased. Moreover, it was shown that the lack of PLP in the diet resulted in reduced fecal H2S level by approximately 50% [39]. It indicates that enzymatic formation of H2S by PLP-dependent enzymes (CBS and CSE) in colonic tissues is responsible for about a half of the total production of fecal H2S.
피리독살 인산 의존성 효소.
내인성 H2S 합성에 관여하는 주요 효소(CBS, CSE, MST)는 많은 조직에서 발현됩니다. 세 가지 효소 모두 간에서 가장 많이 발현되는 것으로 알려져 있습니다. CBS는 주로 뇌, 간, 신장, 췌장에서 발현됩니다. CSE는 주로 간, 신장, 흉부 대동맥, 회장, 문맥, 자궁, 췌장 섬, 태반에서 발견됩니다. CSE의 활동과 표현이 뇌에서 이루어지고 있는 것으로 나타났습니다. MST는 간을 제외하고 뇌, 결장, 신장, 심장, 폐, 췌장에서도 발현됩니다 [12]. 따라서 CBS와 MST가 신경계에서 지배적인 역할을 하는 반면, CSE와 MST는 심혈관계에서 지배적인 역할을 한다고 결론을 내릴 수 있습니다 [13-15].
활성 상태의 CBS는 4개의 63kDa 소단위로 구성되어 있으며, 각 소단위는 PLP 결합 영역, 헴 결합 영역(N-말단) 및 조절 영역(C-말단)의 세 가지 영역을 포함합니다 [16]. CBS의 활동은 내인성 및 외인성 요인에 의해 영향을 받습니다 [17]. CBS의 알로스테릭 활성화제인 S-아데노실메티오닌(SAM)은 CBS의 활동을 증가시킬 수 있습니다 [16,18]. 또한, SAM은 다른 가스 전달 물질인 NO와 CO가 헴에 결합하는 것을 조절하는 것으로 나타났으며, 이러한 방식으로 CBS의 효소 활성을 조절할 수도 있습니다 [19]. CBS의 활성은 S-글루타티온화 [20], 인산화 [21], 스모일화 [22]에 의해서도 조절됩니다. CBS 구조에서 두 개의 유비퀴틴화 부위가 발견되었으며[23], 이 단백질의 일반적인 변형도 CBS 활성의 조절에 중요한 역할을 하는 것으로 추정됩니다.
CBS와 마찬가지로 CSE도 4량체이지만, 그 단량체의 분자량은 45kDa입니다[12]. CSE의 조절 메커니즘은 아직 잘 알려져 있지 않습니다. 체외 연구에 따르면 CSE의 번역 후 변형으로 스모일레이션이 제안되었습니다 [24]. CSE의 발현은 인산화 [25]와 프로모터 메틸화 [26]에 의해 조절됩니다. CSE의 발현 증가는 염증 [27], 산화 스트레스 [28,29] 및 영양실조 [30]와 같은 일부 병리학적 조건에서 관찰되었습니다.
MST 조절의 경우, Trxred 또는 DHLA와 같은 환원 화합물의 영향이 중요한 측면입니다 [10]. 또한, MST/CAT 경로는 Ca2+ 이온에 의해 억제되어 CAT 억제를 통해 H2S 합성을 제한합니다 [31].
H2S의 또 다른 중요한 공급원은 인간과 동물의 장에 있는 미생물 군집입니다. 고생대 바다에 서식하는 황산염 환원 박테리아(SRB)도 포유류의 장에서 발견되었으며, 그곳은 적절한 혐기성 환경입니다. SRB는 포유류 결장의 보편적인 구성원이며, 지배적인 속인 데설로비리오(Desulfovibrio)를 가진 여러 종류의 박테리아를 포함합니다. SRB는 비효소적으로 황산염을 H2S로 환원할 수 있습니다. 일부 SRB 종(Desulfovibrio piger)은 황산염화된 글리칸을 기질로 사용할 수 있습니다 [32]. 황산염이 풍부한 식단은 쥐의 결장에서 H2S 생산을 증가시킨다는 사실이 밝혀졌습니다 [33]. SRB 외에도 대장균, 살모넬라 장염, 클로스트리듐, 엔테로박터 에어로제네스를 포함한 소화관에 서식하는 여러 혐기성 세균이 시스테인 탈황효소에 의해 촉매되는 반응에서 시스테인을 H2S, 피루브산, 암모니아로 대사할 수 있습니다 [34,35]. 위에서 언급한 대부분의 박테리아와 일부 다른 박테리아(클레브시엘라, 바실러스, 포도상구균, 코리네박테리움, 로도코쿠스)에 의한 H2S 생성의 또 다른 가능성은 아황산염 환원효소에 의한 아황산염의 환원에 있습니다 [36,37].
일부 저자들은 장내 미생물총에 의한 H2S 생성이 시스테인으로부터의 내인성 생성을 초과한다고 주장합니다 [38]; 그러나 다른 연구자들은 두 가지 원인의 비율이 동일하다고 지적합니다 [39]. 대장의 내강 내용물에서 황화물의 총 농도는 포유류의 경우 밀리몰 농도 범위 내에 있는 것으로 보고되었지만, 대변의 결합력이 크기 때문에 총 황화물의 8% 미만이 자유 형태로 존재합니다[40,41]. 흥미롭게도 무균 생쥐와 일반 생쥐를 대상으로 한 연구에 따르면 미생물총의 존재는 혈장(단백질 1mg당 0.03nmol 대 0.009nmol)과 장에서 H2S 농도를 증가시키는 것으로 나타났습니다[41]. 또한 무균 생쥐의 혈장에서는 H2S 저장 방법으로 간주되는 결합된 황화황의 수준이 현저하게 감소(0.005 nmol/mg 대 0.03 nmol/mg 단백질)하는 것이 관찰되었습니다 [42]. 반면에 무균 동물의 대부분의 기관에서는 시스테인 수준이 증가하고 CSE의 활동이 감소하는 것이 발견되었습니다 [43]. 이러한 관찰 결과는 포유류 체내 H2S 생산에 있어 미생물총의 중요한 역할을 강력하게 뒷받침하는 동시에, 장내 미생물총이 시스테인의 생체 이용률과 CSE의 활성에 영향을 미친다는 것을 시사합니다. Flannigan 등은 건강한 쥐와 대장염 쥐의 대장 조직과 박테리아에서 유래된 대장 H2S 합성을 조사했습니다 [39]. 그들은 대변에 있는 H2S의 약 절반이 대장 세포에서 유래된다는 것을 발견했습니다. 조직이 손상되거나 염증이 있을 때, 결장 세포에 의한 H2S의 생성이 현저하게 증가합니다. 또한, 식이요법에서 PLP가 부족하면 대변 H2S 수치가 약 50% 감소하는 것으로 나타났습니다 [39]. 이는 결장 조직에서 PLP 의존성 효소(CBS 및 CSE)에 의한 H2S의 효소적 형성이 대변 H2S 총 생산량의 약 절반을 담당한다는 것을 나타냅니다.
Catabolism of H2S
H2S plays a signaling role only at physiological, relatively low concentrations. It is well known that in higher concentrations H2S is toxic as an inhibitor of complex IV of the respiratory chain, namely, cytochrome oxidase, and in this way, it impairs cell respiration [44,45]. In order to prevent this toxicity, bioavailability of H2S must be tightly regulated by its safe and nontoxic storage in the form of bound sulfane sulfur (see the next chapter) and by efficient catabolism. In mammals, H2S is oxidized mainly in mitochondria via the ‘sulfide oxidizing pathway’ with thiosulfate and sulfate being the end products of this pathway. Three enzymes are involved in the catabolism of H2S: sulfide quinone oxidoreductase (SQR), persulfide dioxygenase (ETHE1) and rhodanese also known as thiosulfate sulfurtransferase (TST) [46].
The H2S oxidizing pathway begins with the reaction catalyzed by SQR resulting in the formation of SQR persulfide (SQR-SSH) at one of its two Cys-SH residues [47–49]. SQR is a flavoprotein bound to the inner mitochondrial membrane, and while it is oxidized a noncovalently bound FAD is reduced and electrons are transferred to coenzyme Q contributing to the electron transport chain and ATP production. For this reason, H2S was regarded as the first inorganic substrate for oxidative phosphorylation comparable to succinate [44,50]. SQR is characterized by a low KM value and high catalytic rate, which contributes to effective catabolism of H2S [48]. The sulfur atom from SQR-SSH can be transferred to glutathione (GSH) forming glutathione persulfide (GSSH) and next it can be oxidized to sulfite (SO32−) by ETHE1 or transferred to sulfite by TST resulting in thiosulfate formation (Figure 5). Sulfite is further oxidized to sulfate by sulfite oxidase (SO). Sulfite can also be a direct acceptor of sulfur atom from SQR-SSH producing thiosulfate. Some authors suggested that endogenous reducing compounds other than GSH (i.e. DHLA) could accept the sulfur atom from SQR-SSH [51]. GSSH can also donate its sulfur atom to proteins forming protein persulfides [52].
H2S는 생리학적으로 상대적으로 낮은 농도에서만 신호 전달 역할을 합니다. 높은 농도의 H2S는 호흡 사슬의 복합 IV, 즉 시토크롬 산화효소의 억제제로서 독성을 나타내며, 이러한 방식으로 세포 호흡을 손상시키는 것으로 잘 알려져 있습니다 [44,45]. 이러한 독성을 방지하기 위해서는 H2S의 생체 이용률을 결합된 황화황의 형태로 안전하고 무독성 상태로 저장하고(다음 장 참조) 효율적인 이화 작용을 통해 엄격하게 조절해야 합니다. 포유류에서 H2S는 주로 미토콘드리아에서 티오설페이트와 황산염을 최종 산물로 생성하는 '황화물 산화 경로'를 통해 산화됩니다. H2S의 이화 작용에는 황화물 퀴논 산화 환원 효소(SQR), 퍼설파이드 디옥시게나제(ETHE1), 티오설페이트 황 전이 효소(TST)로도 알려진 로다네이즈 등 세 가지 효소가 관여합니다 [46].
H2S 산화 경로는 SQR에 의해 촉매 작용을 받는 반응으로 시작되어, 두 개의 Cys-SH 잔기 중 하나에서 SQR 과황화물(SQR-SSH)이 형성됩니다 [47-49]. SQR은 미토콘드리아 내막에 결합된 플라보단백질이며, 산화되는 동안 비공유 결합된 FAD가 환원되고 전자가 코엔자임 Q로 전달되어 전자 수송 사슬과 ATP 생성에 기여합니다. 이러한 이유로 H2S는 숙시네이트와 비교할 수 있는 산화적 인산화반응을 위한 최초의 무기 기질로 간주되었습니다 [44,50]. SQR은 낮은 KM 값과 높은 촉매 속도를 특징으로 하며, 이는 H2S의 효과적인 이화 작용에 기여합니다 [48]. SQR-SSH의 황 원자는 글루타티온(GSH)으로 옮겨져 글루타티온 퍼설파이드(GSSH)를 형성하고, 그 다음에는 ETHE1에 의해 아황산염(SO32−)으로 산화되거나 TST에 의해 아황산염으로 옮겨져 티오황산염이 형성될 수 있습니다(그림 5). 아황산염은 아황산염 산화효소(SO)에 의해 황산염으로 더 산화됩니다. 설파이트는 또한 티오황산염을 생성하는 SQR-SSH로부터 황 원자를 직접 받아들이는 역할을 할 수 있습니다. 일부 저자들은 GSH(즉, DHLA) 이외의 내인성 환원 화합물이 SQR-SSH로부터 황 원자를 받아들이는 역할을 할 수 있다고 제안했습니다 [51]. GSSH는 또한 단백질 과황화물을 형성하는 단백질에 황 원자를 기증할 수 있습니다 [52].
Figure 5. Mitochondrial oxidation of H2S.
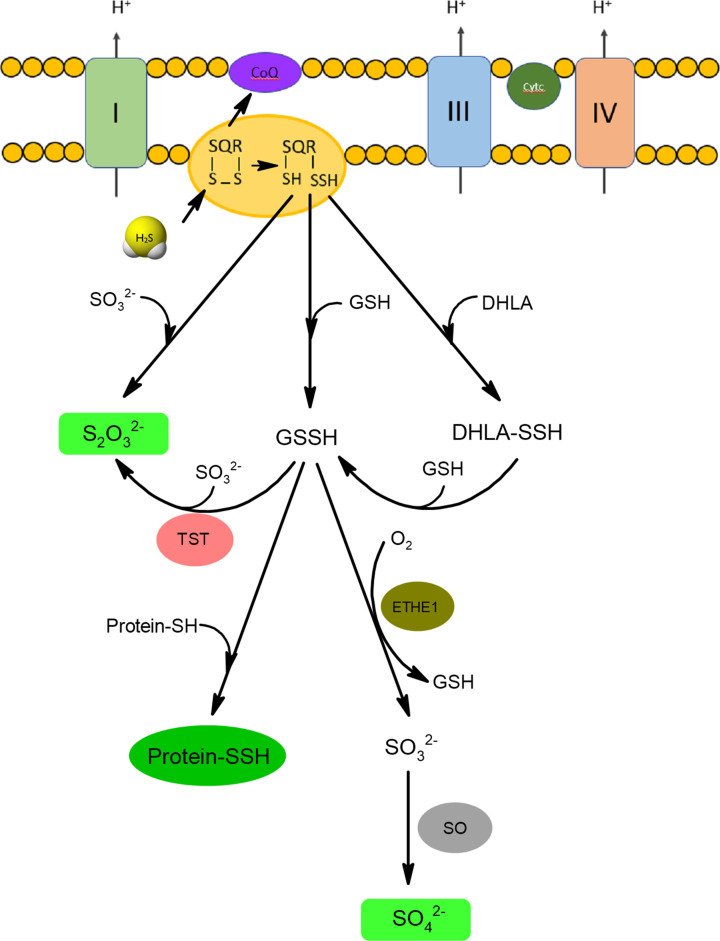
H2S in the reaction with oxidized form of SQR forms persulfide that can be a donor of sulfur atom for glutathione (GSH), DHLA or sulfite anions forming appropriate persulfide forms: GSSH and DHLA-SSH or thiosulfate, respectively. Sulfur atom of GSSH can be next oxidized by persulfide dioxygenase (ETHE1) to sulfite (SO32−) that is further oxidized to sulfate by sulfite oxidase (SO). Sulfur atom from GSSH can be also transferred to sulfite in the reaction catalyzed by rhodanese (TST) resulting in thiosulfate formation. Thiosulfate and sulfate anions are the main end products of H2S catabolism.
산화된 형태의 SQR과 반응하는 H2S는 글루타티온(GSH), DHLA 또는 아황산염 음이온을 형성하는 적절한 퍼설파이드 형태인 GSSH 및 DHLA-SSH 또는 티오황산염을 형성하는 황 원자를 기증할 수 있는 퍼설파이드를 형성합니다. GSSH의 황 원자는 과산화이산화황산화효소(ETHE1)에 의해 아황산염(SO32−)으로 산화될 수 있으며, 아황산염산화효소(SO)에 의해 황산염으로 산화됩니다. GSSH의 황 원자는 또한 로다네이즈(TST)에 의해 촉매되는 반응에서 아황산염으로 이동되어 티오황산염을 형성할 수 있습니다. 티오설페이트와 황산염 음이온은 H2S 분해의 주요 최종 산물입니다.
It is worth mentioning that H2S can also be oxidized by hemoproteins. In this case, the end products of H2S oxidation, i.e. thiosulfate and inorganic polysulfides, are bound to heme iron [53]. Initially, H2S binds to Fe3+ in heme (MetHb) and then another molecules of H2S combine forming persulfide or polysulfide chain. The persulfide can be oxidized in the presence of oxygen to thiosulfate, which can be released as the end product (Figure 6). Involvement of Hb in the H2S catabolism seems to be necessary in erythrocytes which do not have mitochondria. It has been documented that other proteins utilizing oxygen or H2O2 as an electron acceptor (i.e. catalase, superoxide dismutase, SOD) can also oxidize H2S to polysulfide [54,55].
H2S는 또한 헤모단백질에 의해 산화될 수 있다는 점을 언급할 필요가 있습니다. 이 경우, H2S 산화의 최종 산물인 티오설페이트와 무기 다황화물은 헴철에 결합합니다 [53]. 처음에, H2S는 헴(MetHb)의 Fe3+에 결합한 다음, 또 다른 H2S 분자가 결합하여 퍼설파이드 또는 폴리설파이드 사슬을 형성합니다. 퍼설파이드는 산소가 있는 상태에서 티오설페이트로 산화될 수 있으며, 최종 생성물로 방출될 수 있습니다(그림 6). 미토콘드리아가 없는 적혈구에서는 H2S의 대사에 Hb가 관여하는 것이 필요한 것으로 보입니다. 산소 또는 H2O2를 전자 수용체로 이용하는 다른 단백질(즉, 카탈라제, 슈퍼옥사이드 디스뮤타제, SOD)도 H2S를 폴리설파이드로 산화시킬 수 있다는 사실이 밝혀졌습니다 [54,55].
