글루타민은 암세포의 밥...
하지만 인체에서 스스로 합성하는 비필수 아미노산이므로
음식으로 차단하는 것이 아니다.
그럼 어떻게?

Targeting glutamine metabolism as a therapeutic strategy for cancer
- Review Article
- Open access
- Published: 03 April 2023
Targeting glutamine metabolism as a therapeutic strategy for cancer
Experimental & Molecular Medicine volume 55, pages706–715 (2023)Cite this article
Abstract
Proliferating cancer cells rely largely on glutamine for survival and proliferation. Glutamine serves as a carbon source for the synthesis of lipids and metabolites via the TCA cycle, as well as a source of nitrogen for amino acid and nucleotide synthesis. To date, many studies have explored the role of glutamine metabolism in cancer, thereby providing a scientific rationale for targeting glutamine metabolism for cancer treatment. In this review, we summarize the mechanism(s) involved at each step of glutamine metabolism, from glutamine transporters to redox homeostasis, and highlight areas that can be exploited for clinical cancer treatment. Furthermore, we discuss the mechanisms underlying cancer cell resistance to agents that target glutamine metabolism, as well as strategies for overcoming these mechanisms. Finally, we discuss the effects of glutamine blockade on the tumor microenvironment and explore strategies to maximize the utility of glutamine blockers as a cancer treatment.
초록
증식 중인 암 세포는
생존과 증식에 글루타민에 크게 의존합니다.
글루타민은
TCA 회로를 통해 지질 및 대사물 합성의 탄소원으로 작용하며,
아미노산 및 뉴클레오티드 합성의 질소원으로 기능합니다.
현재까지 많은 연구에서
글루타민 대사 과정이 암에 미치는 역할을 탐구해 왔으며,
이는 암 치료를 위해 글루타민 대사를 표적으로 삼는 과학적 근거를 제공해 왔습니다.
이 리뷰에서는
글루타민 운반체부터 환원-산화 균형까지 글루타민 대사 과정의 각 단계에서 관여하는 메커니즘을 요약하고,
임상 암 치료에 활용할 수 있는 분야를 강조합니다.
또한,
글루타민 대사 표적 치료제에 대한 암 세포의 저항 메커니즘을 논의하고,
이러한 메커니즘을 극복하기 위한 전략을 제시합니다.
마지막으로,
글루타민 차단이 종양 미세환경에 미치는 영향을 논의하고,
글루타민 차단제를 암 치료제로 활용하기 위한 전략을 탐구합니다.
Similar content being viewed by others
Glutamine reliance in cell metabolism
Article Open access17 September 2020
Targeting pancreatic cancer metabolic dependencies through glutamine antagonism
Article Open access09 October 2023
Article 09 October 2023
Introduction
Metabolic reprogramming, a hallmark of cancer cells, is a process by which cancer cells ensure a sufficient supply of proteins, nucleotides, and lipids to support rapid growth and proliferation1. The importance of cancer cell metabolism and the limitations of conventional cancer therapies (e.g., resistance to chemotherapy or radiotherapy) have prompted the development of strategies aimed at targeting this biological process2. Several drugs that do just that have been introduced and have shown promising results in animal studies; a few have entered clinical trials2. In particular, glutamine metabolism has attracted much attention as a therapeutic target because cancer cells are heavily reliant on this amino acid for growth and proliferation3.
Glutamine is a nonessential/conditionally essential amino acid that plays a pivotal role in clinical illness and stress conditions4. Glutamine in cancer cells plays critical and diverse roles by providing not only a source of nitrogen for amino acid and nucleotide biosynthesis but also a source of carbon to replenish the tricarboxylic acid (TCA) cycle and lipid biosynthesis pathways; thus, cancer cells are “addicted” to glutamine5. Glutamine metabolism and closely linked metabolic networks involving glutamine transporters, glutaminase, aminotransferase, and redox homeostasis are essential for cancer cell survival6. Targeting each step of glutamine metabolism has shown promising results in cancer treatment, prompting the discovery of druggable targets and the development of anticancer drug candidates3. In addition, given that immune checkpoint inhibitors are now widely used to treat cancer, the role of glutamine blockade within the tumor microenvironment (TME) has gained much attention7.
This review summarizes each step of glutamine metabolism in cancer cells and highlights opportunities for clinical intervention. Furthermore, we discuss resistance mechanisms and the role of glutamine blockade in the TME.
소개
대사 재프로그래밍은 암 세포의 특징적인 현상으로, 암 세포가 빠른 성장과 증식을 지원하기 위해 단백질, 핵산, 지질 등의 충분한 공급을 확보하는 과정입니다1. 암 세포의 대사 과정의 중요성과 전통적인 암 치료법(예: 화학요법이나 방사선 요법에 대한 저항성)의 한계는 이 생물학적 과정을 표적으로 삼는 전략의 개발을 촉진했습니다2. 이러한 목표를 달성하기 위해 여러 약물이 개발되어 동물 실험에서 유망한 결과를 보여주었으며, 일부는 임상 시험 단계에 진입했습니다2. 특히, 암 세포가 성장과 증식에 이 아미노산에 크게 의존하기 때문에 글루타민 대사는 치료 표적으로 많은 관심을 받고 있습니다3.
글루타민은
임상적 질환 및 스트레스 조건에서 중요한 역할을 하는
비필수/조건부 필수 아미노산입니다4.
암 세포 내 글루타민은
아미노산 및 뉴클레오티드 생합성을 위한 질소 공급원뿐 아니라
트리카르복실산(TCA) 회로와 지질 생합성 경로를 보충하기 위한
탄소 공급원으로도 작용합니다.
따라서
암 세포는
글루타민에 '중독'되어 있습니다5.
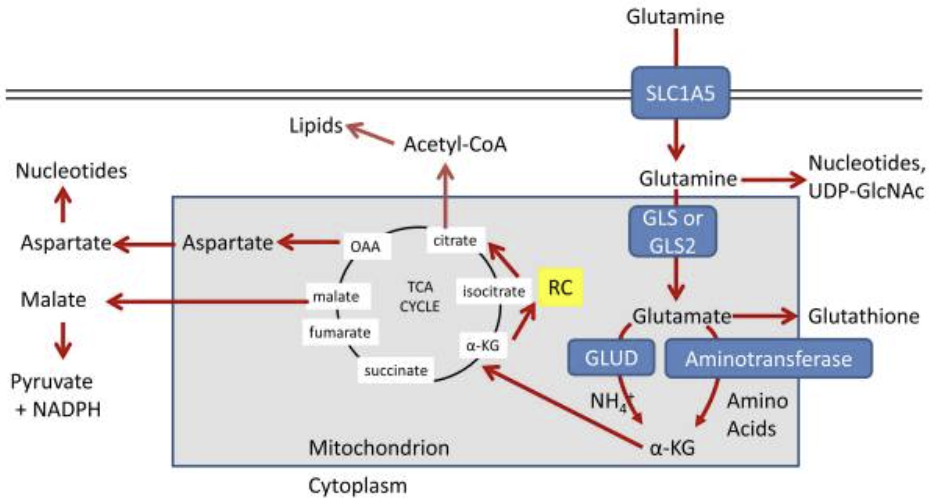
글루타민 대사 및 글루타민 운반체, 글루타미나제, 아미노전달효소,
산화환원 균형과 밀접하게 연관된 대사 네트워크는
암 세포 생존에 필수적입니다6.
글루타민 대사 과정의 각 단계를 표적으로 삼는 접근법은
암 치료에서 유망한 결과를 보여주며,
약물 표적 발견과 항암제 후보물질 개발을 촉진했습니다3.
또한
면역 체크포인트 억제제가 암 치료에 널리 사용됨에 따라
종양 미세환경(TME) 내 글루타민 차단 메커니즘에 대한 관심이 크게 증가했습니다7.
이 리뷰는 암 세포 내 글루타민 대사 과정의 각 단계를 요약하고 임상적 개입 기회를 강조합니다. 또한 저항 메커니즘과 TME 내 글루타민 차단 메커니즘의 역할을 논의합니다.