그림 6. 티오황산염과 무기 폴리설파이드를 최종 산물로 하는 헤모단백질 의존적 H2S 산화.
Figure 6. Hemoprotein-dependent oxidation of H2S with thiosulfate and inorganic polysulfide as end products.
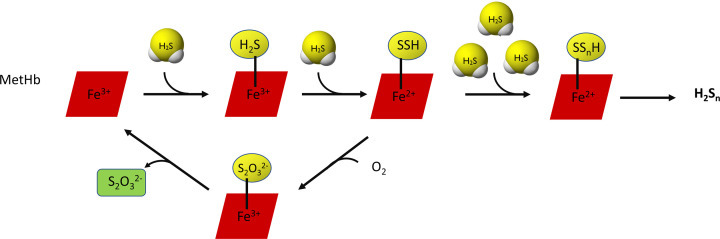
Many molecules of H2S can bind to Fe3+ ion in heme (MetHb) forming persulfide or polysulfide chain. The persulfide can be oxidized in the presence of oxygen to thiosulfate. This pathway of H2S oxidation is important in erythrocytes that do not have mitochondria.
많은 H2S 분자가 헴(MetHb)의 Fe3+ 이온에 결합하여 퍼설파이드 또는 폴리설파이드 사슬을 형성할 수 있습니다. 퍼설파이드는 산소가 있는 상태에서 티오설페이트로 산화될 수 있습니다. H2S 산화의 이 경로는 미토콘드리아가 없는 적혈구에서 중요합니다.
Persulfides (RSSH)
Persulfides (RSSH) in contrast to H2S contain sulfane sulfur atom bound to cysteine thiol. Sulfane sulfur is defined as a sulfur atom occurring in the 0 or -1 oxidation state covalently bound to another sulfur atom. In the RSSH structure, the sulfane sulfur atom is the outer sulfur with -1 oxidation state, while in elemental sulfur (S8) or in polysulfides (RSSSR) the oxidation state of sulfane sulfur atom is 0.
RSSH have aroused interest of researchers because they were proposed to mediate the H2S signaling. The search for the keyword ‘persulfide’ in the PubMed database yielded about 679 records, and since 1997 there has been a steady increase in interest in these compounds (Figure 1). A large body of evidence indicate that persulfides possess a similar physiological role as H2S. For this reason, RSSH have been shown to be responsible for some of the effects originally attributed to H2S. Indeed, H2S at higher concentrations is a toxic compound, while RSSH do not show such properties.
과황화수소(RSSH)
과황화수소(RSSH)는 H2S와는 달리 시스테인 티올에 결합된 설파인 황 원자를 포함합니다. 설파인 황은 다른 황 원자에 공유 결합된 산화 상태가 0 또는 -1인 황 원자로 정의됩니다. RSSH 구조에서 설파인 황 원자는 산화 상태가 -1인 외부 황이고, 원소 황(S8) 또는 폴리설파이드(RSSSR)에서 설파인 황 원자의 산화 상태는 0입니다.
RSSH는 H2S 신호를 매개하는 것으로 제안되어 연구자들의 관심을 불러일으켰습니다. PubMed 데이터베이스에서 'persulfide'라는 키워드를 검색하면 약 679개의 기록이 나옵니다. 1997년 이후로 이러한 화합물에 대한 관심이 꾸준히 증가하고 있습니다(그림 1). 많은 증거가 persulfides가 H2S와 유사한 생리학적 역할을 가지고 있음을 나타냅니다. 이런 이유로, RSSH가 원래 H2S에 기인하는 일부 효과에 대한 책임이 있는 것으로 밝혀졌습니다. 실제로, 고농도의 H2S는 독성 화합물인 반면, RSSH는 그러한 특성을 보이지 않습니다.
General properties of RSSH
The chemical properties of RSSH are often compared with the RSH which are their thiol analogues. Replacing the -SH group in thiols with the -SSH group in persulfides affects energy of S-H bond, that is weaker by 92 kJ/mol in RSSH compared with RSH [56]. This energy difference will result in an increased stability and weaker toxicity of persulfide radicals (RSS●) in relation to thiyl radicals (RS●). The internal sulfur atom in RSS● structure is stabilized by resonance effect [57] (reaction 2).
| [R-¨S¨-˙S¨:⇄R-˙S¨-¨S¨:] | (2) |
Persulfides are more potent H atom donors than thiols, and similarly anionic perthiols (RSS−) proved to be better one-electron reductants compared with corresponding anionic thiol (RS−) [58,59]. It was confirmed by Koppenol and Bounds who based on thermodynamic data estimated that at pH 7.0 the electrode potential of RSS●/RSSH was +0.68V, while the potential for RS●/RSH was +0.96V [60]. For this reason, persulfides are considered to be stronger and more effective antioxidants than thiols. It was demonstrated that GSSH effectively reduced H2O2, while GSH or H2S in the same conditions was not sufficient in this reduction [61]. Recently, Li et al. showed by using resonance synchronous spectroscopy that GSSH was 50-fold more reactive than H2S relative to H2O2 at physiological pH (k values estimated for GSSH and H2S were 23.76 and 0.46 M−1s−1, respectively) [62]. The noticeable increase in the reducing capacity of persulfides relative to thiols can be attributed to the possibility of their tautomerization to thiosulfoxide form (reaction 3) [63].
| R-S-SH⇄:¨S¨:R-SH | (3) |
Bianco et al. examined the ability of RSSH to act as one-electron reductants using an alkyl hydropersulfide model, namely, N-methoxycarbonyl penicillamine persulfide (MCP-SSH). They found that this persulfide was easily oxidized by the moderately strong one-electron oxidant 4-hydroxy-2,2,6,6-tetramethylpiperidine 1-oxyl (TEMPOL) to TEMPOL-OH, while the corresponding thiol was not. MCP-SSH was also efficiently oxidized by K3Fe(CN)6 and Fe(III) of myoglobin, while in the case of the corresponding thiol, reduction of myoglobin Fe(III) did not occur and reduction of K3Fe(CN)6 was less than 50% of that with MCP-SSH [64]. Moreover, a study performed on A549 cells revealed that the cells with overexpression of CSE responsible for persulfide formation showed a better viability after treatment with H2O2 than those with normal CSE expression [61]. All these facts clearly confirm that persulfides are much more powerful reductants compared with corresponding thiols. However, it should be noted that all the above-mentioned facts were derived based on in vitro experiments using comparable concentrations of thiols and persulfides. However, the concentrations of persulfides present in cells are much lower than thiol concentrations what means that persulfides are even more reactive than thiols, in physiological conditions antioxidant reactions involving thiols, especially GSH, predominate due to considerably higher concentration of GSH than GSSH.
Persulfides are characterized by a higher acidity when compared with thiols (reactions 4 and 5) [65].
| RSSH⇄RSS−+H+ | (4) |
| RSH⇄RS−+H+ | (5) |
The pKa value for 2-[3-aminopropyl)amino]ethane persulfide was estimated at 6.2, whereas the pKa for the corresponding thiol was 7.6 [58]. On the other hand, based on computational calculation the pKa of cysteine persulfide (CysSSH) was estimated at 4.3, while the pKa of CysSH is known to be 8.3 [66]. The pKa value of glutathione persulfide (GSSH) is a little controversial because Li et al. using resonance synchronous spectroscopy estimated it at 6.9 [62], while Benchoam et al. in the reaction with mBrB determined it as 5.45 [67]. The pKa of GSH is assumed to be 8.9, so independently of the difference in the estimated values of pKa for GSSH, persulfides possess much stronger acidic character. Based on these data, it can be concluded that at physiological pH anionic perthiol species (RSS−) dominate over RSSH forms, whereas thiols predominantly exist in the protonated form (RSH).
RSSH의 일반적인 특성
RSSH의 화학적 특성은 종종 티올 유사체인 RSH와 비교됩니다. 티올의 -SH 그룹을 퍼설파이드의 -SSH 그룹으로 대체하면 RSSH의 S-H 결합 에너지가 RSH에 비해 92kJ/mol 더 약해집니다 [56]. 이 에너지 차이로 인해 티일 라디칼(RS●)과 관련하여 과황화 라디칼(RSS●)의 안정성이 증가하고 독성이 약해집니다. RSS● 구조의 내부 황 원자는 공명 효과에 의해 안정화됩니다 [57] (반응 2).
[R-¨S¨-˙S¨:⇄R-˙S¨-¨S¨:] (2)
퍼설파이드가 티올보다 더 강력한 H 원자 공여체이고, 유사하게 음이온 페르티올(RSS−)이 해당 음이온 티올(RS−)에 비해 더 나은 일전자 환원제로 입증되었습니다 [58,59]. Koppenol과 Bounds는 열역학 데이터를 바탕으로 pH 7.0에서 RSS●/RSSH의 전극 전위가 +0.68V이고, RS●/RSH의 전극 전위가 +0.96V[60]라고 추정했습니다. 이러한 이유로, 퍼설파이드가 티올보다 더 강력하고 효과적인 항산화제로 간주됩니다. GSSH가 H2O2를 효과적으로 감소시키는 것으로 밝혀졌지만, 같은 조건에서 GSH 또는 H2S는 이 감소에 충분하지 않았습니다 [61]. 최근에 Li 등은 공명 동시 분광법을 사용하여 생리학적 pH에서 GSSH가 H2O2에 비해 H2S보다 50배 더 반응성이 높다는 것을 보여주었습니다(GSSH와 H2S에 대해 추정된 k 값은 각각 23.76과 0.46 M−1s−1이었습니다) [62]. 설파이드의 환원 능력이 티올에 비해 현저하게 증가한 것은 설파이드가 티오설폭사이드 형태로 전이 가능한 가능성 때문일 수 있습니다(반응 3) [63].
R-S-SH⇄:¨S¨:R-SH (3)
Bianco 등은 알킬 하이드로퍼설파이드 모델, 즉 N-메톡시카르보닐 페니실라민 퍼설파이드(MCP-SSH)를 사용하여 RSSH가 1전자 환원제로서 작용하는 능력을 조사했습니다. 그들은 이 퍼설파이드가 중간 정도의 강한 일전자 산화제인 4-하이드록시-2,2,6,6-테트라메틸피페리딘 1-옥실(TEMPOL)에 의해 쉽게 산화되어 TEMPOL-OH로 변하는 반면, 해당 티올은 그렇지 않다는 것을 발견했습니다. MCP-SSH는 또한 K3Fe(CN)6과 미오글로빈의 Fe(III)에 의해 효율적으로 산화되었지만, 해당 티올의 경우, 미오글로빈의 Fe(III) 환원은 일어나지 않았고, K3Fe(CN)6의 환원은 MCP-SSH의 환원율의 50% 미만이었습니다 [64]. 또한, A549 세포에 대한 연구에 따르면, 과산화황 형성에 책임이 있는 CSE의 과발현이 있는 세포는 정상적인 CSE 발현을 가진 세포보다 H2O2 처리 후 생존력이 더 우수하다는 사실이 밝혀졌습니다 [61]. 이러한 모든 사실은 과산화황이 해당 티올에 비해 훨씬 더 강력한 환원제임을 분명히 확인시켜 줍니다. 그러나 위에서 언급한 모든 사실은 비슷한 농도의 티올과 퍼설파이드를 사용한 체외 실험을 기반으로 한 것임을 유의해야 합니다. 그러나 세포에 존재하는 퍼설파이드의 농도는 티올 농도보다 훨씬 낮습니다. 즉, 퍼설파이드가 티올보다 훨씬 더 반응성이 높다는 것을 의미합니다. 생리학적 조건에서 티올, 특히 GSH가 포함된 항산화 반응은 GSSH보다 GSH의 농도가 훨씬 높기 때문에 우세합니다.
과황화물은 티올(반응 4 및 5)에 비해 산도가 더 높은 것이 특징입니다 [65].
RSSH⇄RSS−+H+ (4)
RSH⇄RS−+H+ (5)
2-[3-aminopropyl)amino]ethane persulfide의 pKa 값은 6.2로 추정되었으며, 이에 상응하는 thiol의 pKa는 7.6 [58]입니다. 한편, 계산에 기초한 시스테인 퍼설파이드(CysSSH)의 pKa는 4.3으로 추정되었으며, CysSH의 pKa는 8.3 [66]으로 알려져 있습니다. 글루타티온 퍼설파이드(GSSH)의 pKa 값은 Li et al.이 공명 동시 분광법을 사용하여 6.9 [62]로 추정하고, Benchoam et al.이 mBrB와의 반응에서 5.45 [67]로 결정했기 때문에 약간 논란의 여지가 있습니다. GSH의 pKa는 8.9로 추정되므로, GSSH의 pKa 추정값의 차이와 관계없이, 과황화물은 훨씬 더 강한 산성 특성을 가지고 있습니다. 이러한 데이터를 바탕으로, 생리학적 pH에서 음이온성 페르티올 종(RSS−)이 RSSH 형태보다 우세하고, 티올은 주로 양성자화된 형태(RSH)로 존재한다고 결론을 내릴 수 있습니다.
Nucleophilicity of RSSH
Persulfides, like thiols can act as nucleophiles (reactions 6 and 7).
| RSS−+E+⇄RSSE | (6) |
| RS−+E+⇄RSE | (7) |
They react with electrophilic alkylating agents, such as iodoacetamide (IAA), monobromobimane (mBrB), N-ethylmaleimide (NEM) and dinitrofluorobenzene (DNFB) yielding appropriate disulfides (Figure 7). Thiols in the same reactions form respective thioethers. Reactions with electrophiles are also used for persulfide determination; however, other new methods have been developed [68,69]. It has also been documented that persulfides can efficiently react with 8-nitroguanosine 3′,5′-cyclic monophosphate (8-nitro-cGMP), an endogenously occurring weak electrophile acting as a second messenger in redox signaling [61].
RSSH의 친핵성
설파이드 화합물은 티올과 마찬가지로 친핵체 역할을 할 수 있습니다(반응 6 및 7).
RSS−+E+⇄RSSE (6)
RS−+E+⇄RSE (7)
이들은 요오드아세트아미드(IAA), 모노브로모비만(mBrB), N-에틸말레이미드(NEM), 디니트로플루오로벤젠(DNFB)과 같은 친전자성 알킬화제와 반응하여 적절한 디설파이드를 생성합니다(그림 7). 동일한 반응에서 티올은 각각의 티오에테르를 형성합니다. 친전자체와의 반응은 또한 퍼설파이드 측정에 사용되지만, 다른 새로운 방법들이 개발되었습니다 [68,69]. 또한 퍼설파이드가 산화 환원 신호 전달에서 제2 메신저 역할을 하는 내인성 약한 친전자체인 8-니트로구아노신 3′,5′-사이클릭 모노포스페이트(8-니트로-cGMP)와 효율적으로 반응할 수 있다는 사실이 밝혀졌습니다 [61].
Figure 7. Reactions of RSSH with popular electrophilic alkylating agents.