The role of glutamine in cancer cell growth
Rapidly proliferating cancer cells take up glutamine from plasma via various amino acid transporters, and then it is converted to glutamate in the mitochondria by the two forms of glutaminase: kidney-type glutaminase (GLS)1 and liver-type GLS28. Notably, GLS-mediated deamination of glutamine to glutamate is the first and rate-limiting step of glutaminolysis, making it an attractive druggable target9. GLS1 is overexpressed in various cancer cells, and this phenotype is associated with a higher disease stage and a poor prognosis10. Mechanistically, the expression of GLS1 is regulated indirectly by Myc (via repression of miR-23a and miR-23b) and mTORC111,12. Unlike GLS1, GLS2 suppresses the proliferation and migration of cancer cells13. In hepatocellular carcinoma (HCC), GLS2 inhibited proliferation in vitro and lung metastasis in a xenograft mouse model by inhibiting the small GTPase Rac113. However, several studies have shown that GLS2 is highly expressed in triple-negative basal-like breast cancer (TNBC) and metastatic lung cancer and that it confers radioresistance in advanced human cervical cancer cells, suggesting that GLS2 may reduce reactive oxygen species (ROS) levels by increasing the level of cellular reduced glutathione (GSH), NADH, or NADPH14,15,16. The confounding results regarding the function of GLS2 in cancer metabolism suggest that it may act in a context-specific manner17.
The TCA cycle is an essential hub for several metabolic pathways and for the interconversion of metabolites, which are renewed constantly in rapidly proliferating cancer cells18. Thus, replenishment of metabolic intermediates via the TCA cycle is vital to cancer cells, making them reliant on glutamine, a phenomenon called anaplerosis8. During anaplerosis, mitochondrial glutamate dehydrogenase 1 (GLUD1) plays a key role by catalyzing the conversion of glutamate to alpha-ketoglutarate (α-KG) and releasing ammonia, which regulates autophagy and neutralizes the intracellular pH in cancer cells19,20. α-KG is generated for the TCA cycle and is used for oxidative phosphorylation (OXPHOS)21. In addition, glutamine-derived α-KG is oxidized to succinate and fumarate, which maintain the TCA cycle in cancer cells by providing ATP, NADH, and FADH2 and by acting as oncometabolites22. Indeed, GLUD1 is overexpressed in various cancer cells, promoting epithelial-mesenchymal transition and drug resistance23. Mechanistically, Myc drives glutaminolysis by upregulating GLUD1 and induces a concurrent increase in the expression of GLS and SLC1A524.
Amino acids are required by cancer cells for proliferation under genotoxic, oxidative, and nutritional stress conditions; these amino acids serve as building blocks for protein synthesis and act as substrates for glucose, lipid, and nucleic acid synthesis25,26. In particular, glutamine plays a vital role in this process not only by providing a carbon source to the TCA cycle but also by acting as a nitrogen source for the biosynthesis of alanine, aspartate, and serine (Fig. 1)8. Therefore, the role of aminotransferases such as glutamate pyruvate transaminase (GPT) and glutamate oxaloacetate transaminase (GOT) in glutamine metabolism in cancer cells has been studied extensively8. Regarding GPTs, cytosolic GPT1 and mitochondrial GPT2 play major roles in energy metabolism in cancer cells by providing alanine for protein synthesis and by replenishing TCA cycle intermediates27. Indeed, GPT2 is a significant contributor to tumorigenesis in breast cancer, glioblastoma, and KRAS-driven colorectal cancer (CRC) cells28,29,30,31. Because cancer cells do not take up aspartate very well, GOT fuels tumorigenesis by providing cytosolic aspartate, which is used as a precursor for protein and nucleotide synthesis and for redox homeostasis32. Cytosolic GOT1 and mitochondrial GOT2, which together comprise the malate-aspartate shuttle, interconvert oxaloacetate and aspartate using glutamate or α-KG as substrates33. Indeed, both GOT1 and GOT2 are overexpressed in KRAS-driven pancreatic ductal adenocarcinoma (PDAC) cells34,35. Glutamine is also required for de novo synthesis of asparagine via asparagine synthetase (ASNS), which is induced by either the amino acid response or the unfolded protein response pathways36,37. Asparagine activates mTORC1 and contributes to the biosynthesis of purines and pyrimidines, as well as to the exchange of extracellular amino acids such as histidine, aspartate, and serine37. Indeed, the role of ASNS in tumorigenesis and metastasis has been reported, and it is associated with poor survival in various types of breast cancer, non-small cell lung cancer (NSCLC), and sarcoma38,39,40.
글루타민의 암 세포 성장 역할
빠르게 증식하는 암 세포는
혈장으로부터 다양한 아미노산 운반체를 통해 글루타민을 흡수한 후,
미토콘드리아에서 두 가지 형태의 글루타민아제(신장형 글루타민아제(GLS)1과 간형 GLS28)에 의해
글루타메이트로 전환됩니다.
특히,
GLS에 의한 글루타민의 글루타메이트로의 탈아미노화는
글루타민 분해의 첫 번째이자 속도 제한 단계로,
약물 표적으로서의 잠재력을 지니고 있습니다9.
GLS1은 다양한 암 세포에서 과발현되며,
이 현상은 질병 단계가 높고 예후가 불량한 것과 연관되어 있습니다10.
기전적으로 GLS1의 발현은 Myc(miR-23a 및 miR-23b 억제를 통해)와 mTORC1에 의해 간접적으로 조절됩니다11,12. GLS1과 달리 GLS2는 암 세포의 증식과 이동을 억제합니다13.
간세포암(HCC)에서 GLS2는 in vitro에서 증식을 억제하고 이종 이식 마우스 모델에서 폐 전이를 억제함으로써 소형 GTPase Rac1을 억제했습니다13. 그러나 여러 연구에서 GLS2는 삼중 음성 기저형 유방암(TNBC)과 전이성 폐암에서 고도로 발현되며, 진행성 인간 자궁경부암 세포에서 방사선 저항성을 부여한다는 것이 밝혀져, GLS2가 세포 내 환원형 글루타티온(GSH), NADH, 또는 NADPH 수준을 증가시켜 활성 산소 종(ROS) 수준을 감소시킬 수 있음을 시사합니다14,15,16. GLS2의 암 대사 기능에 대한 모순된 결과는 그것이 맥락에 따라 특이적으로 작용할 수 있음을 시사합니다17.
TCA 회로는 여러 대사 경로의 필수적인 허브이며 대사 산물의 상호 전환을 조절하며,
이는 빠르게 증식하는 암 세포에서 지속적으로 재합성됩니다18.
따라서
TCA 회로를 통한 대사 중간체의 보충은 암 세포에 필수적이며,
이는 글루타민에 의존하는 현상인 아나플로레시스(anaplerosis)를 유발합니다8.
아나플레로시스 동안,
미토콘드리아 글루타메이트 탈수소효소 1 (GLUD1)은
글루타메이트를 알파-케토글루타레이트 (α-KG)로 전환하고
암세포에서 자가포식을 조절하고
포 내 pH를 중화시키는 암모니아를 방출하는 촉매 역할을 합니다19,20.
α-KG는
TCA 사이클을 위해 생성되며 산화적 인산화 (OXPHOS)에 사용됩니다21.
또한 글루타민 유래 α-KG는
수크신산과 푸마르산으로 산화되어
ATP, NADH, FADH2를 공급하고 온코메타볼라이트로 작용하여
암 세포의 TCA 사이클을 유지합니다22.
실제로
GLUD1은 다양한 암 세포에서 과발현되어
상피-간질 전환과 약물 저항성을 촉진합니다23.
기전적으로,
Myc는
LUD1 발현을 증가시켜 글루타민 분해를 촉진하며,
동시에 GLS와 SLC1A5 발현도 증가시킵니다24.
암 세포는
유전독성, 산화적, 영양 결핍 스트레스 조건 하에서 증식하기 위해 아미노산이 필요하며,
이러한 아미노산은 단백질 합성의 구성 요소로 작용하며 글루코스, 지질, 핵산 합성의 기질로 기능합니다25,26.
특히
글루타민은 TCA 회로에 탄소원을 공급하는 것 외에도
알라닌, 아스파르트산, 세린의 생합성에 필요한 질소원으로 작용하여
따라서
암 세포에서의 글루타민 대사에서
글루타메이트 피루vate 트랜스아미나제(GPT) 및 글루타메이트 옥살아세테이트 트랜스아미나제(GOT)와 같은
아미노전달효소의 역할은 광범위하게 연구되어 왔습니다8.
GPT의 경우, 세포질 GPT1과 미토콘드리아 GPT2는
단백질 합성을 위한 알라닌을 공급하고 TCA 회로 중간체를 보충함으로써
암 세포의 에너지 대사에서 주요 역할을 합니다27.
실제로 GPT2는
유방암, 글리오blastoma, KRAS 유도 대장암(CRC) 세포에서
종양 발생에 중요한 역할을 합니다28,29,30,31.
암 세포는
아스파르테이트를 잘 흡수하지 않기 때문에
GOT는 세포질 아스파르테이트를 공급하여 종양 발생을 촉진합니다.
이 아스파르테이트는 단백질 및 핵산 합성의 전구체로 사용되며 산화환원 균형 유지에도 기여합니다32. 세포질 GOT1과 미토콘드리아 GOT2는 말산-아스파르트산 셔틀을 구성하며, 글루타메이트 또는 α-KG를 기질로 사용하여 옥살아세테이트와 아스파르트산을 상호 변환합니다33. 실제로 GOT1과 GOT2는 모두 KRAS에 의해 유발된 췌관 선암(PDAC) 세포에서 과발현됩니다34,35. 글루타민은 아스파라긴 합성효소(ASNS)를 통해 아스파라긴의 de novo 합성에 필수적이며, 이는 아미노산 반응 또는 미접힘 단백질 반응 경로를 통해 유도됩니다36,37. 아스파라긴은 mTORC1을 활성화하고 푸린 및 피리미딘의 생합성에 기여하며, 히스티딘, 아스파르트산, 세린과 같은 세포외 아미노산의 교환에도 관여합니다37. 실제로 ASNS의 종양 발생 및 전이에서의 역할이 보고되었으며, 유방암, 비소세포 폐암(NSCLC), 육종 등 다양한 유형의 암에서 예후가 불량한 것과 연관되어 있습니다38,39,40.
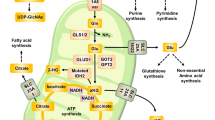
Fig. 1: Interlinked networks involved in glutamine metabolism.
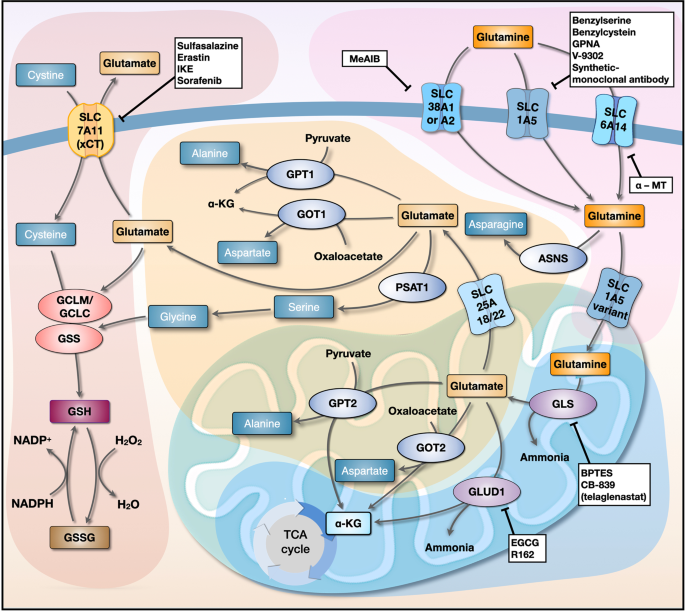
Glutamine transporters (SLC1A5, SLC38A1/SLC38A2, and SLC6A14) expressed on the cell membrane transport glutamine into the cytosol. Next, the SLC1A5 variant transports glutamine to the mitochondrial matrix, where it is converted to glutamate by GLS; this is the rate-limiting step of glutaminolysis. Glutamine-derived glutamate is catalyzed into α-KG by GLUD1, GOT2, and GPT2 to release ammonia, aspartate, and alanine, respectively. Glutamine-derived glutamate in the mitochondria is also transported to the cytosol by SLC25A18/SLC25A22. GOT1, which is part of the malate-aspartate shuttle, contributes to the maintenance of redox homeostasis by converting OAA to aspartate, and GPT1 converts pyruvate to alanine. SLC7A11 transports cysteine to the cytosol in exchange for glutamate. Glutamine-derived glutamate and cysteine are ligated by GCLM/GCLC, which is in turn utilized by GSS to form GSH, which scavenges cellular ROS. Inhibitors of each step of glutamine metabolism are shown in white boxes. GLS, glutaminase; α-KG, α-ketoglutarate; GLUD1, glutamate dehydrogenase 1; GOT, glutamate oxaloacetate transaminase; GPT, glutamate pyruvate transaminase; GCLM, glutamate-cysteine ligase modifier subunit; GCLC, glutamate-cysteine ligase catalytic subunit; GSS, glutathione synthetase; GSH, reduced glutathione; ROS, reactive oxygen species; ASNS, asparagine synthetase; PSAT1, phosphoserine aminotransferase 1.
Targeting glutaminase and transaminase as a treatment for cancer
GLS, which is highly expressed in cancer cells and plays a role in cancer progression, has been investigated extensively as a druggable target41. Bis-2-(5-phenylacetamido-1,2,4-thiadiazol-2-yl)ethyl (BPTES), a potent orally available GLS1 inhibitor that spares GLS2, shows promising antitumor effects against human lymphoma B cells in vitro and in a xenograft mouse model42; it also suppresses the growth of platinum-resistant CRC and ovarian cancer cells, suggesting that combined treatments based on conventional drugs and glutamine-modulating compounds will yield clinically relevant results43,44. Recently, another selective inhibitor of GLS1, CB-839 (telaglenastat), showed no significant side effects in preclinical trials and is currently undergoing full clinical trials45. Previous studies showed that it did not significantly suppress the growth of KRAS-derived PDAC cells in vitro or in vivo because these cells mounted an adaptive metabolomic response, suggesting the importance of combined therapy for avoiding metabolic adaptation in response to GLS inhibition45. Thus, clinical trials are currently testing the following drugs in combination with CB-839: nivolumab as a treatment for melanoma, renal cell carcinoma (RCC), and NSCLC (clinicaltrials.gov ID: NCT02771626); everolimus for RCC (clinicaltrials.gov ID: NCT03163667); palbociclib for KRAS-derived PDAC, NSCLC and CRC (clinicaltrials.gov ID: NCT03965845); and cabozantinib for advanced RCC (clinicaltrials.gov ID: NCT03428217)46. Furthermore, CB-839 increased the radiosensitivity of head and neck squamous carcinoma (HNSCC) and NSCLC cells both in vitro and in a xenograft mouse model by abolishing GSH synthesis47,48, making it useful for concurrent chemotherapy and radiotherapy in a clinical setting.
Previous studies have shown that targeting GLUD1 inhibits the proliferation and migration of cancer cells, suggesting that GLUD1 is a druggable target for cancer therapy23. Epigallocatechin gallate (EGCG), an inhibitor of GLUD1 and 2, suppresses the proliferation of neuroblastoma, glioma, and CRC cells49. Recently, the purpurin analog R162 (an inhibitor of GLUD1) also showed promising results with respect to attenuating the proliferation of breast, NSCLC, and glioma cells in vitro and in patient-derived xenograft mouse models23. In addition, cotreatment of docetaxel-resistant NSCLC with docetaxel plus R162 inhibited cancer cell growth and metastasis both in vitro and in xenograft mouse models, again suggesting that combination therapy with anticancer drugs plus a GLUD1 inhibitor is an effective cancer treatment23.