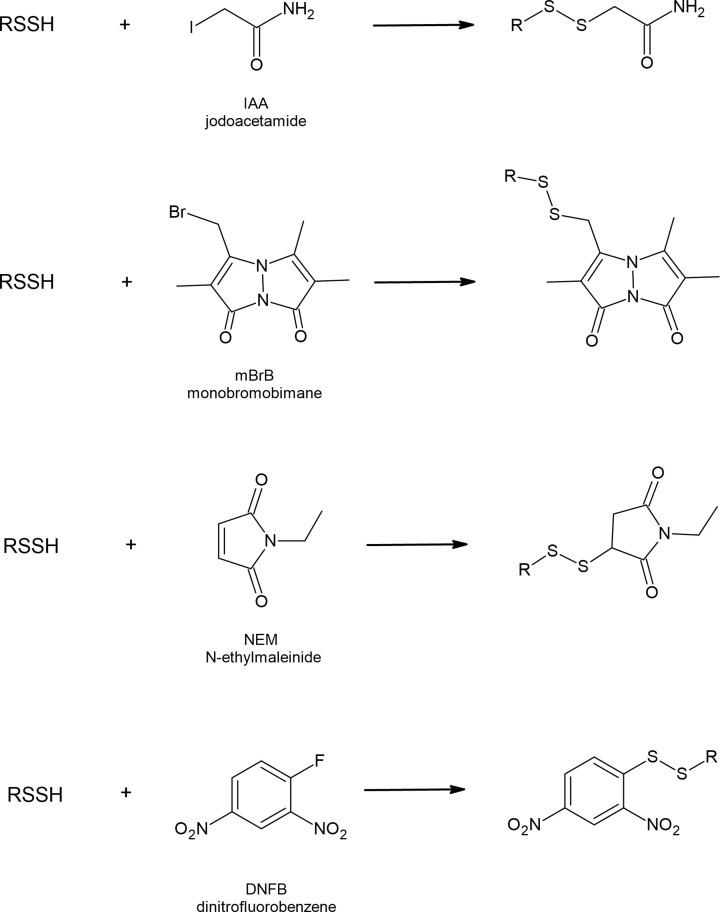
Persulfides are considered to be stronger nucleophiles than corresponding thiols. Benchoam et al. studied the kinetics of GSSH reactions with three various electrophiles and compared the data with parameters for GSH. The results indicate that at physiological pH the reactivity of GSSH toward mBrB, peroxynitrite and H2O2 is enhanced, when compared with GSH. According to the literature the estimated rate constants for GSSH and GSH are: 7.02 × 103 and 208 M−1s−1; 7.5 and 0.42 M−1s−1; 1.25 × 105 and 1360 M−1s−1 toward mBrB, H2O2 and peroxynitrite, respectively [67,70,71]. The increased nucleophilicity of persulfide compared with thiol has also been documented in the case of albumin persulfide in reaction with disulfide 4-4′-dithiodipyridine. Cuevasanta et al. reported the following second-order rate constants: 1.7∙104 M−1 s−1 for HSA-SSH and 7.6 × 102 M−1 s−1 for HSA-SH determined at pH 7.4 and at 25°C [66]. It can be concluded that persulfides react faster than thiols because of increased availability of the anion and so called alfa-effect (increased reactivity of an atom caused by the presence of unshared pairs of electrons in the adjacent atom). A similar effect has been observed for other pairs of compounds with nucleophilic character (NH2NH2/NH3 and HOO−/HO−) [72]. Strong nucleophilic character of RSSH makes them able to be used as a kind of defense against toxins with electrophilic nature. It has been documented that an electrophilic metabolite of acetaminophen N-acetyl-p-benzoquinone imine (NAPQI) reacts with CysSSH or GSSH forming mixed disulfides NAPQIH2-SSCys or NAPQIH2-SSG, respectively [73]. Sulfane sulfur-containing persulfides can also react with other electrophiles, such as methylmercury (MeHg) [74] or 1,4-naphthoquinone (1,4-NQ) [75] yielding various sulfur adducts that may be advantageous in toxicology [76].
Electrophilicity of RSSH
Regardless of the nucleophilic properties, persulfides when protonated possess also electrophilic character what distinguishes them from thiols. Nucleophiles can attack the outer sulfur atom of persulfides that leads to thiol formation; on the other hand, nucleophile attack on the inner sulfur atom produces H2S. The general reaction scheme and reactions of RSSH with popular nucleophiles that attack the outer sulfur atom are presented below in part A, whereas the reaction representing the attack of nucleophile on the inner sulfur atom is presented in part B.
퍼설파이드 화합물은 해당 티올보다 더 강력한 친핵체로 간주됩니다. 벤쵸암(Benchoam) 등은 세 가지 다양한 친전자체와 GSSH 반응의 동역학을 연구하고, 그 데이터를 GSH의 매개 변수와 비교했습니다. 그 결과, 생리학적 pH에서 GSSH가 mBrB, 퍼옥시니트라이트, H2O2에 대한 반응성이 GSH에 비해 향상된다는 사실이 밝혀졌습니다. 문헌에 따르면, GSSH와 GSH의 추정 속도 상수는 다음과 같습니다. 7.02 × 103 및 208 M−1s−1; 7.5 및 0.42 M−1s−1; 1.25 × 105 및 1360 M−1s−1 mBrB, H2O2 및 퍼옥시니트라이트를 향해 각각 [67,70,71]. 디설파이드 4-4′-디티오디피리딘과 반응하는 알부민 퍼설파이드의 경우, 티올에 비해 퍼설파이드의 친핵성이 증가하는 것이 문서화되었습니다. Cuevasanta 등은 pH 7.4, 25°C에서 측정된 HSA-SSH의 1.7∙104 M−1 s−1, HSA-SH의 7.6 × 102 M−1 s−1의 2차 속도 상수를 보고했습니다 [66]. 이는 음이온의 가용성 증가와 알파 효과(인접 원자 내 공유되지 않은 전자쌍의 존재로 인해 원자의 반응성이 증가하는 현상)로 인해 퍼설파이드가 티올보다 빠르게 반응한다는 결론을 내릴 수 있습니다. 친핵성 특성을 가진 다른 화합물 쌍(NH2NH2/NH3 및 HOO−/HO−)에서도 유사한 효과가 관찰되었습니다[72]. RSSH의 강한 친핵성은 친전자성 독소에 대한 일종의 방어 수단으로 사용될 수 있게 해줍니다. 아세트아미노펜의 친전자성 대사 산물인 N-아세틸-p-벤조퀴논이민(NAPQI)이 시스테인SSH 또는 글루타티온SSH와 반응하여 각각 혼합 이황화물 NAPQIH2-SSCys 또는 NAPQIH2-SSG를 형성하는 것으로 알려져 있습니다 [73]. 설파인 황 함유 퍼설파이드도 메틸수은(MeHg) [74] 또는 1,4-나프토퀴논(1,4-NQ) [75]과 같은 다른 친전자성 물질과 반응하여 독성학에 유용할 수 있는 다양한 황 부가물을 생성할 수 있습니다 [76].
RSSH의 친전자성
친핵성 특성에 관계없이, 프로설파이드가 양성자화되면 친전자성 특성도 가지게 되어, 이를 티올과 구별할 수 있습니다. 친핵체는 티올 형성을 유도하는 과황화물의 외부 황 원자를 공격할 수 있습니다; 반면에, 내부 황 원자에 대한 친핵체 공격은 H2S를 생성합니다. 외부 황 원자를 공격하는 친핵체와 RSSH의 일반적인 반응 방식과 반응은 파트 A에, 내부 황 원자에 대한 친핵체 공격의 반응을 나타내는 반응은 파트 B에 제시되어 있습니다.
A
| 𝐑𝐒𝐒𝐇+𝐍𝐮−⇄𝐑𝐒−+𝐍𝐮𝐒𝐇 |
| RS𝐒H+CN−⇄RS−+H𝐒CN | (8) |
| RS𝐒H+SO2−3⇄RS−+H𝐒SO2−3 | (9) |
| RS𝐒H+OH−⇄RS−+H𝐒OH | (10) |
| RS𝐒H+R′3P⇄RS𝐇+R′3P=𝐒 | (11) |
| RS𝐒H+R′S−⇄RS−+R′S𝐒𝐇 | (12) |
B
| 𝐑𝐒𝐒𝐇+𝐍𝐮−⇄𝐇𝐒−+𝐑𝐒𝐍𝐮 |
| R𝐒SH+R'S−⇄RSSR'+HS− | (13) |
The (reaction 8) between persulfide and cyanide yielding thiocyanate is used as the oldest method of sulfane sulfur detection called cyanolysis [77]. In this method, thiocyanate formed by cyanide attack on sulfane sulfur atom reacts then with ferric ions leading to the formation of a colored complex that can be assayed spectrophotometrically. Transfer of reactive sulfane sulfur atom from persulfides to nucleophilic acceptors, such CN−, sulfite or other thiols (RS−) can be catalyzed in biological conditions by rhodanese (TST). The reaction of persulfide with thiol, in which the outer sulfur atom is the target of RS− anion attack, leads to transpersulfidation reaction, where a new persulfide is formed (reaction 12). This process occurs when enzymes involved in H2S and sulfane sulfur metabolism (TST, MST, SQR) accept the sulfur atom forming their intermediate persulfide and then transfer sulfur atom to other acceptors. Moreover, the transpersulfidation process plays an important role in creation of protein -SSH groups. Low molecular weight persulfides, especially GSSH, are regarded as responsible for protein persulfidation [52].
On the other hand, when the inner sulfur atom in the persulfide is the target of RS− anion attack, in this reaction H2S is released and mixed disulfide is formed (reaction 13) [65,78]. This mechanism is preferred by small, low molecular weight persulfides that in reaction with thiol form mainly intermolecular disulfide bonds and release H2S. In the case of MST persulfide (MST-SSH), it has been documented that H2S can be released only by the small protein Trxred or DHLA. Both these compounds as dithiols have a close pair of -SH groups and during the reaction one of the two thiols forms a persulfide (so it is a transpersulfidation reaction), which is then attacked at the inner sulfur by the vicinal thiol leading to the formation of intramolecular disulfide of Trxox or LA, respectively (Figure 8) [79]. It seems that the reduction of protein persulfides by dithiols can also occur in other persulfidated proteins. It has been demonstrated that the activity of yeast aldehyde dehydrogenase (ALDH) inhibited by RSS was recovered much better by DHLA and dithiothreitol (DTT) than by GSH [80].
설파이드와 시안화물이 반응하여 티오시안산염을 생성하는 반응(반응 8)은 시아노리시스라고 불리는 가장 오래된 설파이드 황 검출 방법으로 사용됩니다[77]. 이 방법에서는, 설파이드 황 원자에 대한 시안화물의 공격에 의해 형성된 티오시안산염이 철 이온과 반응하여 분광광도계로 분석할 수 있는 착색된 복합체를 형성합니다. 반응성 황화수소 원자가 퍼설파이드에서 친핵성 수용체(CN-, 아황산염 또는 기타 티올(RS-) 등)로 이동하는 것은 로다나제(TST)에 의해 생물학적 조건에서 촉매될 수 있습니다. 외부 황 원자가 RS- 음이온 공격의 대상이 되는 티올과 퍼설파이드의 반응은 새로운 퍼설파이드가 형성되는 트랜스퍼설파이드화 반응을 일으킵니다(반응 12). 이 과정은 H2S와 설파인 황 대사에 관여하는 효소(TST, MST, SQR)가 황 원자를 받아 중간체인 퍼설파이드를 형성한 다음, 다른 수용체에 황 원자를 전달할 때 발생합니다. 또한, 트랜스퍼설파이드화 과정은 단백질 -SSH 그룹의 생성에 중요한 역할을 합니다. 저분자량 퍼설파이드, 특히 GSSH는 단백질 퍼설파이드화에 관여하는 것으로 알려져 있습니다 [52].
반면에, 이황화물의 내부 황 원자가 RS− 음이온의 공격 대상이 되면, 이 반응에서 H2S가 방출되고 혼합 이황화물이 형성됩니다(반응 13) [65,78]. 이 메커니즘은 주로 분자간 이황화 결합을 형성하고 H2S를 방출하는 티올과 반응하는 저분자량 작은 과황화물에 의해 선호됩니다. MST 과황화물의 경우(MST-SSH), H2S가 작은 단백질 Trxred 또는 DHLA에 의해서만 방출될 수 있다는 사실이 문서화되었습니다. 디티올과 같은 이 두 화합물들은 -SH 그룹의 쌍을 이루고 있으며, 반응 중에 두 티올 중 하나가 퍼설파이드를 형성합니다(즉, 트랜스퍼설파이드 반응입니다). 그러면 인접한 티올에 의해 내부 황에 공격되어 각각 Trxox 또는 LA의 분자 내 디설파이드가 형성됩니다(그림 8) [79]. 디티올에 의한 단백질 퍼설파이드의 감소는 다른 퍼설파이드 단백질에서도 일어날 수 있는 것으로 보입니다. RSS에 의해 억제된 효모 알데히드 탈수소효소(ALDH)의 활성이 GSH보다 DHLA와 디티오트레이톨(DTT)에 의해 훨씬 더 잘 회복된다는 것이 입증되었습니다[80].
Figure 8. Endogenous dithiols: Trx and lipoic acid (LA) are responsible for release of H2S from MST persulfide (MST-SSH).
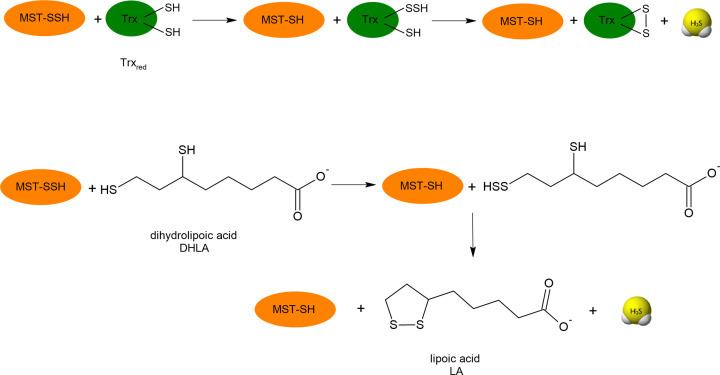
DHLA (reduced form of lipoic acid)
As mentioned earlier, persulfides show an electrophilic character when exist in their protonated form (RSSH). Considering that based on pKa value at physiological pH conditions in the cells anionic perthiol species (RSS−) shall prevail over RSSH forms, it seems that in biological processes nucleophilic character of persulfides dominates over electrophilic.
Synthesis and endogenous occurrence of RSSH
Reactivity and properties of persulfides have been studied using various low molecular weight compounds or protein models. The synthesis of a few organic persulfides, including benzyl persulfide (BnSSH), trityl persulfide (TrtSSH) and adamantyl persulfide (AdSSH) has been described [81–83]. Details of the synthesis of these compounds from acetyl sulfenyl chloride and appropriate thiols have been given in the papers mentioned above. Using these persulfide models, some conclusions regarding structure–reactivity relationship can be drown. For example, it has been established that acidic persulfides are more prone to decomposition than the less acidic ones [82]. Moreover, these studies have also revealed that persulfides can release H2S under the influence of reducing agents [83].
Apart from those model of persulfides which rather are not present in biological conditions, GSSH and CysSSH are the well-known and widespread low molecular weight persulfides in the cells. The most popular, standard method to generate low molecular weight persulfides, such as GSSH or CysSSH, in vitro is the reaction between a disulfide (GSSG or CysSSCys) and Na2S or between sulfenic acids (RSOH) and Na2S [66,67,78,80,84]. This technique is fast and easy, however, it has some weak points, namely it requires high concentrations of the used disulfides and often thiol and sulfide can be found in the reaction mixture apart from persulfides.
Both biologically important persulfides, GSSH and CysSSH, just like other compounds of this kind, are quite unstable what makes them a difficult objects to study. The main products of decomposition of persulfides include thiols, polysulfides and elemental sulfur (S8). H2S is not a direct product of decomposition, however, in the presence of thiols it can also be formed [78]. The reactivity of biological persulfides CysSSH and GSSH should be similar to the reactivity of other low molecular weight persulfides (RSSH) mentioned above [85]. A study by Benchoam et al. examining the acidity and nuclephilic reactivity of persulfides performed using GSSH confirmed it [67].
In vivo persulfides are produced mainly in reactions catalyzed by CBS/CSE and SQR; however, glutathione reductase (GR) can also be responsible for GSSH formation (Figure 9). CBS and CSE can convert cystine (CysSSCys) to CysSSH called also thiocysteine [86]. Thiocysteine can give its sulfane sulfur atom to GSH, the concentration of which in cells is high, forming GSSH (transpersulfidation). As mentioned above, SQR is an enzyme involved in H2S oxidation in mitochondria and it can create GSSH by transferring its sulfane sulfur atom to GSH (Figure 9) [87]. GSSH can also be produced from glutathione polysulfide in reaction catalyzed by GR [61]. CysSSH and GSSH have been detected in various cell cultures including HEK293 [88], A549, SH-SY5Y, C6 [61] and HeLa [57].
RSSH의 합성과 내생적 발생
과황화물의 반응성과 특성은 다양한 저분자량 화합물 또는 단백질 모델을 사용하여 연구되었습니다. 벤질과황화물(BnSSH), 트리티일과황화물(TrtSSH), 아다만틸과황화물(AdSSH)을 포함한 몇 가지 유기과황화물의 합성에 대한 설명이 있습니다 [81-83]. 아세틸설페닐클로라이드와 적절한 티올로부터 이러한 화합물을 합성하는 방법에 대한 자세한 내용은 위에서 언급한 논문에서 확인할 수 있습니다. 이러한 과황화합물 모델을 사용하면 구조-반응성 관계에 관한 몇 가지 결론을 도출할 수 있습니다. 예를 들어, 산성 과황화합물은 덜 산성인 과황화합물보다 분해되기 쉽다는 것이 밝혀졌습니다 [82]. 또한, 이러한 연구들은 과황화합물이 환원제의 영향으로 H2S를 방출할 수 있다는 사실도 밝혀냈습니다 [83].