글루타미나제와 트랜스아미나제를 표적으로 한 암 치료법
암 세포에서 고도로 발현되며 암 진행에 역할을 하는 GLS는 약물 표적으로서 광범위하게 연구되어 왔습니다41. Bis-2-(5-phenylacetamido-1,2,4-thiadiazol-2-yl)ethyl (BPTES), GLS2를 선택적으로 억제하는 강력한 경구 투여 가능한 GLS1 억제제로, 체외 실험 및 이종 이식 마우스 모델에서 인간 림프종 B 세포에 대한 유망한 항종양 효과를 보여주었습니다42; 또한 백금 내성 대장암 및 난소암 세포의 성장도 억제하여, 전통적 약물과 글루타민 조절 화합물을 결합한 치료법이 임상적으로 의미 있는 결과를 가져올 수 있음을 시사합니다43,44. 최근 GLS1의 선택적 억제제인 CB-839 (telaglenastat)는 전임상 시험에서 유의미한 부작용을 보이지 않았으며 현재 완전한 임상 시험 중입니다45. 이전 연구에서 이 약물은 체외 및 체내에서 KRAS 변이 PDAC 세포의 성장을 유의미하게 억제하지 못했으며, 이는 이러한 세포가 GLS 억제에 대한 적응성 대사 반응을 보였기 때문으로, GLS 억제에 대한 대사 적응을 방지하기 위해 복합 치료의 중요성을 시사합니다45. 따라서 현재 CB-839와 결합하여 다음과 같은 약물을 임상 시험에서 테스트 중입니다: 멜라노마, 신세포암(RCC), 및 비소세포폐암(NSCLC) 치료를 위한 니볼루맙(clinicaltrials.gov ID: NCT02771626); RCC 치료를 위한 에버롤리무스(clinicaltrials.gov ID: NCT03163667); KRAS 변이 PDAC 치료를 위한 팔보시클리브, NSCLC 및 CRC(임상시험 등록번호: NCT03965845); 그리고 진행성 RCC에 대한 cabozantinib(임상시험 등록번호: NCT03428217)46. 또한 CB-839는 GSH 합성을 억제함으로써 체외 및 이종 이식 마우스 모델에서 두경부 편평상피암(HNSCC) 및 NSCLC 세포의 방사선 감수성을 증가시켰습니다47,48, 이는 임상 환경에서 화학요법과 방사선 요법의 병용 치료에 유용할 수 있습니다.
이전 연구들은 GLUD1을 표적화하는 것이 암 세포의 증식 및 이동을 억제함을 보여주었으며, 이는 GLUD1이 암 치료를 위한 약물 표적일 수 있음을 시사합니다23. GLUD1 및 2의 억제제인 에피갈로카테킨 갈레이트(EGCG)는 신경모세포종, 뇌종양, 대장암 세포의 증식을 억제합니다49. 최근 GLUD1 억제제인 푸르푸린 유사체 R162도 체외 실험 및 환자 유래 이종 이식 마우스 모델에서 유방암, NSCLC, 뇌종양 세포의 증식을 억제하는 유망한 결과를 보여주었습니다23. 또한, 도세탁셀 내성 NSCLC에 도세탁셀과 R162를 병용 투여했을 때 체외 및 이종 이식 마우스 모델에서 암 세포의 성장과 전이를 억제했으며, 이는 항암제와 GLUD1 억제제의 병용 요법이 효과적인 암 치료법임을 다시 한 번 시사합니다23.
Role of glutamine metabolism in redox homeostasis
ROS levels are elevated persistently in proliferating cancer cells, and ROS damage DNA and cellular components; therefore, redox homeostasis plays a pivotal role in protecting cancer cells against them. Notably, GSH acts as a critical antioxidant that protects cancer cells from any form of programmed cell death (i.e., autophagy, apoptosis, necroptosis, and ferroptosis)50,51,52. Given that glutathione is a tripeptide composed of glutamate, glycine, and cysteine, glutamine-derived glutamate and cysteine need to be ligated by glutamate-cysteine ligase (GCL), which itself comprises two separately encoded proteins: a catalytic subunit (GCLC) and a modifier subunit (GCLM)53,54. Next, glutathione synthetase (GSS) adds glycine to the ligated glutamate-cysteine (Fig. 1)53,54. While glutamate and glycine are abundant in cells, cysteine is the least abundant amino acid; therefore, it must be transported into the cells by SLC7A11 (xCT) in exchange for glutamate, which implies that SLC7A11-mediated GSH biosynthesis largely relies on glutamine metabolism55. Intriguingly, GOT1 and malic enzyme 1 (ME1) are also associated with redox homeostasis; in PDAC, GOT1 and ME1 maintain the NADPH/NADP+ ratio via the malate-aspartate shuttle by maintaining GSH levels56. In addition, we previously showed that upon inhibition of glutamine, cancer cells reduce the amount of GSH by exporting oxidized glutathione (GSSG) out of the cell via GSSG transporters and multiple-drug resistance-associated proteins57 and by extracellular degradation of GSSG58. Given that glutamine metabolism increases the amount of GSH in cells by maintaining the NADPH/NADP+ ratio and by preventing export and extracellular degradation of GSSG, glutamine is the primary amino acid that controls cellular GSH homeostasis.
산화 환원 항상성에서 글루타민 대사의 역할
ROS 수준은 증식하는 암세포에서 지속적으로 상승하며, ROS는 DNA 및 세포 성분을 손상시킵니다. 따라서 산화 환원 항상성은 암세포를 이러한 손상으로부터 보호하는 데 중요한 역할을 합니다. 특히, GSH는 모든 형태의 프로그램된 세포사(자가포식, 아폽토시스, 네크로토시스, 페로토시스)로부터 암세포를 보호하는 중요한 항산화제 역할을 합니다50,51,52. 글루타티온은 글루타메이트, 글리신, 시스테인으로 구성된 트리펩티드이기 때문에, 글루타민에서 유래한 글루타메이트와 시스테인은 글루타메이트-시스테인 리가아제(GCL)에 의해 연결되어야 합니다. GCL은 두 개의 별도로 암호화된 단백질, 즉 촉매 서브 유닛(GCLC)과 변형 서브 유닛(GCLM)으로 구성되어 있습니다53,54. 다음으로 글루타티온 합성효소(GSS)는 결합된 글루타메이트-시스테인에 글리신을 추가합니다(그림 1)53,54. 글루타메이트와 글리신은 세포 내 풍부하지만, 시스테인은 가장 적은 아미노산이기 때문에, 시스테인은 글루타메이트와 교환되어 SLC7A11(xCT)에 의해 세포 내로 운반되어야 합니다. 이는 SLC7A11 매개 GSH 생합성이 글루타민 대사55에 크게 의존함을 의미합니다. 흥미롭게도 GOT1과 말산 효소 1(ME1)도 환원-산화 균형과 연관되어 있습니다. PDAC에서 GOT1과 ME1은 말산-아스파르트산 셔틀을 통해 GSH 수준을 유지함으로써 NADPH/NADP+ 비율을 조절합니다56. 또한, 우리는 이전 연구에서 글루타민 억제 시 암 세포가 GSSG 운반체와 다약제 내성 관련 단백질57을 통해 세포 외로 산화 글루타티온(GSSG)을 배출하고, 세포 외 분해58을 통해 GSSG의 양을 감소시켜 GSH 수준을 낮춘다는 것을 보여주었습니다. 글루타민 대사는 NADPH/NADP+ 비율을 유지하고 GSSG의 세포 내 배출 및 세포 외 분해를 방지함으로써 세포 내 GSH 양을 증가시키므로, 글루타민은 세포 내 GSH 항상성을 조절하는 주요 아미노산입니다.