생물학적 조건에서는 존재하지 않는 퍼설파이드 모델과는 별개로, GSSH와 CysSSH는 세포에서 잘 알려져 있고 널리 퍼져 있는 저분자량 퍼설파이드입니다. 체외에서 저분자량 퍼설파이드를 생성하는 가장 인기 있고 표준적인 방법은 이황화(GSSG 또는 CysSSCys)와 Na2S 또는 설펜산(RSOH)과 Na2S 사이의 반응입니다 [66,67,78,80,84]. 이 기술은 빠르고 쉽지만, 약점이 있습니다. 즉, 사용되는 이황화물의 농도가 높아야 하고, 종종 티올과 황화물이 퍼설파이드를 제외한 반응 혼합물에서 발견될 수 있습니다.
생물학적으로 중요한 두 개의 퍼설파이드인 GSSH와 CysSSH는 다른 이런 종류의 화합물들처럼 매우 불안정하기 때문에 연구하기가 어렵습니다. 퍼설파이드의 주요 분해 산물에는 티올, 폴리설파이드, 그리고 원소 황(S8)이 포함됩니다. H2S는 분해의 직접적인 산물은 아니지만, 티올이 존재할 때 형성될 수도 있습니다 [78]. 생물학적 퍼설파이드 CysSSH와 GSSH의 반응성은 위에서 언급한 다른 저분자량 퍼설파이드(RSSH)의 반응성과 유사해야 합니다 [85]. 벤촐람(Benchoam) 등의 연구에서 GSSH를 사용하여 퍼설파이드의 산성도와 친핵성 반응을 조사한 결과, 이를 확인했습니다 [67].
생체 내의 퍼설파이드(persulfide)는 주로 CBS/CSE와 SQR에 의해 촉매 작용을 받는 반응에서 생성되지만, 글루타티온 환원효소(glutathione reductase, GR)도 GSSH 형성에 관여할 수 있습니다(그림 9). CBS와 CSE는 시스틴(cystine, CysSSCys)을 티오시스테인(thiocysteine)이라고도 불리는 CysSSH로 전환할 수 있습니다[86]. 티오시스테인은 세포 내 농도가 높은 GSH에 설파인 황 원자를 제공하여 GSSH(트랜스퍼설파이드화)를 형성할 수 있습니다. 위에서 언급한 바와 같이, SQR은 미토콘드리아에서 H2S 산화에 관여하는 효소이며, 설파인 황 원자를 GSH로 옮김으로써 GSSH를 생성할 수 있습니다(그림 9) [87]. GSSH는 또한 GR [61]에 의해 촉매 작용을 받는 반응에서 글루타티온 폴리설파이드로부터 생성될 수 있습니다. CysSSH와 GSSH는 HEK293 [88], A549, SH-SY5Y, C6 [61] 및 HeLa [57]를 포함한 다양한 세포 배양에서 검출되었습니다.
Figure 9. Endogenous synthesis of low molecular weight and protein persulfides.
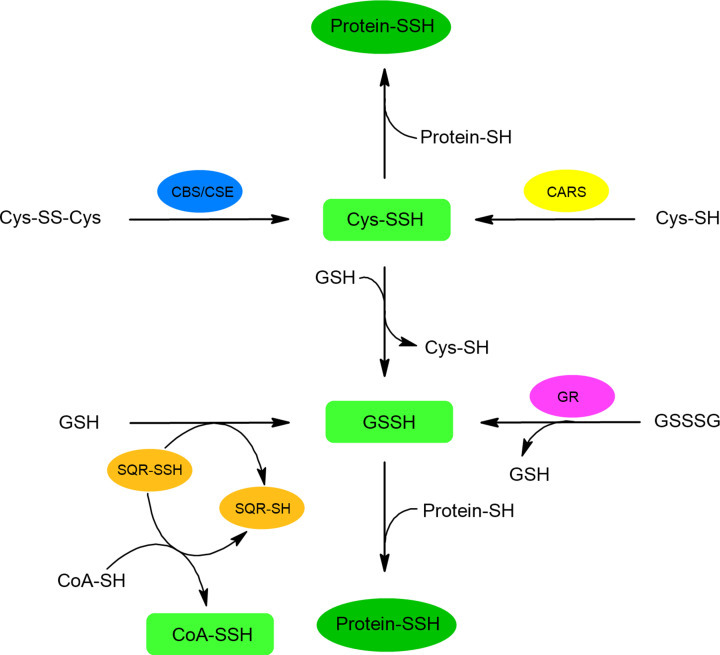
Biological persulfides are produced mainly in reactions catalyzed by CBS and CSE that convert cystine (CysSSCys) to cysteine persulfide (CysSSH, thiocysteine). CARS catalyzes the formation of CysSSH from CysSH during translation process. SQR, involved in mitochondrial oxidation of H2S, produces glutathione persulfide (GSSH) by transferring sulfane sulfur atom to GSH. SQR can also form persulfide of CoA-SH. Moreover, GR can produce GSSH from glutathione polysulfide (GSSSG).
Recently, it has been found that the formation of another important endogenous thiol CoA-SH in its persulfide form can also be mediated by SQR. A study by Landry et al. using human SQR supported the conversion of CoA-SH to CoA-SSH indicating that this enzyme can utilize CoA-SH as an alternative sulfur acceptor. CoA-SSH formed in this reaction inhibits butyryl-CoA dehydrogenase (ACADS) and butyrate oxidation [89,90]. According to literature data, the GSSH concentration in animal tissues is in a range 10–100 μM in most organs, while the highest concentration of approximately 150 μM was found in the brain [61,91]. The level of CysSSH has also been estimated in μM range and the level of both endogenous persulfides (CysSSH and GSSH) was dependent on the availability of sulfur compounds (e.g. methionine) in the diet [61] as well as on the expression of enzymes involved in their synthesis [61,88]. The presence of GSSH and CysSSH has also been demonstrated in various human tissues, including plasma [61,92], lung resident cells and epithelial lining fluid [93], sputum [94], aqueous and vitreous humor [95], tear, saliva, and nasal discharge [96]. Information concerning enzymes involved in physiological formation of persulfides is summarized in Table 1.
Protein persulfides
Protein persulfides (protein-SSH) are formed mainly by reaction with low molecular weight persulfides, such as GSSH or CysSSH that means in a transpersulfidation reaction. It seems that GSSH is the most widespread low molecular weight persulfide, and it plays a dominant role in the protein persulfide forming reaction. In this process called protein persulfidation or sulfuration, the protein CysSH residue is oxidized to CysSSH yielding protein persulfide. This process is considered as a reversible, post-translational, covalent modification of proteins which plays an important regulatory and protective role [52,97–99]. Regarding the mechanism of protein persulfidation, two pathways are possible: protein CysSH reacts with RSSH or another compound containing sulfane sulfur atom or protein CysSH oxidized to sulfenic acid, disulfide, mixed disulfide or nitrosothiol is persulfidated with H2S [100].
More recently, it has been documented that protein persulfides are formed in vivo also in reactions catalyzed by cysteine persulfide synthase called cysteinyl-tRNA synthetase (CARS). This enzyme catalyzes the formation of CysSSH from CysSH as a substrate during translation process [88]. CysSSH-tRNA formed by CARS can be a substrate for protein synthesis in ribosomes and can lead to persulfidation of newly synthesized proteins. It means that protein persulfidation can be a post-translational modification and a translation-coupled process. It seems that both mechanisms play important roles in maintenance of proper physiological level of protein persulfidation.
Polysulfides
Polysulfides including inorganic hydrogen polysulfides (H2Sn, n>2) and organic polysulfides (RSnH and RSnR, where n>2), just like persulfides, contain reactive sulfane sulfur atom in their structure. This kind of RSS became the focus of interest after it has been revealed that they exist endogenously and participate in RSS redox signaling. The great interest in these forms is evidenced by the fact that the search for the keyword ‘polysulfide’ in PubMed currently results in over 2900 records (Figure 1). In many cases, polysulfides are being considered together with persulfides since both H2S-related RSS are linked to some physiological and pathological processes. Moreover, some methods used for persulfide and polysulfide determination do not distinguish between these two kinds of RSS. However, from chemical point of view, there are a lot of points that differ these two kinds of RSS and this is why polysulfides should be described separately. In recent years, several new methods have been developed to selectively determine polysulfides in biological samples [101–104].
Synthesis of polysulfides
Biosynthesis of inorganic and organic polysulfides is closely related to the synthesis of persulfides. Inorganic polysulfides were first detected in mouse brains by Kimura et al. [105]. Their studies revealed the presence of H2S3 in the cytosol of brain cells. The enzyme responsible for the production of these polysulfides was recognized as MST, which uses 3-MP as a substrate (Figure 10A). Besides H2S3, H2S2 and H2S5 were also identified in the studied brain samples; however, they were minor products [105]. The second enzyme that can contribute to the production of polysulfides is CARS mentioned above in the persulfide section. It was shown by Akaike et al. that CARS could effectively catalyze the production of not only Cys-SSH but also Cys-SnSH using Cys-SH as a substrate (Figure 10B) (Table 1) [88]. Cys-SnSH formed in this reaction either in the free form or incorporated in protein structure is an example of organic polysulfides; however, on the other hand, it can be a precursor of inorganic H2Sn.
Figure 10. Reactions responsible for polysulfide synthesis.
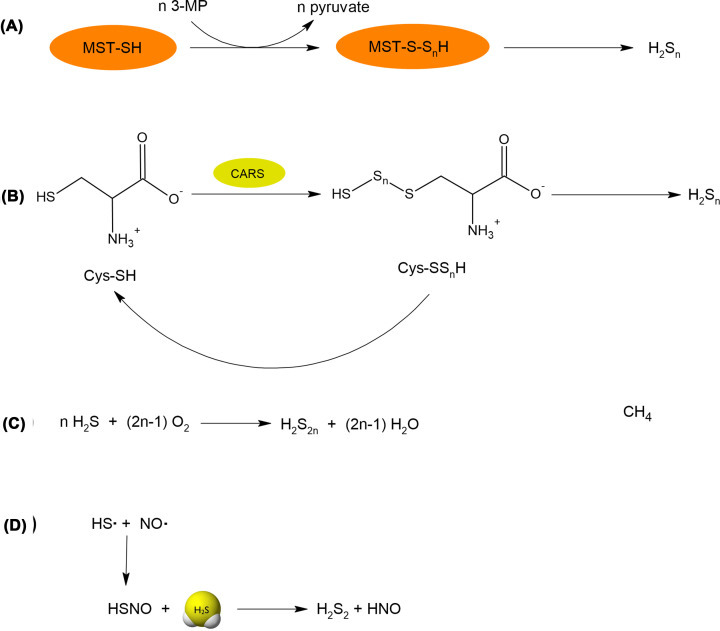
(A) Inorganic polysulfides are produced by MST, which uses 3-MP as a substrate. (B) CARS using L-cysteine as a substrate effectively produces cysteine hydropolysulfides (CysSSnH), which can release inorganic polysulfides. (C) An example of nonenzymatic generation of inorganic polysulfides by simple oxidation of H2S. (D) Inorganic polysulfides can also be produced in reaction of H2S with thionitrous acid (HSNO) formed in reaction of two radicals (NO• and HS•).
Inorganic polysulfides can also be generated non-enzymatically by oxidation of H2S (Figure 10C) [106]. This reaction is rather slow, that is why it is not an important source of polysulfides inside the cells. However, this reaction is often responsible for contamination of commercially available sulfides by inorganic polysulfides. Non-enzymatic reactions generating H2S2 include also the reaction between radical forms of two gasotransmiters: H2S and nitric oxide (NO). Thionitrous acid (HSNO) formed in this reaction can next react with the H2S molecule leading to H2S2 and nitroxyl (HNO) (Figure 10D) [107]. Since NO and H2S both occur in the cells, this reaction can be at least partially responsible for the production of polysulfide in the mammalian body.
Polysulfides can also be generated in heme-catalyzed reactions. As presented in Figure 5, H2S can be oxidized by heme of Mb and Hb or other hemoproteins to polysulfides bound to iron atom [53]. Free inorganic polysulfides can be next released spontaneously under the influence of endogenous reductors. Olson et al. found that cytosolic form of SOD could catalyze the oxidation of H2S yielding H2S2, H2S3 and H2S5 [55]. Another pathway of polysulfide formation involves the reaction between H2S and RSSR, which results in the production of RSSSR together with RSH and RSSH [108].
Properties of polysulfides
Polysulfides are rather unstable species. According to the results of experiments performed with glutathione polysulfide (GSSnSG) [88] and alkylated polysulfides (RSSnSR) [109], sulfur-sulfur bonds in such compounds are susceptible to hydrolysis at physiological and alkaline conditions. The hydrolysis of polysulfides results in formation of reduced thiols and polysulfenic acids or hydropolysulfides and sulfenic acids (reactions 14-19).
| RSSnSR(n>1)+H2O⇄RSH+RSnSOH | (14) |
| RSSnSR(n>1)+H2O⇄RSSnH+RSOH | (15) |
| RSSR+H2O⇄RSH+RSOH | (16) |
| HSSR+H2O⇄H2S+HSOH | (17) |
| HSnH+H2O⇄H2Sn−1+HSOH | (18) |
| HSnH+H2O⇄H2S+(n−1)8 S8 | (19) |
This high susceptibility of polysulfides (RSSnSR, n≥1) to hydrolysis distinguishes them from disulfides (RSSR), which undergo a hydrolysis reaction only under strong alkaline conditions. The alkaline hydrolysis of RSSR leads to production of reduced thiol (RSH) and sulfenic acid (RSOH) [104,110]. At neutral pH, the equilibrium of the reaction of RSSR hydrolysis is shifted to the left, so disulfides at physiological pH rather do not undergo hydrolysis.
Inorganic polysulfides (H2Sn) are characterized by low stability at neutral pH. Moreover, it has been documented that various salts of inorganic polysulfides often used as polysulfide donors, differ in stability. For example, Na2S2 and Na2S4 were found to be relatively stable compounds but Na2S3 can readily decompose forming a mixture of Na2S2 and Na2S4 [111]. As long as the first product of this decomposition is consistent with the (reaction 18) (sulfenic acid is the second product), the formation of Na2S4 may be surprising. It can be explained by taking into account the suggestions of some authors that H2Sn can also decompose to H2S and elemental sulfur according to (reaction 19) [102]. In this way, HS− ion and elemental sulfur present in the solution as a result of H2Sn decomposition can combine forming a new polysulfide (as Na2S4, more stable than Na2S3).
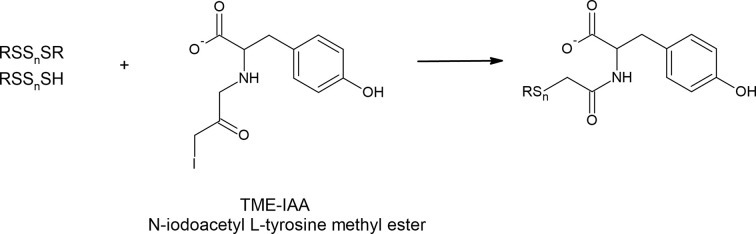
It has been shown that various alkylating reagents including IAA, NEM, mBrB or dimedone can enhance the hydrolysis of polysulfides [104,112]. During the hydrolysis, there is some equilibrium between RSSnSR and its products, i.e. RSSH and RSSOH. Alkylating agents reacting with RSSOH or RSSH shift this equilibrium to the right leading to enhanced decomposition of RSSnR. Interestingly, it was revealed that such alkaline hydrolysis of polysulfides could be significantly prevented by hydroxyl group-containing compounds such as β-(4-hydroxyphenyl)ethyl iodoacetamide (HPE-IAA), tyrosine or serine [104,112]. This observation suggests that moieties of naturally occurring amino acids containing hydroxyl groups (tyrosine and serine) may be involved in maintaining the polysulfide stability and in the regulation of their homeostasis.