Targeting redox homeostasis for cancer treatment
Approximately one-third of glutamine taken up by human fibroblast cells is exchanged for cysteine by SLC7A1159,60. This suggests that SLC7A11 not only plays a critical role in protein and GSH synthesis through cysteine uptake but also dictates glutamine dependence61. Therefore, targeting SLC7A11 is a promising therapeutic option, and its efficacy can be increased by combining it with drugs that target glutaminolysis60. Indeed, sulfasalazine (which inhibits SLC7A11) effectively suppresses the proliferation of glutamine-depleted TNBC in vitro and in vivo60. Moreover, glutamine-dependent PDAC is sensitive to the SLC7A11 inhibitor erastin, which induces ferroptosis62,63. However, although erastin shows antitumor effects, it has not entered clinical trials because it is poorly soluble in water, and its metabolism in vivo is unpredictable; therefore, imidazole ketone erastin (IKE) and piperazine erastin were developed (which are more soluble in water), and both show strong antitumor effects against diffuse large B-cell lymphoma (DLBCL) and fibrosarcoma64,65. Intriguingly, and as mentioned above, PDAC cells are dependent on GOT1 and the malate-aspartate shuttle; GOT1 knockout combined with cysteine depletion by erastin or IKE showed potent antitumor effects against these cells by reducing GSH and increasing ferroptosis66. In addition, sorafenib, a kinase inhibitor approved for the treatment of RCC and HCC, inhibits SLC7A11 to suppress the growth of these tumors via the induction of ferroptosis67,68.
산화환원 항상성을 표적으로 한 암 치료
인간 섬유아세포가 흡수한 글루타민의 약 1/3은 SLC7A1159,60에 의해 시스테인으로 교환됩니다. 이는 SLC7A11이 시스테인 흡수를 통해 단백질 및 GSH 합성에 중요한 역할을 할 뿐만 아니라 글루타민 의존성을 결정한다는 것을 시사합니다61. 따라서 SLC7A11을 표적화하는 것은 유망한 치료 옵션이며, 글루타민 분해(glutaminolysis)를 표적화하는 약물과 결합하면 그 효능이 향상될 수 있습니다60. 실제로, SLC7A11을 억제하는 설파살라진(sulfasalazine)은 체외 및 체내에서 글루타민 결핍 TNBC의 증식을 효과적으로 억제합니다60. 또한, 글루타민 의존성 PDAC는 철분 사멸(ferroptosis)을 유도하는 SLC7A11 억제제 에라스틴(erastin)에 민감합니다62,63. 그러나 에라스틴은 항종양 효과를 보이지만, 물에 용해도가 낮고 체내 대사 과정이 예측하기 어려워 임상 시험에 진입하지 못했습니다. 이에 물에 더 용해되는 이미다졸 케톤 에라스틴(IKE)과 피페라진 에라스틴이 개발되었으며, 두 물질 모두 확산성 대세포 B세포 림프종(DLBCL)과 섬유육종에 대해 강력한 항종양 효과를 나타냈습니다64,65. 흥미롭게도, 위에서 언급된 바와 같이 PDAC 세포는 GOT1과 말레이트-아스파르테이트 셔틀에 의존합니다. GOT1 결손과 에라스틴 또는 IKE에 의한 시스테인 고갈을 결합하면 GSH 감소와 페로프토시스 증가를 통해 이러한 세포에 대한 강력한 항종양 효과를 나타냈습니다66. 또한, RCC 및 HCC 치료에 승인된 키나제 억제제인 소라페닙은 SLC7A11을 억제하여 페로토시스를 유도함으로써 이러한 종양의 성장을 억제합니다67,68.
Targeting glutamine transporters as a treatment for cancer
Cancer cells require an abundant supply of glutamine from the extracellular milieu; therefore, upregulation of glutamine transporters SLC1A5, SLC38A1, SLC38A2, and SLC6A14 at the cell membrane is required (Fig. 1)69. Indeed, high expression of these transporters contributes to cancer cell growth and is a marker of clinically poor outcomes for patients with NSCLC, prostate cancer, breast cancer, and acute myeloid leukemia70,71,72,73. Thus, cancer treatment strategies have focused on pharmacological inhibition of these transporters.
글루타민 운반체를 표적으로 한 암 치료법
암 세포는
세포 외 환경으로부터 풍부한 글루타민 공급이 필요하며,
따라서 세포막에서 글루타민 운반체
SLC1A5, SLC38A1, SLC38A2, 및 SLC6A14의 발현이 증가해야 합니다(그림 1)69.

실제로 이러한 운반체의 고발현은
암 세포의 성장에 기여하며,
비소세포폐암(NSCLC), 전립선암, 유방암, 급성 골수성 백혈병 환자의
임상적 예후가 불량한 지표로 작용합니다70,71,72,73.
따라서
암 치료 전략은 이러한 운반체의
약리학적 억제에 초점을 맞추고 있습니다.
SLC1A5
SLC1A5 (ASCT2) is an obligatory sodium-dependent transporter of neutral amino acids, which are exchanged for asparagine, threonine, or serine74. SLC1A5 has high affinity for glutamine, particularly in an acidic environment75, and is thus more effective at transporting glutamine into cancer cells that thrive in acidic environments76. Indeed, SLC1A5 is highly expressed in various solid cancers73,76,77, such as squamous lung cancer, in which it is responsible for approximately 50% of glutamine uptake73. The expression of SLC1A5 is regulated by various transcriptional regulators, including ATF4 and Myc78,79. In TNBC, high expression of ATF4 and Myc is associated with overexpression of SLC1A5 and indicates poor survival outcomes77. In addition, Myc-dependent expression of ATF4 in DLBCL cells, human colon adenocarcinoma cells, and mouse embryonic fibroblasts drives the expression of SLC1A5 during metabolic adaptation to stress conditions73,80,81. A recent study showed that HIF-2α-mediated overexpression of SLC1A5 variants in mitochondria plays an essential role in glutamine metabolism in pancreatic cancer cells by inducing chemotherapy resistance82.
Therefore, SLC1A5 is a promising druggable target83. Benzylserine and benzylcysteine were the first molecules found to inhibit SLC1A5 in breast and gastric cancer cells, but they are nonspecific84,85. L-γ-glutamyl-p-nitroanilide (GPNA) suppresses the growth of TNBC, different types of lung cancer, and neuroblastoma cells73,77,81. In addition, combined treatment with GPNA and a monoclonal antibody (cetuximab) targeting EGFR effectively suppressed the growth of gastric cancer cells in vitro and in vivo86. However, amino acid analogs are unsuitable for clinical use due to their low affinity, lack of specificity, and toxicity87. V-9302 (2-amino-4-bis (aryloxy benzyl) aminobutanoic acid) was originally discovered as an SLC1A5 inhibitor; it showed a 100-fold increase in potency over GPNA and attenuated the growth of cancer cells, including HCC, CRC, lung cancer, and breast cancer cells83. Recent studies have shown that synthetic monoclonal antibodies specific for SLC1A5 (i.e., KM4008, KM4012, and KM4018) are an effective therapeutic option for suppressing glutamine-dependent growth of CRC cells, but their efficacy in other cancer cells remains unclear88. Despite the significance of SLC1A5 in some cancer cells, there are few specific and effective SLC1A5-inhibiting drugs89.
SLC38A1 and SLC38A2
SLC38A1 (SNAT1) and SLC38A2 (SNAT2) are sodium-dependent neutral amino acid transporters that drive glutamine influx into cells90. SLC38A1 is overexpressed in melanoma, breast, gastric, osteosarcoma, and endometrial cancer cells, showing a close association with proliferation and migration91,92,93. SLC38A2 is highly expressed in prostate cancer, HCC, and TNBC cells, thereby contributing to tumorigenesis94,95. Interestingly, silencing of SLC1A5 does not suppress the proliferation of epithelial cervical cancer and osteosarcoma cells; rather, it induces an amino acid starvation response by upregulating the expression of SLC38A1, suggesting that SLC38A1 is a major importer of glutamine into these cells90. In addition, amino acid starvation upregulates SLC38A2 via activation of GCN2 and ATF4, which help to maintain the intracellular glutamine pool36,90. Therefore, strategies designed to target glutamine metabolism should consider the combined blockade of these transporters.
N-methyl-aminoisobutyric acid (MeAIB) has been investigated in studies attempting to elucidate the function of SLC38A1 and/or SLC38A2 in various cells; indeed, MeAIB exerts an antitumor effect against various cancer cells90,96. Recently, it was proposed that the aforementioned drug V-9302 targets SLC38A2 and SLC7A5 rather than SLC1A587. A previous study supported this, showing that treatment of SLC1A5-knockdown HNSCC cell lines with V-9302 led to marked inhibition of glutamine metabolism, thereby suppressing growth and proliferation both in vitro and in vivo97. Therefore, combination therapy with V-9302 and SLC1A5-specific inhibitors may be a promising therapeutic option for some cancers97.