The finding that the hydroxyl group-containing agents efficiently protect polysulfides from hydrolytic degradation led the Akaike’s research group to develop the new alkylating agent namely, N-iodoacetyl L-tyrosine methyl ester (TME-IAA) (Figure 11). This compound specifically traps and stabilizes polysulfides and protects them against hydrolysis, and used with mass spectrometry allows for successful polysulfide analysis [103,104]. Kasamatsu et al. noted a loss of nearly 40% of glutathione tetrasulfide after 1 h of incubation at physiological pH while in the presence of mBrB, the total loss reached nearly 70% of the initial amount of tetrasulfide. In contrast, in the presence of TME-IAA this degradation was not observed [103]. The same authors reported higher amounts of various polysulfides and smaller amounts of sulfides in mouse tissues when they were detected with TME-IAA compared with amounts determined with HPE-IAA. It confirms that the determined level of RSS, especially polysulfides and H2S can be influenced by specific alkylating agents.
Figure 11. N-iodoacetyl L-tyrosine methyl ester (TME-IAA) is an alkylating agent which reacts with polysulfides and protects them from hydrolytic degradation.
Reactive sulfur species in diseases
As RSS play an important regulatory and protective role, it is obvious that disruption of their homeostasis can lead to some pathological conditions. It has been quite well documented that many diseases are associated with a disturbance of the endogenous RSS pool, most often with its decrease. Development of specific methods for the determination of H2S and especially persulfides and polysulfides enabled the study of their significance in some diseases. However, it is worth mentioning that an assay of any kind of RSS is a challenge due to their reactivity and instability. Many factors, including, pH, oxygen content, presence of oxidants or reductors can influence the final assay results. Moreover in the case of persulfides, the problem with appropriate stable standards exists. In the case of poly- or persulfides there are only limited studies reporting the levels of these RSS in physiological conditions (Table 2).
Table 2. Disturbances of RSS level and enzymes expression in some pathologies.
PathologyRSS or enzymeTissueHealthDiseaseReference
| Cardiovascular disease | |||||
| Spontaneous hypertension | H2S | Rat plasma | 48 μM | 20 μM* | [116] |
| Portal hypertension | H2S | Human plasma | 43 μM | 42 μM Child-Pugh score A 33 μM* Child-Pugh score B 22 μM* Child-Pugh score C | [115] |
| Chronic kidney disease (CKD) | |||||
| Hemodialysis | H2S | Human plasma | ≈15 μM | ≈11 μM* | [119] |
| CKD (non-dialysed) | H2S | Human plasma | 14 μM | 7 μM* | [43] |
| Hemodialysis and diabetic nephropathy | H2S | Human plasma | 57 μM | 23 μM* CKD + diabetic nephropathy 29 μM* μM CKD | [120] |
| Metabolic disorders | |||||
| Overweight | H2S | Human plasma | 39 μM | 22 μM* | [122] |
| Overweight and DM2 | H2S | Human plasma | 39 μM | 10 μM* | [122] |
| DM2 | H2S | Human plasma | 130 μM | 110 μM* | [125] |
| Streptozotocin-induced diabetes | H2S | Rat plasma | 78 μM | 45 μM* | [125] |
| Fructose-induced diabetes | H2S | Mice plasma | ≈1.3 μM | ≈1.0 μM* | [126] |
| Fructose-induced diabetes | H2S | Mice heart | ≈0.20 μM | ≈0.14 μM* | [126] |
| Streptozotocin-induced diabetes | H2S CSE expression CBS expression | Rat liver | ≈37 μM | ≈65 μM* ↑* ↑* | [128] |
| H2S CSE expression CBS expression | Rat pancreas | ≈30 μM | ≈45 μM* ↑ ↑* | [128] | |
| Diabetic retinopathy | CysSSH | Human aqueous humor | 0.14 | 0.28*μM | [95] |
| Diabetic retinopathy | CysSSH | Human vitreous humor | 0.07 | 0.18*μM | [95] |
| Diabetic retinopathy | GSSSG | Human aqueous humor | 0.07 | 0.19* μM | [95] |
| Neurodegenerative diseases | |||||
| Mouse model of Parkinson’s disease | H2S | Mice striatum | ≈ 39 μM | ≈ 29 μM* | [129] |
| Mouse model of Parkinson’s disease | H2S | Mice plasma | ≈ 22 μM | ≈ 8.5 μM* | [129] |
| Parkinson disease | Parkin persulfides | Human striatum | control value | 40% of the control value | [154] |
| Alzheimer disease | H2S | Human plasma | 45 μM | 34 μM* | [130] |
| Alzheimer disease | H2S | Human plasma | below 1 μMA | below 1 μMA | [131] |
| Alzheimer disease | Bound sulfur (persulfides) | Human plasma | below 1 μMA | above 1 μMA ↑* | [131] |
| Amyotrophic lateral sclerosis (ALS) | H2S | Human cerebrospinal fluid | 3.6 mg/l | 7.5 mg/l* | [132] |
| Down syndrome | Thiosulfate | Human urine | 3.3 mmol/mol creatinine | 7.6 mmol/mol creatinine* | [133] |
| Rat model of Down syndrome | CBS expression | Brain | ↑* | [135] | |
| Respiratory diseases | |||||
| Asthma in children | H2S | Human serum | 53 μM | 44 μM | [137] |
| Asthma | H2S | Human serum | 152 μM | 280 μM* non-severe 283 μM*severe | [138,142] |
| Asthma | H2S | Human sputum | 11 μM | 27 μM* non-severe 28 μM* severe | [138,140] |
| COPD | H2S | Human sputum | 20 μM smokers 12 μM non-smokers | 32 μM* stable COPD 50 μM* acute exacerbation COPD | [141] |
| COPD | H2S | Human serum | 91 μM smokers 91 μM non-smokers | 149 μM* stable COPD 49 μM acute exacerbation COPD | [141] |
| COPD | GSSH | Bronchial epithelial | 71 nmol/g protein | 51 nmol/g protein* | [93] |
| Bronchial epithelial CysSSH | 31 nmol/g protein | 21 nmol/g protein* | [93] | ||
| Asthma–chronic obstructive pulmonary disease overlap | Persulfides and polysulfides | Human sputum | 16 arbitrary units (AU) | 5.2* arbitrary units (AU) | [94] |
| COVID-19 | |||||
| COVID-19 | H2S | Human plasma | 0.31 μM | 0.19* μM | [144] |
| COVID-19 | Bound sulfur | Human plasma | ≈ 0.50 μM | ≈ 0.62 μM NS | [144] |
| Osteoarthritis | |||||
| Osteoarthritis | H2S MST expression CBS expression | Human cartilage | 0.46 nmoles/g of cartilage | 0.06 nmoles/g of cartilage* ↓* ↓ NS | [143] |
| Osteoarthritis | H2S | Human serum | 69 μM | 45 μM NS | [143] |
*
statistically significant difference between control healthy and pathology; NS- non-significant difference; A it was difficult to read the exact value.
Some methods of H2S determination use acidification during procedure that may promote release of H2S from acid-labile sulfur and overestimate the concentration of free H2S. For this reason, it is difficult to talk about the credible and absolute value of individual RSS. However, most reports present the levels of RSS determined using the same method for control and tested biological samples, therefore, based on these data comparisons may be done. Data reporting disturbances in the RSS levels in pathological conditions refer to both low molecular weight RSS, such as H2S, GSSH and CysSSH as well as to the total protein persulfidation or persulfidation of specific proteins.
Disturbances in H2S level
The role of H2S in the proper functioning of the body is well understood and disturbances in plasma H2S levels or its synthesis in some tissues have been described in many pathologies. In this review only a few such examples will be presented. Most authors estimated the concentrations of free H2S in the plasma of healthy humans within the range 15–90 µM. Rarely, the higher level of 130–150 µM was reported. Surprisingly, some authors presented that the level of free H2S in healthy human plasma was as low as >1 µM or less [131,144]. The reason of such a big discrepancy of the reported values is the fact that various methods were used to evaluate the amount of H2S. The most commonly used methods include colorimetric method based on the methylene blue formation, fluorometric assays using different fluorescence dyes (e.g. mBrB, HSip-1, 7-nitro-1,2,3-benzoxadiazole [NBD] and many others) and electrochemical determinations using ion-selective electrodes. It seems that the colorimetric method gives higher values of H2S compared with more specific fluorescent methods. As mentioned earlier, RSS are very difficult to assay because of their instability and due the fact that different forms of RSS coexist. Bound sulfide is released as H2S from its bound form (persulfides) under reducing agents or in acidic environments (acid-labile sulfur). Therefore, during the assay procedure some kind of bound sulfur can be determined as free H2S. Moreover, H2S level in plasma can be influenced by its interaction with plasma proteins or erythrocytes; therefore, the time of collection and preparation method of plasma can also influence the estimated level of H2S. The next factor which affects the detected H2S concentration is pH. In physiological pH, H2S exists mainly as HS− and acidification of the samples facilitates the formation of H2S, which can be assayed by diffusion but contributes to bound sulfur release. The pH value is very important also in the determination with the ion-selective electrodes which are specific to the S2− ions and, therefore, alkalization of the sample plays a crucial role. Another factor affecting the end value of the estimated H2S level is a good standard. Most often used standards, i.e. Na2S and NaHS are very susceptible to oxidation and may contain a lot of sulfane sulfur. When the standard with a relatively high sulfane sulfur content is used for S2− calibration curve, the estimated values can be incorrect. Therefore, comparisons of the H2S and other RSS levels between healthy and pathological samples may be done only when the same method is used for control and tested samples.
A large body of evidence shows that pathogenesis of a variety of vascular diseases is connected with the down-regulation of the H2S pathway. In plasma of patients with hypertension as well as in patients with heart failure or ischemic heart disease, the level of H2S is reduced compared with the plasma of control patients [113–116]. For example, the H2S plasma levels of portal hypertension patients were 33.5 μM (Child–Pugh score B) and 22.2 μM (Child–Pugh score C) compared with 43.5 μM detected in control subjects (Table 2) [115]. Moreover, the plasma H2S level in the patients with vascular diseases was correlated with the severity of symptoms—it was significantly lower in patients with acute myocardial infarction and unstable angina compared with patients with a stable form of the disease [117]. Similarly, markedly reduced H2S plasma level has been reported in chronic kidney disease (CKD) patients and animal models of CKD [43,118,119]. For example, Li et al. reported the H2S level of 56.6 µM in healthy patients, whereas the level of H2S in patients with CKD was 22.9 μM and 29.1 μM in patients with or without diabetic nephropathy, respectively [120]. Additionally, several lines of evidence suggest that H2S can exert protective effects against CKD progression [121]. Disturbances in the synthesis and catabolism of H2S leading to reduced H2S plasma level were shown in overweight patients (22.0 μM vs. 38.9 μM in controls) and in patients with Type 2 of diabetes mellitus (DM2) (10.5 μM vs. 38.9 μM in controls) [122,123]. Moreover, it was found that low H2S levels in patients with DM2 correlated with a higher risk of cardiovascular disease [124]. Similar results were obtained in rat and mouse animal models, where the level of H2S was lower in individuals with induced diabetes, both in plasma and in some organs, such as the heart, vessels, kidneys, bone marrow and central nervous system [123,125,126]. On the other hand, studies in a model of induced diabetes in rats showed a higher concentration of H2S in the liver and pancreas compared with healthy control, which was accompanied by an increase in the expression of enzymes responsible for H2S synthesis in these organs [127,128].
Accumulating evidence demonstrated an important role of H2S in neurological diseases. Parkinson’s disease (PD) is manifested by motor system abnormalities, and decreased H2S production caused by down-regulation of CBS in the substantia nigra in PD animal models has been documented. Moreover, a decline of H2S level was observed in the striatum and plasma in the mouse models of PD (Table 2)[129]. Alzheimer’s disease (AD) leads to the cognitive dysfunction and loss of memory. It has been reported previously that plasma level of free H2S tends to be decreased in AD patients (34 μM vs. 45 μM in controls) [130], while another study published recently has not confirmed this suggestion. The latter study showed no differences in the free plasma H2S level in AD patients compared with the plasma of control individuals; however, disturbances in the total sulfide (acid-labile and bound) have been found by these authors in the AD patients [131]. Amyothrophic lateral sclerosis (ALS) is a neurodegenerative disease causing a selective degeneration of upper and lower motor neurons. Higher levels of H2S have been found in the spinal fluid of ALS patients when compared with the controls (7.53 mg/l vs. 3.64 mg/l in controls) (Table 2). Similarly, an increase in the tissue levels of H2S was also observed in a mouse model of ALS [132]. An elevated H2S production and increased urine level of thiosulfate, the main product of H2S catabolism, were also found in Down syndrome, a disease caused by trisomy of chromosome 21 [133–135]. The role of RSS in neurological diseases has been recently described in detail [136].
In the case of asthma and chronic obstructive pulmonary disease (COPD), deviations in the physiological level of H2S were also documented, however, the published data are divergent. Some studies found that serum H2S levels in asthmatic children were significantly decreased compared with those in non-asthmatic children (44.2 μM vs. 52.6 μM) [137], while other authors reported elevated serum H2S levels in severe and non-severe asthmatic patients compared with healthy subjects (280–283 μM vs. 152 μM) [138]. The diminished expression of CSE was also reported in the lung of mice in animal model of asthma [139]. There can be some possible reasons for this discrepancy including different methods used for H2S assay and various factors which can influence the H2S level, such as exacerbation of disease and degree of inflammation. In parallel, the level of H2S in asthmatic patients measured in the sputum was clearly elevated compared with healthy donors: 26–27 μM vs.11.4 μM [138,140] and 31.9 μM vs 19.7 μM [141,142], (Table 2). It seems that sputum H2S particularly correlates with the degree of airflow limitation and it has been suggested that H2S has potential as a novel biomarker for COPD and asthma [140]. It has also been found that mRNA and protein levels of MST were significantly reduced in cartilage of patients suffering from osteoarthritis (OA) compared with healthy donors. It was accompanied with a lower H2S biosynthesis in OA cartilage; however, no differences were found in the H2S concentration in serum from OA patients and OA-free donors (Table 2) [143].
More recently Dominic et al. studied the levels of H2S in plasma of COVID-19 patients and compared them with healthy controls. They found that plasma levels of free H2S were significantly reduced in patients suffering from COVID-19 in comparison with healthy controls (0.31 μM vs. 0.19 μM) [144]. It was true in both Caucasian and African American patients. The total sulfide levels were also decreased in the plasma of patients affected by COVID-19 (1.37 μM vs. 1.15 μM), just like plasma NO levels. The authors described also a case study of a single patient who was initially a control subject, but a few days later contracted a COVID-19 infection. The total sulfide level in this patient was significantly reduced during COVID-19 infection (from 1.11 μM before infection to 0.85 μM during infection); however following antiviral therapy with remdesivir the total sulfide level increased to 1.07 μM on the 5th day of therapy and to 1.45 μM 14 days post-treatment. The authors performed receiver-operating characteristic analysis (ROC) to determine the accuracy of H2S as an indicator of COVID-19. The analysis of plasma H2S between COVID-19 patients and controls revealed areas under the curve (AUC) of 0.87 (P<0.0001) for free sulfide and 0.75 (P<0001) for total sulfide. It suggests that especially free H2S can be regarded as a strong predictor of COVID-19 in an overall population; however, the AUC values were a little higher for Caucasian than for African American patients [144]. Besides free and total sulfide, the authors assayed also acid-labile sulfur, which can release free H2S under acidic conditions and consists mainly of iron-sulfur clusters contained in iron-sulfur proteins. The level of acid-labile sulfur was significantly decreased in patients with COVID-19 when compared with healthy controls (0.45 μM vs. 0.59). This study for the first time reported the reduction in two biochemical sulfide forms (H2S and acid-labile sulfur) in COVID-19 patients compared with healthy controls; however, it is only a single study and more research is needed to confirm this thesis. On the other hand, a study by Mete et al. revealed the decrease in total and native (reduced) thiols in plasma of patients with COVID-19 compared with healthy controls (282.5 μM vs. 410.5 μM for total and 169.7 μM vs. 279.0 μM for reduced thiols) [145]. This fact suggests that in the course of COVID-19 the availability of substrates for H2S synthesis: L-cysteine and homocysteine is limited what seems to confirm the diminished synthesis of H2S. Renieris et al. studied the level of H2S in serum of patients with COVID-19 on the 1st and 7th day after hospital admission. This study showed a correlation between the serum H2S level and the severity of the disease progression and final outcome. The mortality of patients with COVID-19 was significantly higher among patients with decreased H2S level, while in those who survived a higher serum level of H2S was detected on the 1st (≈195 μM for survivors vs. ≈130 μM for non-survivors) and 7th day after admission to hospital (≈190 μM for survivors vs. ≈75 μM for non-survivors) [146]. This study also revealed that serum H2S level was negatively correlated with the level of C-reactive protein (CRP) and IL-6 considered as the main and relevant parameter in predicting the most severe course of respiratory failure, lung injury and death in COVID-19 [147].
On the other hand, the above-cited study by Dominic et al. also performed analysis of correlation between the plasma H2S level and severity of COVID-19 course (estimated by cardiac injury, thrombosis and inflammatory) and found an increase in total H2S level in patients with severe course of COVID-19 compared with the patients with mild-to-moderately severe course of the illness [144]. These conflicting results could be due to a difference between plasma and serum, method of H2S estimation and other important factors affecting H2S level (e.g. age or accompanying diseases). It is difficult to estimate the impact of COVID-19 on plasma H2S level since most of patients admitted to the hospital due to SARS-CoV-2 infection suffer from other diseases associated with H2S disorders including DM, COPD, CKD or cardiovascular disease. In the study of Dominic et al., 75% of COVID-19 patients had hypertension while DM was recognized in 42% of patients [144]. In the Renieris study, the situation was clearer since in this group only 8% of patients suffered from DM and 21% from cardiovascular disease including chronic heart failure and coronary heart disease. Severe respiratory failure was the most frequent failure in this group of patients and affected nearly 34% of all patients [146]. The next aspect that should be taken into consideration is age of the study participants. In the study of Dominic et al., patients were in the wide age range 18–89, in the Renieris study there is an information that 47% of patients were 64 or more years old. As reported by Giustarini et al., the low molecular weight thiols and protein thiol/disulfide ratio declines in human plasma with age [148]. In the case of infection induced by SARS-CoV-2 with associated inflammation, this decline with age can be even more rapid. To clarify the correlation between plasma H2S levels, severity of COVID-19 and final outcome additional investigations are required.
Disturbances in low molecular weight persulfide and polysulfide levels
Like in the case of H2S, disturbances in the endogenous levels of low molecular weight persulfides and polysulfides can be associated with certain diseases, although this area is much less explored. For example, it has been found that patients with diabetic retinopathy show elevated levels of CysSSH in aqueous (0.28 μM vs. 0.14 μM) and vitreous (0.18 μM vs. 0.07 μM) humor compared with healthy patients [95]. In aqueous humor, an increased level of glutathione polysulfide (GSSSG) was also observed (0.19 μM vs. 0.07 μM in controls). It seems that in the eye, some RSS are up-regulated in DM, however, these disturbances were not reflected in persulfide levels in plasma of these patients [95].
A study by Numakura et al. was designed to investigate endogenous levels of low molecular weight persulfides in the lungs in patients with chronic obstructive pulmonary disease (COPD) and to compare them with control patients. They have found reduced levels of GSSH and CysSSH in lung-resident cells and epithelial lining fluid derived from COPD patients airways (Table 2) [93]. Similarly, a decrease in the level of total (nonprotein and protein) persulfides and polysulfides was observed in the sputum of the patients suffering from COPD overlapped with asthma when compared with healthy controls (5.2 arbitrary units [AU] vs. 16.4 AU) [94]. It is interesting especially in the context of previously cited reports about the elevated H2S levels in the sputum of asthmatic patients. It seems that in the case of asthma and COPD disturbances in the levels of H2S and persulfides may result from imbalance between H2S and its storage in the form of persulfides rather than up- or down-regulation of RSS synthesis; however, further studies are needed to clarify this problem.
In the COVID-19 cases, there was no specific research to estimate low molecular weight persulfides or polysulfides; however, one above-mentioned paper by Dominic et al. tested also the level of bound sulfane sulfur in the plasma of COVID-19 patients. This form of reactive sulfur releases H2S under reducing conditions and includes various persulfides and polysulfides. This study did not reveal significant differences in the level of bound sulfane sulfur in the plasma of patients with COVID-19 when compared with the controls (Table 2) [144]. It means that infection induced by SARS-CoV-2 affects only free or total H2S without the effect on the bound forms of sulfur. It is also possible that other analytical methods more specific to persulfides or polysulfides could be more appropriate to demonstrate a disturbance in the level of these RSS in patients with COVID-19.
Disturbances in protein persulfidation
Protein persulfidation is well described in the literature and disturbances in the level of protein persulfidation associated with various pathological conditions are much better known than those connected with the level of low molecular weight persulfides. This problem is particularly important as persulfidation plays a protective role and it most often affects protein activity. It makes protein persulfidation an important regulatory process, and especially in the case of specific protein targets, it can be essential in maintaining the health of the body and in treating some disease.
It has been reported that the total protein persulfidation decreases with aging. It is known that during aging an increase in reactive oxygen species (ROS) production with an accompanying decrease in antioxidant defense is observed [149]. It results in aggravation of oxidative damage and promotes development of age-related diseases, such as Parkinson’s disease and Alzheimer’s disease. Persulfides possessing powerful antioxidant properties and thiols that have weaker antioxidant properties but exist in higher concentrations in cells, are together an important cellular defense system against oxidative injury. Oxidation of cysteine -SH groups in proteins to sulfonic acids (-SO3H) can led to the loss of protein's activity, so protein persulfidation is regarded as a reversible modification and a kind of protection against irreversible oxidation [150]. Unfortunately, the levels of total plasma sulfane sulfur and total cysteine, the main substrate for RSS synthesis, decline with age [151]. Moreover, a study by Zivanovic et al. using rats of different ages found that protein persulfidation levels in brain extracts was reduced with age reaching approximately 50% in 24-month-old rats relative to the level of persulfidation observed in 1-month-old rats. It was accompanied by the reduced brain expression of CSE, CBS and MST. Additionally, these authors reported also an age-related decrease in protein persulfidation and expression of CSE and MST in the hearts of animals [152].
Aging is regarded as one of the main risk factors for development of neurodegenerative diseases. It has been shown that persulfidation process is dysregulated in some neurodegenerative disorders, such as PD and AD [136]. PD which is connected mainly with lesions in the motor nervous system, can be caused by a mutation of parkin protein gene. Parkin acts as a ubiquitin E3 ligase and its activity has been documented to decrease in PD [153]. Then, it has been demonstrated that parkin is persulfidated by physiological RSS and this process enhances its catalytic activity through protecting it from inactivation by S-nitrosylation. In the brains of patients with PD, a 60% decrease in parkin persulfidation was found what confirms continuing reduction of parkin activity in PD and suggests that it can be a pathological factor contributing to PD development [154].
Recently, Giovinazzo et al. studying the molecular mechanisms of neuroprotective effects of H2S in AD, reported that the activity of CSE, which can bind wild-type microtubule-associated protein Tau, was diminished in the 3xTg-AD mouse model as well as in AD human brains. In physiological conditions, RSS produced by CSE enhanced catalytic activity of Tau by persulfidation of its kinase, glycogen synthase kinase 3β (GSK3β) thereby preventing the hyperphosphorylation of Tau. The authors revealed that sulfhydration of GSK3β was diminished in AD by nearly 50%, while administration of RSS donors to 3xTg-AD mice, which prevent hyperphosphorylation of Tau, ameliorated motor and cognitive deficits in these mice [155].
It seems that persulfidation of specific proteins has a great significance not only in chronic neurodegenerative diseases but in many other pathologies. In the cardiovascular system, vasorelaxation is the most important activity of RSS. Among proteins involved in this process, ATP-sensitive potassium channels (KATP) and TRPV4 channels that can activate Ca2+ ion-activated K+ (Kca) channels have been shown to be regulated by persulfidation [156]. The decreased level of H2S found in patients with hypertension [117,157] suggests that in this pathology the level of protein persulfidation is also decreased, especially of the aforementioned channels. The administration of RSS donors has been shown to lead to persulfidation of specific cysteine residues in the KATP channel, inducing hyperpolarization and vasorelaxation [158].
Kelch-like-ECH-associated protein 1 (Keap 1) is a repressor protein that binds to nuclear factor erythroid 2-related factor 2 (Nrf2) and promotes its degradation by the ubiquitin proteasome pathway. The Keap1–Nrf2 pathway is an important regulator of protective response to electrophilic and oxidative stress [159]. Keap1 is a Cys-rich protein, and its persulfidation has been well documented. It has been shown that persulfidation of Keap 1 by RSS suppressed diabetes-accelerated atherosclerosis [123] and showed protective effect against cellular aging [160]. Recently, it has also been reported that persulfidation of Keap 1 in the livers of patients with fatty liver under diabetic conditions was decreased [161].
Disturbances in protein persulfidation have been recently proposed as a mechanism that occurs in obesity. In this pathology, hypertrophy of the adipose tissue leads to oxidative stress that favors oxidation of Cys-SH residues to sulfenic acids. The latter can react with H2S forming protein persulfides. However, in the obesity a decreased synthesis of H2S means an oxidizing environment in which protein sulfenic acids are oxidized to sulfonic acid leading to protein inactivation [162]. This hypothesis seems to be confirmed by a recent study that determined protein persulfidation in various tissues of mice under dietary restriction (DR) [163]. It is well documented that DR increases lifespan of organisms [164] but Bithi et al. found that DR in mid-life could boost H2S production and increase protein persulfidation in many tissues. They observed enriched protein persulfidation in the liver, kidney, muscle and brain, while in the heart a decrease in protein persulfidation was observed. DR did not affect protein persulfidation in plasma of mice used in this experiment [163]. Moreover, the same authors performed a similar study introducing DR in late-life which confirmed that fasting even in late-life increased protein persulfidation in the liver, kidney, skeletal muscle and brain, although not always to the same extent as mid-life DR intervention. In this study, a decrease in protein persulfidation in the heart was also observed [165]. Interestingly, these effects were sex-dependent—they were observed only in male individuals. It suggests that the protein persulfidation is a complex process that is influenced by hormones and more studies aimed to elucidate this phenomenon is required.
Reactive sulfur donors
As discussed above, the disturbances in homeostasis of RSS, including H2S, persulfides and polysulfides are involved in aging and many different pathologies. For this reason, effective and safe precursors are being developed. It is impossible here to describe all of studied compounds to be used as H2S releasing agents or per- and polysulfide prodrugs. It is clear that inorganic sulfides and polysulfide salts, like Na2S and Na2Sn used commonly as experimental H2S and polysulfides donors are not suitable for use in in vivo studies due to fast and uncontrollable release of RSS, what is dangerous especially in the case of free H2S.
H2S donors
Among all designed donors of various sulfur species, studies aimed to develop effective donors of H2S were the earliest and are the most advanced. They led to development of commonly known and often used: sodium polythionate SG-1002, which in fact is a mixture of various sulfur compounds including elemental sulfur and acts as an RSS donor rather than as a specific H2S donor, Lawesson’s reagent (2,4-bis(4-methoxyphenyl)1,3,2,4-dithiaphosphetane-2,4-disulfide) [166,167], and a new generation, slow-releasing H2S donor morpholin-4-ium-4 methoxyphenyl(morpholino)phosphinodithioate known as GYY4137 [167,168] (Figure 12). Then, many other reagents have been proposed that can release H2S using different mechanisms, of which the following should be mentioned here: H2S donors controlled by pH, hydrolysis-based donors, redox-activated H2S donors, photosensitive or photoactivated H2S donors and H2S donors activated by esterase. Moreover, donors based on labile S-N bonds have been developed, and it was revealed that thiols (e.g. Cys) are essential to trigger H2S release from these molecules [169]. The next important group of H2S prodrugs, some of which entered the clinical phase, includes H2S-releasing modifications of many clinically used drugs with the well-known ATB-346 which is a H2S-releasing derivative of naproxen [170,171] (Figure 12). An important representative of H2S precursors carbonyl sulfide (COS) is hydrolyzed in mammalian cells to H2S by carbonic anhydrase [172]. COS production in mammalian cells has yet to be confirmed; however, COS application as H2S donor for biological applications can be accepted due to enzymatic conversion of COS to H2S [173]. The list of compounds designed and tested as potential H2S donors is much longer and it was the subject of several review papers [174–178].
Figure 12. Structures of popular H2S donors.
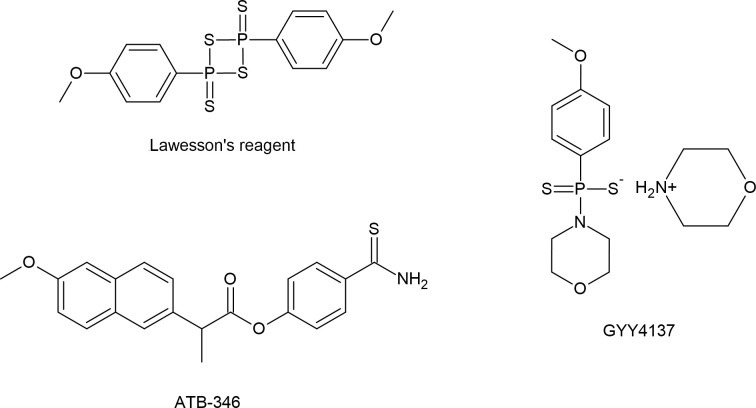
Recently, it has been suggested that the mechanism of action of the well-known drug N-acetylcysteine (NAC) is connected with its conversion to H2S and other RSS containg sulfane sulfur [179]. NAC easily penetrates into the cells where it is deacetylated to Cys, the main substrate for H2S synthesis. Ezerina et al. were the first who noticed that NAC triggered the production of intracellular H2S and other sulfane sulfur-containing compounds [180]. NAC is clinically used in various pathologies including neurological disorders, liver failures, spermatogenesis disorders and the spectrum of its pharmacological activity is constantly expanding [181–183] but the mechanism of its action is not fully explained.
A few clinical studies reported the effectiveness of NAC as an adjunctive therapy in COVID-19. Some patients with moderate or severe COVID-19 pneumonia received orally NAC apart from standard care. The obtained results revealed that NAC slowed down the progression to severe respiratory failure, reduced inflammation and caused a clinical improvement when compared with the control patients who received only standard treatment without NAC. A lower mortality was also observed in the group of patients treated with NAC [184,185]. Unfortunately, none of these reports investigated the plasma H2S level in patients with COVID-19 before and during therapy of NAC, so it could be only hypothesized that beneficial action of NAC in patients with severe COVID-19, is largely due to the increase in H2S concentration.
최근에 잘 알려진 약물인 N-아세틸시스테인(NAC)의 작용 기전이 H2S 및 황화황을 함유한 기타 RSS로의 전환과 관련이 있다는 주장이 제기되었습니다 [179]. NAC는 세포 내로 쉽게 침투하여 H2S 합성의 주요 기질인 Cys로 탈아세틸화됩니다. Ezerina 등은 NAC가 세포 내 H2S 및 기타 황 함유 화합물의 생성을 촉발한다는 사실을 최초로 발견했습니다 [180]. NAC는 신경 장애, 간부전, 정자 형성 장애를 포함한 다양한 병리학에서 임상적으로 사용되고 있으며, 그 약리학적 활동의 스펙트럼은 지속적으로 확장되고 있습니다 [181-183] 그러나 그 작용 메커니즘은 완전히 밝혀지지 않았습니다.
몇몇 임상 연구에서 COVID-19의 보조 요법으로서의 NAC의 효과를 보고했습니다. 중등도 또는 중증 COVID-19 폐렴 환자 중 일부는 표준 치료와 별개로 NAC를 경구 투여받았습니다. 그 결과, NAC는 표준 치료만 받은 대조군과 비교했을 때, 중증 호흡 부전으로의 진행을 늦추고, 염증을 감소시키며, 임상적 개선을 가져오는 것으로 나타났습니다. NAC로 치료받은 환자 그룹에서도 사망률이 낮았습니다 [184,185]. 안타깝게도, 이 보고서들 중 어느 것도 COVID-19 환자의 혈장 H2S 수치를 NAC 치료 전과 치료 중 조사하지 않았기 때문에, COVID-19 중증 환자의 경우 NAC가 유익한 작용을 하는 이유는 주로 H2S 농도의 증가 때문이라고 추측할 수 있을 뿐입니다.
Persulfide donors
One of the most important mechanism of RSS action in biological systems is related to persulfidation of proteins. This reaction can be triggered by H2S if target -SH groups in proteins are reversibly oxidized to sulfenic acid or S-nitrosothiols. When the protein -SH groups are in reduced state, only persulfides or polysulfides can sulfhydrate target proteins. It is assumed that persulfides and polysulfides are regarded as more efficient persulfidating agents than H2S. As mentioned earlier, persulfides are also more active and safer sulfur species than H2S what makes them good candidates for sulfur redox signaling. However, due to their low stability it is not easy to use them directly for biological application. Therefore, some efforts have been made in making persulfide donors suitable for use in biological systems.