SLC6A14
SLC6A14, also known as amino acid transporter B0,+ (ATB0,+), maintains a unidirectional influx of glutamine, coupled with 2 Na+ and 1 Cl-, along a transmembrane gradient98. Given the functional role of SLC6A14 in extending the range of amino acid uptake (including glutamine and leucine, both of which are activators of mTORC1), as well as providing substrates for SLC1A5 and SLC7A5, the molecule has attracted much attention99,100. Indeed, SLC6A14 is overexpressed in colon, cervical, ER-positive breast, and pancreatic cancer cells and is associated with their proliferation101,102,103,104. High expression of SLC6A14 in PDAC and CRC cells is closely associated with metastasis and a poor outcome101,105,106. Mechanistically, SLC6A14 expression is regulated by the Wnt signaling pathway, and genetic or pharmacological inhibition of the transporter and its downstream effectors suppresses the growth of CRC cells both in vitro and in vivo102.
Given that tryptophan is a substrate for SLC6A14, the inhibitor α-methyltryptophan (α-MT) suppresses the growth of SLC6A14-positive breast cancer, PDAC, and CRC cells but not SLC6A14-negative cells107,108,109. Combined treatment of pancreatic cancer cells with gemcitabine and α-MT significantly inhibited proliferation and migration110. Although the role of SLC6A14 in cancer cells is becoming clearer, few compounds targeting SLC6A14 have been developed; thus, an effective drug targeting this transporter needs to be developed.
SLC1A5
SLC1A5 (ASCT2)는 중성 아미노산을 나트륨에 의존하여 운반하는 필수 운반체로, 아스파라긴, 트레오닌, 또는 세린과 교환됩니다74. SLC1A5는 특히 산성 환경에서 글루타민에 높은 친화성을 보입니다75, 따라서 산성 환경에서 번영하는 암 세포로 글루타민을 운반하는 데 더 효과적입니다76. 실제로 SLC1A5는 편평상피 폐암을 포함한 다양한 고형 암에서 고도로 발현되며73,76,77, 이 중 편평상피 폐암에서는 글루타민 섭취의 약 50%를 담당합니다73. SLC1A5의 발현은 ATF4와 Myc78,79와 같은 다양한 전사 조절 인자에 의해 조절됩니다. 삼중 음성 유방암(TNBC)에서 ATF4와 Myc의 고발현은 SLC1A5의 과발현과 연관되어 있으며, 이는 불량한 생존 예후를 나타냅니다77. 또한, DLBCL 세포, 인간 대장 선암 세포, 마우스 배아 섬유아세포에서 Myc에 의존적인 ATF4 발현은 스트레스 조건에 대한 대사적 적응 과정에서 SLC1A5 발현을 촉진합니다73,80,81. 최근 연구에서 미토콘드리아 내 HIF-2α에 의해 매개되는 SLC1A5 변이체의 과발현이 췌장암 세포에서 글루타민 대사 및 화학요법 저항성을 유도하는 데 필수적인 역할을 한다는 것이 밝혀졌습니다82.
따라서 SLC1A5는 유망한 약물 표적입니다83. 벤질세린과 벤질시스테인은 유방암 및 위암 세포에서 SLC1A5를 억제하는 첫 번째 분자로 발견되었지만, 이들은 비특이적입니다84,85. L-γ-글루타밀-p-니트로아닐라이드 (GPNA)는 삼중 음성 유방암 (TNBC), 다양한 유형의 폐암, 및 신경모세포종 세포의 성장을 억제합니다73,77,81. 또한, GPNA와 EGFR을 표적으로 하는 단일클론 항체(cetuximab)의 병용 치료는 체외 및 체내에서 위암 세포의 성장을 효과적으로 억제했습니다86. 그러나 아미노산 유사체는 낮은 친화력, 비특이성, 독성으로 인해 임상 사용에 부적합합니다87. V-9302 (2-아미노-4-비스(아릴옥시 벤질) 아미노부타노산)는 원래 SLC1A5 억제제로 발견되었으며, GPNA보다 100배 높은 효능을 보였고 HCC, CRC, 폐암, 유방암 세포의 성장을 억제했습니다83. 최근 연구에서 SLC1A5에 특이적인 합성 단일클론 항체(예: KM4008, KM4012, KM4018)는 CRC 세포의 글루타민 의존성 성장을 억제하는 효과적인 치료 옵션으로 나타났지만, 다른 암 세포에서의 효능은 아직 명확하지 않습니다88. SLC1A5가 일부 암 세포에서 중요한 역할을 함에도 불구하고, 특정적이고 효과적인 SLC1A5 억제제는 여전히 부족합니다89.
SLC38A1 및 SLC38A2
SLC38A1(SNAT1) 및 SLC38A2(SNAT2)는 세포 내 글루타민 유입을 촉진하는 나트륨 의존성 중성 아미노산 운반체입니다90. SLC38A1은 멜라노마, 유방, 위, 골육종, 자궁내막암 세포에서 과발현되며, 증식 및 이동성과 밀접한 연관성을 보여줍니다91,92,93. SLC38A2는 전립선 암, 간세포암(HCC), 삼중음성 유방암(TNBC) 세포에서 고도로 발현되어 종양 발생에 기여합니다94,95. 흥미롭게도, SLC1A5의 발현을 억제해도 상피성 자궁경부암 및 골육종 세포의 증식을 억제하지 않으며, 오히려 SLC38A1의 발현을 증가시켜 아미노산 결핍 반응을 유도합니다. 이는 SLC38A1이 이러한 세포로의 글루타민 주요 수입체임을 시사합니다90. 또한, 아미노산 결핍은 GCN2 및 ATF4 활성화를 통해 SLC38A2 발현을 증가시켜 세포 내 글루타민 풀을 유지하는 데 도움을 줍니다36,90. 따라서 글루타민 대사 표적 전략은 이러한 운반체에 대한 복합적 차단 전략을 고려해야 합니다.
N-메틸-아미노이소부티르산(MeAIB)은 다양한 세포에서 SLC38A1 및/또는 SLC38A2의 기능을 규명하기 위한 연구에서 조사되었습니다. 실제로 MeAIB는 다양한 암 세포에 대해 항종양 효과를 발휘합니다90,96. 최근에, 앞서 언급된 약물 V-9302가 SLC1A5 대신 SLC38A2와 SLC7A5를 표적한다는 제안이 제기되었습니다87. 이전 연구는 이를 뒷받침했으며, SLC1A5 발현을 억제한 HNSCC 세포주에 V-9302를 처리했을 때 글루타민 대사 억제가 현저히 증가하여 체외 및 체내에서 성장과 증식이 억제되었다고 보고되었습니다97. 따라서 V-9302와 SLC1A5 특이적 억제제의 병용 요법은 일부 암에 대한 유망한 치료 옵션이 될 수 있습니다97.
SLC6A14
SLC6A14(아미노산 운반체 B0,+ (ATB0,+))는 2 Na+와 1 Cl-와 결합하여 세포막을 통해 글루타민의 일방적 유입을 유지합니다98. SLC6A14가 아미노산 흡수 범위(글루타민과 류신 포함, 둘 다 mTORC1 활성화제)를 확장하는 기능적 역할과 SLC1A5 및 SLC7A5의 기질 공급원으로서의 역할 때문에 이 분자는 많은 관심을 받았습니다99,100. 실제로 SLC6A14는 대장, 자궁경부, ER 양성 유방, 췌장 암 세포에서 과발현되며, 이들의 증식과 연관되어 있습니다101,102,103,104. PDAC 및 CRC 세포에서의 SLC6A14 고발현은 전이와 불량한 예후와 밀접하게 연관되어 있습니다101,105,106. 기전적으로, SLC6A14 발현은 Wnt 신호전달 경로에 의해 조절되며, 운반체 및 그 하류 효과기의 유전적 또는 약리학적 억제는 체외 및 체내에서 CRC 세포의 성장을 억제합니다102.
트립토판이 SLC6A14의 기질이기 때문에, 억제제 α-메틸트립토판 (α-MT)은 SLC6A14 양성 유방암, PDAC, 및 CRC 세포의 성장을 억제하지만 SLC6A14 음성 세포에는 영향을 미치지 않습니다107,108,109. 췌장암 세포에 gemcitabine과 α-MT를 병용 투여하면 증식 및 이동이 유의미하게 억제되었습니다110. SLC6A14의 암 세포 내 역할이 점차 명확해지고 있지만, SLC6A14를 표적하는 화합물은 거의 개발되지 않았습니다. 따라서 이 운반체를 표적하는 효과적인 약물이 개발되어야 합니다.
Mechanisms that induce resistance to glutamine-targeting therapies
Although targeting glutamine metabolism is a promising therapeutic approach, few drugs have been developed. Tumor metabolism is affected by a multitude of microenvironmental factors, including nutrient availability. There are several mechanisms by which cancer cells escape the effects of inhibitors of glutamine metabolism; these include increased metabolic flexibility, uptake of extracellular amino acids via compensatory transporters and macropinocytosis, and expression of nutrient stress-response proteins (Fig. 2).
글루타민 표적 치료에 대한 저항성을 유발하는 메커니즘
글루타민 대사 표적화는
유망한 치료 접근법이지만,
개발된 약물은 거의 없습니다.
종양 대사는
영양소 가용성을 포함한 다양한 미세환경 요인에 의해 영향을 받습니다.
암 세포가 글루타민 대사 억제제의 효과를 회피하는 메커니즘에는
대사 유연성 증가,
보상성 운반체를 통한 세포외 아미노산 흡수 및 대식세포 내포작용,
영양 스트레스 반응 단백질 발현 등이 포함됩니다(그림 2).
Fig. 2: Resistance mechanisms used by cancer cells in response to glutamine starvation.
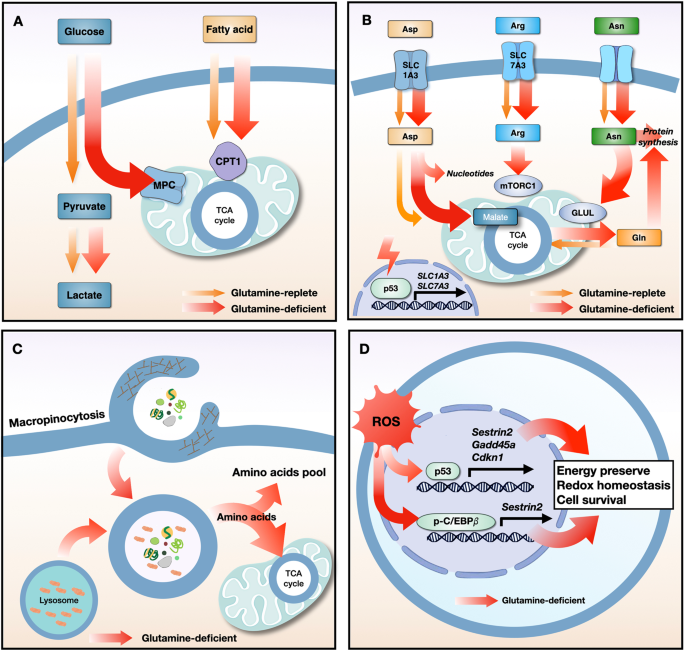
a Glutamine starvation induces metabolic flexibility, in which the influx of glucose-derived pyruvate via MPC and fatty acid-derived acyl-CoA via CPT1 into the mitochondria drives TCA cycle activity. b Under conditions of glutamine deprivation, the tumor suppressor protein p53 induces the expression of the SLC1A3 and SLC7A2 transporters. Aspartate uptake through SLC1A3 transporters increases the amount of malate, which is a TCA cycle intermediate, leading to an increase in oxidative phosphorylation and glutamine synthesis. Aspartate is used for nucleotide synthesis. Arginine uptake through SLC7A3 transporters restores mTORC1 expression, which is suppressed by glutamine depletion. The high level of intracellular asparagine increases the expression of GLUL proteins, thereby increasing glutamine and protein synthesis. c Under conditions of nutrient stress, macropinocytosis internalizes extracellular macromolecules to supply amino acids. Membrane ruffling aids in the uptake of extracellular macromolecules, such as serum albumin, via the formation of macropinosomes. After fusion between macropinosomes and lysosomes, albumin is degraded to supply amino acids to the cytosol and the mitochondrial TCA cycle. d Glutamine deprivation increases the expression of p53 and its target genes (Sestrin2, Gadd45a, and Cdkn1) and increases the phosphorylation of C/EBPβ and its target gene (Sestrin2), all of which maintain energy and redox balance and increase cancer cell survival. MPC, mitochondrial pyruvate carrier; CPT1, carnitine palmitoyltransferase I; TCA, tricarboxylic acid cycle; Asp, aspartate; Arg, arginine; Asn, asparagine; Gln, glutamine; ROS, reactive oxygen species; GLUL, glutamate-ammonia ligase; C/EBPβ, CCAAT/enhancer binding protein β.
Metabolic flexibility
Although glutamine is the primary carbon source for the TCA cycle in some cancer cells, replenishment of TCA cycle intermediates using alternative anaplerotic substrates reduces bioenergetic stress, thereby enabling resistance to inhibition of glutamine metabolism. There are two main anaplerotic flux pathways that feed the citric acid cycle: glutamine flux via glutaminase and glucose flux via pyruvate carboxylase110. Upon interruption of glutamine metabolism, glutamine-addicted tumor cells employ compensatory anaplerotic mechanisms via pyruvate carboxylase, which generates the oxaloacetate required to maintain TCA cycle flux; thus, the levels of pyruvate carboxylase can greatly affect the sensitivity of tumor cells to inhibition of glutamine metabolism111. In addition, deletion of GLS1 genes from Myc-driven liver tumors upregulates several metabolic compensatory pathways, including glycolysis and aminotransferases111. Thus, combined inhibition of glycolytic genes encoding hexokinase II or aminotransferases increases the efficacy of the GLS1 inhibitor CB-839111. In contrast, CB-839 showed no antitumor activity in PDAC mouse models due to the use of alternative metabolic pathways (e.g., fatty acid and lipid metabolism) by these cancer cells45. Integrated metabolomics and proteomics platforms revealed a marked increase in fatty acid oxidation-related metabolites, as well as proteome changes, in PDAC treated with GLS1 inhibitors, suggesting that treatments should target multiple metabolic pathways to overcome metabolic plasticity112,113.
Extracellular amino acid uptake via compensatory transporters
Amino acids, including aspartate, arginine, and asparagine, are associated with resistance to glutamine depletion. The tumor suppressor protein p53 increases the expression of the aspartate/glutamate transporter SLC1A3 and the arginine transporter SLC7A3 upon glutamine depletion, leading to resistance to treatments that deplete extracellular glutamine114,115. Increases in the levels of intracellular aspartate via SLC1A3 contribute to nucleotide synthesis and maintain the electron transport chain and TCA cycle114. Although the uptake of arginine by SLC7A3 transporters does not maintain TCA cycle flux under conditions of glutamine depletion, arginine activates mTORC1 and contributes to metabolic adaptation and tumor growth115. Uptake of extracellular asparagine prevents the death of glioblastoma cells in response to glutamine depletion by blocking the apoptotic function of a glutamine-induced endoplasmic reticulum stress marker protein, ATF4, and by increasing glutamate-ammonia ligase (GLUL)-mediated glutamine and protein synthesis116,117. Thus, blocking amino acid transporters or depleting amino acids such as L-asparaginase may be effective therapeutic strategies to overcome resistance to glutamine withdrawal.