Currently, several small molecules have been described as persulfide donors, which release persulfides in response to a different stimuli. A series of esterase-sensitive persulfide prodrugs was developed including a GSSH donor [186]. Powell et al. developed and reported the ROS-responsive persulfide donor termed Bpin-disulfide prodrug (BDP-NAC) which is a derivative of NAC (Figure 13). This compounds released NAC-SSH in response to H2O2 [175]. The same research group developed then another derivative of NAC, termed superoxide-responsive, persulfide donor, SOPD-NAC, which reacts with superoxide radical anion (O2●−) releasing NAC-SSH (Figure 13) [187]. Continuing their research on effective persulfide prodrugs, these authors next synthesized a small molecule which acts as an NAC-SSH donor in response to esterase and termed it ester disulfide prodrug, EDP-NAC. Moreover, they designed and synthesized its polymeric form termed poly(EDP-NAC) (Figure 13). The performed experiments confirmed that polymeric form of this prodrug released persulfides much more slowly and protected cardiomiocytes more effectively than EDP-NAC [188]. More examples of persulfide prodrugs including photosensitive agents, molecules activated by ROS and other structures were described in the literature [68,178,186,189–193].
과황화물 기증자
생물학적 시스템에서 RSS 작용의 가장 중요한 메커니즘 중 하나는 단백질의 과황화 작용과 관련이 있습니다. 이 반응은 단백질의 -SH 그룹이 설펜산 또는 S-니트로소티올로 가역적으로 산화될 때 H2S에 의해 촉발될 수 있습니다. 단백질의 -SH 그룹이 환원 상태에 있을 때, 과황화물 또는 폴리황화물만이 표적 단백질을 황화할 수 있습니다. 과황화물과 폴리황화물이 H2S보다 더 효율적인 과황화제로 간주된다고 가정합니다. 앞서 언급한 바와 같이, 과황화물은 H2S보다 더 활동적이고 안전한 황 종류이기 때문에 황 산화 환원 신호 전달에 적합한 후보입니다. 그러나, 과황화물의 안정성이 낮기 때문에 생물학적 응용에 직접 사용하는 것이 쉽지 않습니다. 따라서, 생물학적 시스템에 사용하기 적합한 과황화물 공여체를 만들기 위한 노력이 이루어졌습니다.
현재, 여러 가지 작은 분자가 다른 자극에 반응하여 퍼설파이드를 방출하는 퍼설파이드 공여체로 설명되었습니다. GSSH 공여체를 포함하여 일련의 에스테라아제 민감성 퍼설파이드 전구약물이 개발되었습니다 [186]. Powell 등은 NAC의 유도체인 Bpin-disulfide 전구약물(BDP-NAC)이라는 ROS 반응성 퍼설파이드 공여체를 개발하고 보고했습니다(그림 13). 이 화합물은 H2O2에 반응하여 NAC-SSH를 방출합니다 [175]. 같은 연구 그룹이 슈퍼옥사이드 반응성, 퍼설파이드 기증자, SOPD-NAC라고 불리는 또 다른 NAC 유도체를 개발했는데, 이 물질은 슈퍼옥사이드 라디칼 음이온(O2●−)과 반응하여 NAC-SSH를 방출합니다 (그림 13) [187]. 효과적인 퍼설파이드 전구약물에 대한 연구를 계속하면서, 이 저자들은 에스테라아제에 반응하여 NAC-SSH 공여체 역할을 하는 작은 분자를 합성하고 이를 에스터 디설파이드 전구약물(EDP-NAC)이라고 명명했습니다. 또한, 그들은 폴리(EDP-NAC)라는 고분자 형태를 설계하고 합성했습니다(그림 13). 수행된 실험을 통해 이 전구약물의 중합체 형태가 EDP-NAC보다 훨씬 더 느리게 퍼설파이드를 방출하고 심장근세포를 더 효과적으로 보호한다는 사실이 확인되었습니다 [188]. 광과민성 물질, ROS에 의해 활성화되는 분자, 기타 구조를 포함하는 퍼설파이드 전구약물에 대한 더 많은 예가 문헌에 기술되어 있습니다 [68,178,186,189-193].
Figure 13. Examples of various persulfide prodrugs.
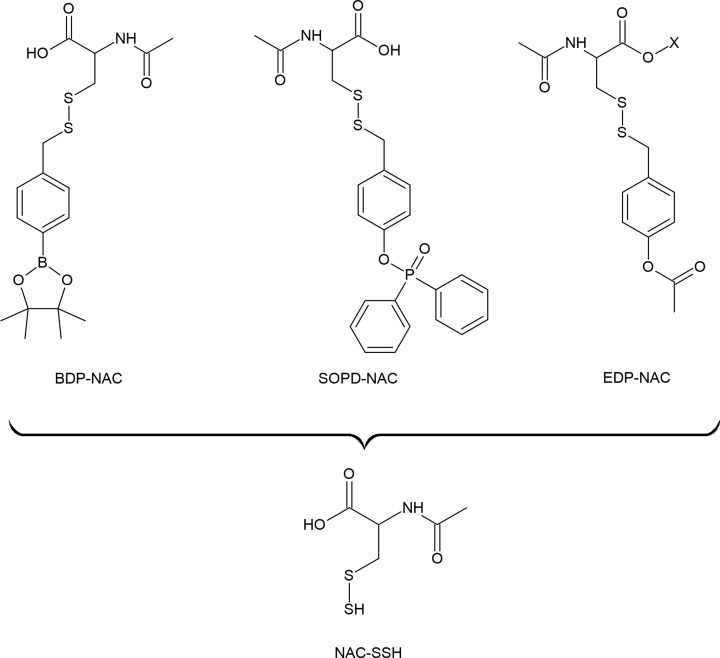
Polysulfide donors
In relation to the above-described compounds, NAC-derivatives have been studied as persulfide prodrugs, and NAC polysulfides were tested as polysulfide prodrugs. It was demonstrated that NAC polysulfides (Figure 14) efficiently increased intracellular levels of RSS, however, a significant increase in the content of persulfides, such as CysSSH and GSSH was also observed [194]. It means that NAC polysulfides are good, stable and effective RSS precursors but they are not selective for polysulfides. It seems to be a general rule for per- and polysulfide prodrugs due to a very close relationship between these two kinds of RSS. Moreover, the equilibrium between H2S, persulfides and polysulfides is dynamic and depends on various factors including pH and redox status of the cells. It was also confirmed by a study utilizing a synthetic slow-releasing RSS donor, which is chemically derived from penicillamine [191]. This compound generates a persulfide and a mixture of polysulfides as end-products. Moreover, it can also release H2S, but only in the presence of thiols, such as Cys-SH or GSH [195].
폴리설파이드 공여체
위에서 설명한 화합물과 관련하여 NAC-파생물이 퍼설파이드 전구체로 연구되었고, NAC 폴리설파이드가 폴리설파이드 전구체로 시험되었습니다. NAC 폴리설파이드(그림 14)가 세포 내 RSS 수준을 효율적으로 증가시키는 것으로 입증되었지만, CysSSH 및 GSSH와 같은 퍼설파이드의 함량도 상당히 증가하는 것으로 관찰되었습니다[194]. 이는 NAC 폴리설파이드가 좋은, 안정적이고 효과적인 RSS 전구물질이지만, 폴리설파이드에 선택적이지 않다는 것을 의미합니다. 이 두 종류의 RSS 사이의 매우 밀접한 관계 때문에, 퍼설파이드와 폴리설파이드 전구물질에 대한 일반적인 규칙인 것 같습니다. 게다가, H2S, 퍼설파이드, 폴리설파이드 사이의 평형은 동적이며, 세포의 pH와 산화 환원 상태를 포함한 다양한 요인에 따라 달라집니다. 또한 페니실라민[191]에서 화학적으로 유도된 합성 서방성 RSS 기증자를 이용한 연구에서도 확인되었습니다. 이 화합물은 최종 생성물로서 퍼설파이드와 폴리설파이드의 혼합물을 생성합니다. 또한, H2S를 방출할 수도 있지만, Cys-SH 또는 GSH와 같은 티올이 존재하는 경우에만 가능합니다[195].
Figue 14. Examples of polysulfide donors.
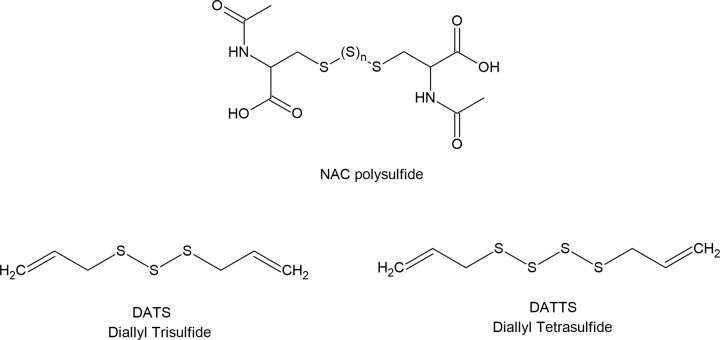
Among natural rich sources of polysulfides, garlic deserves special attention, as it contains organopolysulfides, including diallyl trisulfide (DATS) and diallyl tetrasulfide (DATTS) which can be used as RSS donors (Figure 14). These compounds after polysulfide bond cleavage form persulfides (RSSnH), which can further react with thiols leading to H2S production [196]. This reaction can be repeated and other polysulfides are produced as byproducts. The effectiveness of DATS as an RSS precursor was documented in vivo in animal models. DATS administered intraperitoneally to mice elevated the total sulfane sulfur pool (persulfides and polysulfides) and increased the activity of enzymes involved in their metabolism in the liver and kidney [197,198]. Interestingly, Tocmo et al. studied the effect of cooking on garlic's organopolysulfides and H2S-releasing activity. They found that the amounts of allyl polysulfides increased in crushed garlic boiled for 6–10 min; however, longer thermal treatment decreased their concentrations [199]. Garlic-derived allyl polysulfides as well as isothiocyanates derived from Brassicae have been recognized as polysulfide or H2S donors of great pharmacological potential. Recently, growing attention has been focused on these dietary organosulfur compounds in relation to the possibilities of use in the treatment of diseases with reduced RSS synthesis [200,201]. Moreover, garlic-derived polysulfides are proposed as a potential therapeutic food to fight COVID-19 [202–204].
H2Sn donors
Like in the case of H2S, persulfides and polysulfides, slow and controllable donors of inorganic polysulfides (H2Sn) are looked for but this area is not well explored. Yu et al. developed esterase- and phosphatase-sensitive H2S2 prodrugs with tenable release rates, termed BW-HP-302 and BW-HP-303, respectively (Figure 15). The utility of these compounds in RSS donating was confirmed by S-persulfidation of glyceraldehyde 3-phosphate dehydrogenase (GAPDH) [205]. H2Sn donors, like BW-HP compounds, are based on the template of highly reactive acyl disulfide, they can form polysulfides and release H2S under the influence of thiols [206]. Acyl disulfides can also react with nucleophiles, like amines, which suggests that these H2Sn donors in biological systems may react with naturally existing molecules, like amino acids [102]. Recently, Xu et al. demonstrated that diacyl disulfides could produce H2S2 in reaction with various nucleophiles including amines in both aqueous and organic environment [206].
자연적으로 풍부한 폴리설파이드의 원천 중에서도 마늘은 특별한 관심을 기울일 필요가 있습니다. 마늘에는 RSS 공여체로 사용할 수 있는 디알릴 트리스루파이드(DATS)와 디알릴 테트라설파이드(DATTS)를 포함한 유기폴리설파이드가 포함되어 있기 때문입니다(그림 14). 이러한 화합물은 폴리설파이드 결합이 절단된 후 퍼설파이드(RSSnH)를 형성하며, 이는 티올과 더 반응하여 H2S를 생성할 수 있습니다 [196]. 이 반응은 반복될 수 있으며, 다른 폴리설파이드가 부산물로 생성됩니다. RSS 전구체로서의 DATS의 효과는 동물 모델에서 생체 내에서 입증되었습니다. 마우스에게 복강 내 투여한 DATS는 총 황화황 풀(과황화물과 폴리황화물)을 증가시키고 간과 신장에서 그 대사에 관여하는 효소의 활성을 증가시켰다[197,198]. 흥미롭게도, Tocmo 등은 마늘의 유기폴리황화물과 H2S 방출 활동에 대한 조리 효과를 연구했습니다. 그들은 으깬 마늘을 6-10분 동안 삶으면 알릴 폴리설파이드의 양이 증가한다는 것을 발견했습니다. 그러나 더 오래 열처리하면 농도가 감소했습니다 [199]. 마늘에서 추출한 알릴 폴리설파이드와 십자화과 식물에서 추출한 이소티오시아네이트는 약리학적으로 큰 잠재력을 지닌 폴리설파이드 또는 H2S 공여체로 인식되고 있습니다. 최근에는 RSS 합성 감소로 인한 질병 치료에 사용할 수 있는 가능성과 관련하여 이러한 유기황 화합물에 대한 관심이 높아지고 있습니다 [200,201]. 또한, 마늘에서 추출한 폴리설파이드가 코로나19에 대항할 수 있는 잠재적인 치료 식품으로 제안되고 있습니다 [202-204].
H2Sn 기증자
H2S, 과황화수소, 폴리황화수소와 마찬가지로, 무기 폴리황화수소(H2Sn)의 느리고 제어 가능한 기증자가 필요하지만, 이 분야는 아직 잘 연구되지 않았습니다. Yu 등은 각각 BW-HP-302와 BW-HP-303이라고 불리는, 유지 가능한 방출 속도를 가진 에스테라제 및 포스파타제 민감성 H2S2 전구체를 개발했습니다(그림 15). RSS 기증에서 이러한 화합물의 유용성은 글리세르알데히드 3인산 탈수소효소(GAPDH)의 S-퍼설파이드화에 의해 확인되었다[205]. BW-HP 화합물과 같은 H2Sn 공여체는 반응성이 높은 아실 디설파이드의 템플릿을 기반으로 하며, 폴리설파이드를 형성하고 티올의 영향으로 H2S를 방출할 수 있다[206]. 아실 디설파이드도 아민과 같은 친핵체와 반응할 수 있습니다. 이는 생물학적 시스템에서 이러한 H2Sn 공여체가 아미노산과 같은 자연적으로 존재하는 분자와 반응할 수 있음을 시사합니다 [102]. 최근에 Xu 등은 디아실 디설파이드가 수성 및 유기 환경에서 아민을 포함한 다양한 친핵체와 반응하여 H2S2를 생성할 수 있음을 입증했습니다 [206].
Figure 15. Structures of the two developed H2Sn donors.
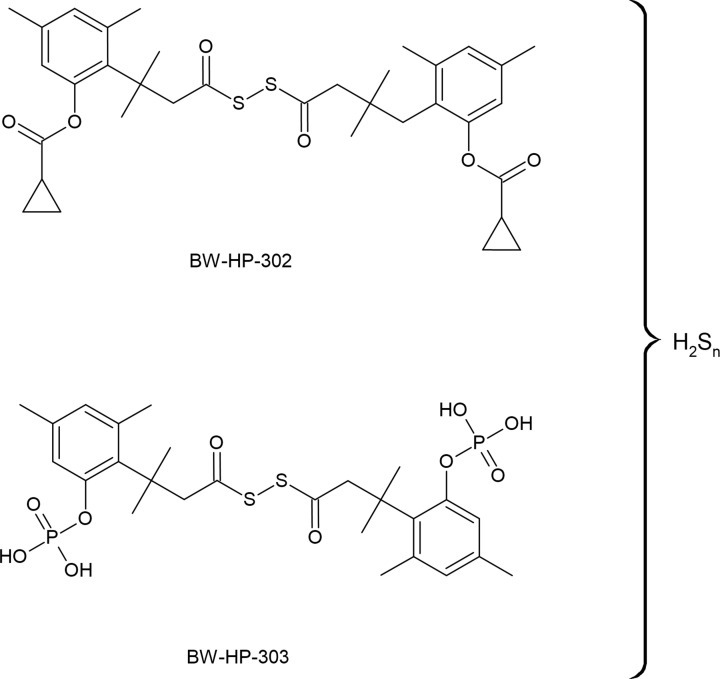
The more is known about the biological role of RSS and disorders of homeostasis in many pathological conditions, the greater the need to develop slow- and controlled-release RSS donors which could be used in experimental studies and, in the longer term, could be applicable in clinical trials to treat some pathologies. The list of potential developed H2S donors is impressive, but only a few of them in experimental studies showed features predisposing them to clinical applications. Among the recently developed precursors, NAC derivatives seem to be the most promising molecules. As NAC is a drug approved for clinical use, it is quite safe and well tolerated. Therefore, stable NAC derivatives that are easily transformed in cells into NAC persulfides, providing RSS for protein persulfidation and a powerful antioxidant and reductive force, open up possibilities for their use to treat diseases. The second promising source of RSS derives from polysulfides and isothiocyanates naturally occurring in garlic and Brassicae. A diet rich in these foods may be effective in protecting against diseases associated with reduced RSS synthesis, such as cardiovascular diseases, cancer or aging.
RSS의 생물학적 역할과 여러 병리학적 조건에서 항상성 장애에 대해 더 많이 알려질수록, 실험 연구에 사용될 수 있고, 장기적으로는 일부 병리를 치료하기 위한 임상 시험에 적용될 수 있는 서방성 및 제어 방출 RSS 기증자를 개발해야 할 필요성이 커집니다. 개발 가능한 H2S 기증자 목록은 인상적이지만, 실험 연구에서 임상 적용에 적합한 특성을 보이는 기증자는 소수에 불과합니다. 최근에 개발된 전구체들 중에서 NAC 유도체가 가장 유망한 분자인 것 같습니다. NAC는 임상용으로 승인된 약물이므로, 안전성이 매우 높고 내약성도 좋습니다. 따라서, 세포에서 쉽게 NAC 퍼설파이드로 전환되어 단백질 퍼설파이드화를 위한 RSS를 제공하고 강력한 항산화 및 환원력을 제공하는 안정적인 NAC 유도체는 질병 치료에 사용할 수 있는 가능성을 열어줍니다. RSS의 두 번째 유망한 공급원은 마늘과 십자화과에 자연적으로 발생하는 폴리설파이드와 이소티오시아네이트에서 비롯됩니다. 이러한 식품이 풍부한 식단은 심혈관 질환, 암, 노화와 같은 RSS 합성 감소와 관련된 질병을 예방하는 데 효과적일 수 있습니다.
Reactive sulfur inhibitors
For understanding the biological function of H2S and other forms of RSS, pharmacological inhibitors of the main enzymes involved in synthesis of H2S and other RSS may play a helpful role. They include three sulfurases: two PLP-dependent enzymes: CSE and CBS as well as one PLP-independent MST. Moreover, some reports mentioned above and cited in Table 2 have documented that activity of these enzymes is up-regulated in some pathologies leading to overproduction of H2S and other RSS. Therefore, compounds able to selectively inhibit individual enzyme will be needed.
In the case of CSE, which is a major source of H2S in peripheral tissues and cardiovascular system, the most popular compounds used to inhibit this enzyme include DL-propargyl-glycine (PAG), β-cyano-L-alanine (BCA) and L-aminoethoxyvinylglycine (AVG) [174]. These compounds preferentially inhibit CSE, however, they have been shown to inhibit also other PLP-dependent enzymes [207]. Of these compounds, PAG is the most commonly used drug of choice to pharmacologically inhibit CSE. In animal models, this compound elevated blood pressure [116] and increased: cardiac damage [208], ischemia/reperfusion injury [209] and mortality in sepsis [210]. Interestingly, Peng et al. suggested that inhibition of CSE by PAG might be a useful therapeutic intervention to prevent carotid body- driven sleep apnea [211]. Due to poor selectivity and poor efficiency (high concentration and limited ability to permeate the cell membrane) of PAG, new potent and selective inhibitors are searched for. In this aspect, Hu et al. studied recently several potential compounds and found that compound named NSC4056, which is a derivative of salicylic acid, was the most potent, active and selective CSE inhibitor [212]. This result offers new opportunities for development of new pharmacologically active remedies to selectively inhibit CSE; however, more studies in vivo using NSC4056 are needed.
CBS is predominantly present in the central nervous system but it has also been found in other tissues including the liver, pancreas and kidney. Despite many studies aimed to discover new, selective CBS inhibitors, to date aminooxyacetic acid (AOAA) CBS remains the most commonly used inhibitor of CBS [17]. This compound has been shown to normalize H2S overproduction mediated by CBS, to restore normal mitochondrial function in Down syndrome [213] and to inhibit proliferation, angiogenesis and cell bioenergetics in cancer cells [214,215]. Moreover it has been also found that AOAA reduced vascular relaxation induced by acetylcholine what confirms implication of H2S in the regulation of vascular tone [216]. As mentioned above, AOAA is regarded as ‘classical CBS inhibitor’; however, it has been well documented that AOAA reacts with PLP and in this way it can also inhibit other PLP-dependent enzymes including CSE [217]. Moreover, study by Asimakopoulou et al. using recombinant CBS and CSE revealed that AOAA exhibited more potency toward CSE when compared with CBS (IC 1.1 vs. 8.5 µM, respectively) [207].
Thus, AOAA and PAG, commonly used inhibitors of CBS and CSE, despite of their usefulness have the limitations associated mainly with the lack of the selectivity and low cell uptake.
For many years, pharmacological inhibitors have not been identified. L-aspartate has been documented to inhibit CAT [217], so the use L-aspartate can suppress H2S production by limitation of 3-MP, the substrate for MST. Recently, the role of MST/H2S has been studied in some aspects using the novel pharmacological, potent and selective MST inhibitor (2-[(4-hydroxy-6-methylpyrimidin-2-yl)sulfanyl]-1-(naphthalen-1-yl)ethan-1-one (HMPSNE). This compound in a concentration-dependent way suppressed H2S production and exerted antiproliferative effects in murine colon cancer [218]. Moreover, HMPSNE produced a bell-shaped effect on several cellular bioenergetic parameters related to oxidative phosphorylation, while other bioenergetic parameters were either unaffected or inhibited at the highest concentration of the inhibitor [218]. On the other hand, Casili et al. using HMPSNE revealed that inhibition of MST significantly enhanced lipid accumulation into the maturing adipocytes [219]. All the studied bioenergetic parameters, including ATP production, were significantly lower in mice adipocyte tissue pretreated with HMPSNE compared with untreated tissue [219]. These results are interesting and it seems that HMPSNE is a promising candidate for selective inhibition of MST; however, more studies including in vivo experiments are needed to confirm its effectiveness and safety.
Overproduction of RSS is found in a smaller number of diseases when compared with the deficiency of RSS synthesis. However, it seems that finding safe and selective inhibitors of enzymes participating in the RSS synthesis, in the future would enable their use in diseases, the course of which is associated with overproduction of RSS, such as Down syndrome or ALS.
반응성 황 억제제
H2S와 다른 형태의 RSS의 생물학적 기능을 이해하기 위해, H2S와 다른 RSS의 합성에 관여하는 주요 효소의 약리학적 억제제가 도움이 될 수 있습니다. 여기에는 세 가지 황화효소, 즉 PLP 의존성 효소인 CSE와 CBS, 그리고 PLP 비의존성 MST가 포함됩니다. 또한, 표 2에 인용된 일부 보고서에서는 이러한 효소의 활동이 H2S 및 기타 RSS의 과잉 생산을 유발하는 일부 병리학에서 상향 조절된다는 사실이 문서화되어 있습니다. 따라서 개별 효소를 선택적으로 억제할 수 있는 화합물이 필요할 것입니다.
말초 조직과 심혈관계에서 H2S의 주요 공급원인 CSE의 경우, 이 효소를 억제하는 데 가장 많이 사용되는 화합물에는 DL-프로파르길-글리신(PAG), β-시아노-L-알라닌(BCA), L-아미노에톡시비닐글리신(AVG) 등이 있습니다[174]. 이 화합물들은 우선적으로 CSE를 억제하지만, 다른 PLP 의존성 효소도 억제하는 것으로 나타났습니다 [207]. 이 화합물들 중에서, PAG는 CSE를 약리학적으로 억제하는 데 가장 일반적으로 사용되는 약물입니다. 동물 모델에서, 이 화합물은 혈압을 상승시켰고 [116], 심장 손상 [208], 허혈/재관류 손상 [209], 패혈증 사망률 [210]을 증가시켰습니다. 흥미롭게도, Peng 등은 PAG에 의한 CSE 억제가 경동맥 체성 수면 무호흡증을 예방하는 데 유용한 치료적 개입이 될 수 있다고 제안했습니다 [211]. PAG의 낮은 선택성과 낮은 효율성(높은 농도와 세포막 투과 능력의 제한) 때문에, 새롭고 강력하고 선택적인 억제제가 연구되고 있습니다. 이와 관련하여, Hu 등은 최근 몇 가지 잠재적 화합물을 연구한 결과, 살리실산의 유도체인 NSC4056이라는 화합물이 가장 강력하고, 활성적이고, 선택적인 CSE 억제제라는 것을 발견했습니다 [212]. 이 결과는 CSE를 선택적으로 억제하는 새로운 약리학적 활성 치료제 개발에 새로운 기회를 제공합니다. 그러나 NSC4056을 사용한 생체 내 연구가 더 필요합니다.
CBS는 주로 중추신경계에 존재하지만 간, 췌장, 신장 등 다른 조직에서도 발견되었습니다. 새로운 선택적 CBS 억제제를 발견하기 위한 많은 연구에도 불구하고, 현재까지 아미노옥시아세트산(AOAA)이 가장 일반적으로 사용되는 CBS 억제제입니다 [17]. 이 화합물은 CBS에 의해 매개되는 H2S 과잉 생산을 정상화하고, 다운증후군에서 정상적인 미토콘드리아 기능을 회복하며 [213], 암세포의 증식, 혈관형성 및 세포 생체 에너지를 억제하는 것으로 나타났습니다 [214,215]. 또한 AOAA가 아세틸콜린에 의해 유발되는 혈관 이완을 감소시킨다는 사실도 밝혀졌는데, 이는 H2S가 혈관 긴장 조절에 관여한다는 것을 확인시켜 줍니다 [216]. 위에서 언급한 바와 같이, AOAA는 '고전적인 CBS 억제제'로 간주됩니다; 그러나, AOAA가 PLP와 반응하여 CSE를 포함한 다른 PLP 의존성 효소를 억제할 수 있다는 사실이 잘 알려져 있습니다 [217]. 또한, Asimakopoulou 등의 연구에 따르면 재조합 CBS와 CSE를 사용한 연구에서 AOAA가 CBS에 비해 CSE에 더 많은 효능을 나타냈습니다(각각 IC 1.1 vs. 8.5 µM) [207].
따라서, 일반적으로 CBS와 CSE의 억제제로 사용되는 AOAA와 PAG는 유용성에도 불구하고 선택성의 부족과 낮은 세포 흡수율과 관련된 한계가 있습니다.
수년 동안 약리학적인 억제제는 확인되지 않았습니다. L-아스파르트산은 CAT를 억제하는 것으로 알려져 있습니다 [217], 따라서 L-아스파르트산을 사용하면 MST의 기질인 3-MP의 제한을 통해 H2S 생성을 억제할 수 있습니다. 최근에, MST/H2S의 역할은 새로운 약리학적이고, 강력하고 선택적인 MST 억제제(2-[(4-hydroxy-6-methylpyrimidin-2-yl)sulfanyl]-1-(naphthalen-1-yl)ethan-1-one(HMPSNE))를 사용하여 일부 측면에서 연구되었습니다. 이 화합물은 농도 의존적으로 H2S 생성을 억제하고 쥐의 결장암에 항증식 효과를 나타냈습니다 [218]. 또한, HMPSNE는 산화적 인산화와 관련된 여러 세포 생체 에너지 매개 변수에 종 모양의 효과를 나타냈으며, 억제제의 최고 농도에서 다른 생체 에너지 매개 변수는 영향을 받지 않거나 억제되었습니다 [218]. 반면, Casili 등은 HMPSNE를 사용하여 MST의 억제가 성숙 지방세포로의 지질 축적을 상당히 향상시킨다는 것을 밝혀냈습니다 [219]. ATP 생산을 포함한 모든 연구된 생체 에너지 매개변수는 HMPSNE로 전처리된 마우스 지방세포 조직에서 처리되지 않은 조직에 비해 현저히 낮았습니다 [219]. 이러한 결과는 흥미롭고, HMPSNE가 MST의 선택적 억제에 유망한 후보인 것으로 보입니다. 그러나 그 효과와 안전성을 확인하기 위해서는 생체 내 실험을 포함한 더 많은 연구가 필요합니다.
RSS 과잉생산은 RSS 합성 결핍과 비교했을 때, 더 적은 수의 질병에서 발견됩니다. 그러나, 향후에 RSS 합성에 관여하는 효소의 안전하고 선택적인 억제제를 찾는 것이 다운증후군이나 ALS와 같이 RSS 과잉생산과 관련된 질병에 사용될 수 있을 것으로 보입니다.
Conclusions
Mounting evidence suggest that RSS play an important role in redox regulation and are potent physiological mediators. Initially, this role was mainly assigned to H2S, but now it is known that these are persulfides and polysulfides, closely related to H2S, that are responsible for many biological effects, including protein persulfidation, and they possess more potent antioxidant and reductive properties. Since all RSS coexist in biological systems and per- and polysulfides are rather unstable molecules, research on the role of particular forms of RSS is challenging. The high reactivity of these compounds makes this task even more difficult; however, constantly developing methods and tools for specific detection of individual RSS forms offer hope for a more reliable analysis and a better understanding of the physiological role of RSS. Over the past 20 years, tremendous progress has been made in developing methods and clarifying the role of RSS in many pathologies. Moreover, many stable and efficient-release donors of RSS have been developed, however, most of them have been tested under in vitro conditions. Among the recently developed RSS precursors, NAC derivatives seem to be the most promising molecules for clinical use since NAC is quite safe, cheap, well-tolerated and most importantly it has been approved for clinical use. In several studies, NAC was also used to treat patients with severe COVID-19 with a success. It seems reasonable that in the aspect of SARS-CoV-2, the beneficial effect of NAC is at least partly related to RSS generation and modulation of their physiological level. This, together with documented anti-inflammatory and antiviral properties of NAC, provides the basis for considering it or its derivatives as a beneficial adjuvant therapy in the course of COVID-19 and other pathologies.
결론
증거가 쌓이면서, 산화 환원 조절에 중요한 역할을 하고 강력한 생리적 매개체 역할을 한다는 사실이 밝혀졌습니다. 처음에는 주로 H2S가 이 역할을 담당하는 것으로 여겨졌지만, 이제는 단백질 과황화 작용을 포함한 많은 생물학적 효과를 담당하는 물질이 H2S와 밀접하게 관련된 과황화물과 폴리황화물이라는 사실이 밝혀졌습니다. 모든 RSS가 생물학적 시스템에 공존하고 있고, 퍼설파이드와 폴리설파이드가 불안정한 분자이기 때문에 특정 형태의 RSS의 역할에 대한 연구는 매우 어렵습니다. 이러한 화합물의 높은 반응성은 이 작업을 더욱 어렵게 만듭니다. 그러나 개별 RSS 형태의 특정 검출을 위한 방법과 도구를 지속적으로 개발하고 있기 때문에, 보다 신뢰할 수 있는 분석과 RSS의 생리적 역할에 대한 더 나은 이해를 기대할 수 있습니다. 지난 20년 동안, 많은 병리학 분야에서 RSS의 역할을 명확히 하고 그 방법을 개발하는 데 엄청난 진전이 있었습니다. 또한, 안정적이고 효율적인 RSS 공급자가 많이 개발되었지만, 대부분 체외 조건에서 테스트되었습니다. 최근 개발된 RSS 전구체 중 NAC 유도체는 임상 사용에 가장 유망한 분자로 보입니다. NAC는 안전하고 저렴하며 내약성이 좋고, 무엇보다도 임상 사용 승인을 받았기 때문입니다. 여러 연구에서 NAC가 중증 COVID-19 환자 치료에 성공적으로 사용된 바 있습니다. SARS-CoV-2의 측면에서 볼 때, NAC의 유익한 효과는 적어도 부분적으로는 RSS 생성 및 생리적 수준의 조절과 관련이 있는 것으로 보입니다. 이것과 함께 NAC의 항염증 및 항바이러스 특성이 입증된 것을 고려할 때, NAC 또는 그 유도체를 COVID-19 및 기타 병리의 과정에서 유익한 보조 요법으로 고려할 수 있는 근거가 됩니다.
AbbreviationsAOAA
aminooxyacetic acid
AVG
L-aminoethoxyvinylglycine
BCA
β-cyano-L-alanine
CARS
cysteinyl-tRNA synthetase
CBS
cystathionine β-synthase
CSE
cystathionine γ-lyase
HMPSNE
2-[(4-hydroxy-6-methylpyrimidin-2-yl)sulfanyl]-1-(naphthalen-1-yl)ethan-1-one
MST
3-mercaptopyruvate sulfurtransferase
NAC
N-acetylocysteine
PAG
DL-propargyl-glycine
PLP
pyridoxal phosphate
RSS
reactive sulfur species
Competing Interests