Macropinocytosis
Macropinocytosis, a nutrient-scavenging pathway, is a compensatory route that supplies amino acids to nutrient-starved cancer cells harboring oncogenic mutations in KRAS or PTEN118,119,120. Experiments using isotope-labeled extracellular proteins show that when supplied with extracellular serum albumin, Ras-transformed cells, which rely on glutamine metabolism to support growth, utilize macropinocytosis to maintain proliferation under glutamine-limiting conditions120. More recent studies have shown that regional depletion of glutamine from PDAC tumors stimulates macropinocytosis by activating EGFR/PAK signaling and supplying glutamine via degradation of extracellular proteins in lysosomes121. Macropinocytosis also facilitates the survival of hypoxic HCC cells. Thus, HCC cells can internalize extracellular proteins by increasing the expression of a membrane ruffling protein called EH domain-containing protein 2, leading to resistance to glutamine deprivation under hypoxic conditions122. Although targeting macropinocytosis could be a key strategy for overcoming resistance to glutamine uptake blockade, further studies are necessary to examine whether macropinocytosis can overcome tumor cell resistance to glutamine antimetabolites or GLS inhibitors that target enzymes involved directly in glutamine metabolism.
Nutrient stress-response proteins
Limiting glutamine utilization regulates nutrient stress-response proteins and transcription factors. Upon glutamine deprivation, lung cancer cells increase the phosphorylated CCAAT/enhancer binding protein β (p-C/EBPβ)-dependent metabolic protein called Sestrin2 to maintain ATP levels and prevent excessive production of ROS through differential regulation of mTORC1 and mTORC2123. Sestrin2-mediated suppression of mTORC1 and mTORC2 activation reprograms lipid metabolism to limit ATP and NADPH consumption, thereby enabling cancer cells to survive under glutamine-depleted conditions. Other studies have shown that ROS production in response to glutamine deprivation increases the expression of p53-dependent genes (Gadd45a, Cdkn1, and Sestrin2) via B55α or IKKβ124,125. Upregulation of Gadd45a and Cdkn1 induces cell cycle arrest in response to glutamine deprivation, which alleviates oxidative stress and reduces energy consumption126,127. Cotargeting proteins involved in glutamine metabolism and the stress response under conditions of glutamine depletion would therefore be a promising therapeutic strategy for overcoming adaptive/resistance mechanisms in cancer cells.
Effects of targeting glutamine metabolism in the TME
The TME is a complex milieu that surrounds tumor cells, often providing immunosuppressive cover that facilitates immune invasion. Specifically, competition for nutrients or cell-intrinsic programming between cancer cells and immune cells induces nutrient deficiency and metabolic reprogramming of immune cells, leading to modulation of antitumor immunity128,129. Given that activation and differentiation of immune cells are coupled to metabolic reprogramming, regulating the metabolic activity of immune cells should be considered in the development of potential strategies that target glutamine metabolism130,131.
Glutamine metabolism in immune cells
Accumulating evidence shows that glutamine is an immunomodulatory nutrient in immune cells. Naïve T cells are metabolically quiescent, undergoing basal levels of glycolysis and glutaminolysis sufficient to maintain minimal biosynthesis; however, T-cell receptor (TCR)-stimulated activation increases the expression of the Myc transcription factor, glutamine transporters (SLC38A1, SLC38A2), and glutaminolysis-related enzymes (GLS, GLUD1, GOT, GPT) to meet bioenergetic and biosynthetic requirements, resulting in T-cell proliferation130,131,132,133. TCR-induced activation of mTORC1 and metabolic signaling pathways requires SLC1A5-dependent uptake of glutamine; indeed, an SLC1A5-deficient mouse model shows decreased induction of T helper 1 (Th1) and Th17 cells but not Th2, regulatory T (Treg), or CD8(+) T cells, leading to a decrease in proinflammatory T-cell responses134. Recent studies have shown that glutamine-derived α-KG regulates the differentiation of CD4(+) T cells into Th1-type effector T cells or Treg cells via DNA methylation and lipid homeostasis135,136. Mechanistically, α-KG decreases Treg differentiation by inhibiting FOXP3 and upregulating inflammatory cytokines such as IFN-γ, Tbet, and Rorc, suggesting that Th1-type effector T cells are more dependent on glutaminolysis than Treg cells136. Moreover, effector T cells are capable of adapting their metabolism in response to nutrient limitation. Activated T cells rely on glutamine-dependent OXPHOS to maintain energetic homeostasis under energy-related stress (e.g., low glucose levels)137.
The antitumor functions of natural killer (NK) cells are upregulated by the expression of c-Myc proteins, which are required for IL-2/IL-12-induced NK cell metabolism and function138. Although amino acids are essential for the function of NK cells, their main role in NK cells is the maintenance of signaling (e.g., via c-Myc or mTOR)139. Unlike other lymphocyte subsets, glutaminolysis and the TCA cycle do not sustain OXPHOS in activated NK cells. Glutamine withdrawal, but not inhibition of glutaminolysis, results in loss of c-Myc protein, reduced cell growth, and impaired NK cell responses138. Consistent with this, receptor-simulated production of IFN-γ by NK cells is not impaired under glutamine-limited conditions140.
In macrophages, glutamine metabolism is a critical metabolic pathway for differentiation. Macrophages undergo metabolic switching during differentiation into inflammatory (M1) or anti-inflammatory (M2) phenotypes. Tumor-associated macrophages (TAMs) can exhibit either an antitumor M1-like phenotype or a protumor M2-like phenotype. Glutamine starvation inhibits M2 polarization but not M1 polarization by suppressing UDP-GlcNAc biosynthesis and N-glycosylation of M2-related proteins such as Relmα, CD206, and CD301141. Consistent with this, glutaminolysis-derived α-KG promotes M2 activation by increasing fatty acid oxidation and Jmjd3-dependent epigenetic reprogramming of M2-related genes142. In contrast to the inhibition of glutaminolysis, pharmacological or genetic targeting of GLUL in macrophages reprograms M2-polarized macrophages to an M1-polarized phenotype143. Mechanistically, macrophage-specific inhibition of GLUL leads to accumulation of succinate and HIF-1α via glutamine-dependent γ-aminobutyric acid (GABA) shunting (thereby inhibiting vessel sprouting and metastasis) and via stimulation of T effector cells; however, IL-10-induced expression of GLUL promotes vessel sprouting, immunosuppression, and metastasis143.
Given the importance of glutamine metabolism to immune cells, including activated lymphocytes, it is crucial to determine whether blockade of glutamine metabolism in tumor cells hampers anticancer immune responses; the answer may be key to the success of therapeutic strategies targeting glutamine metabolism.
Glutamine blockade in the TME
The metabolism of cancer cells and immune cells in the TME is regulated by cell-intrinsic programs through mTORC1 signaling128. PET tracers showed that cancer cells rely heavily on glutamine uptake via mTORC1 signaling, while myeloid cells in the TME are more dependent on glucose, as are T cells and cancer cells (but to a lesser extent)128. Although administration of V-9302 to decrease glutamine availability increases glucose uptake by cancer cells and immune cells in allograft models, the growth of tumors harboring tumor-infiltrating Tregs, CD8(+) T cells, and NK cells is suppressed128. Given that cancer cells are much more dependent on glutamine than immune cells and that V-9302 does not impair CD8(+) T-cell viability and activation83, pharmacological inhibitors of SLC1A5 might have high therapeutic potential.
Accumulating evidence shows that inhibitors of glutamine metabolism, such as V-9302, JHU-083, and CB-839, elicit stronger antitumor effects when used in combination with immune checkpoint inhibitors144,145,146 (Fig. 3). In a previous study, we showed that V-9302 induces the expression of PD-L1 by tumor cells and augments immune evasion in synergistic murine models58. Mechanistically, glutamine limitation decreases GSH levels and sarco/endoplasmic reticulum Ca2+-ATPase (SERCA) glutathionylation, resulting in reduced SERCA activity58. Upregulation of cytosolic Ca2+ activates calcium/calmodulin-dependent protein kinase II (CAMKII), leading to aberrant NF-κB signaling and downstream expression of PD-L158. Therefore, agents that target glutamine utilization may, when used in combination with an anti-PD-L1 antibody, boost antitumor immunity58. Similar results were reported for several tumors147,148,149,150. Glutamine starvation increases the expression of PD-L1 in RCC and bladder cancer cells via the EGFR/ERK/c-Jun signaling pathway147,148. Furthermore, bladder tumors in mice supplemented with glutamine showed lower PD-L1 levels than control tumors148. In natural killer T-cell lymphoma (NKTCL), blocking SLC1A1-mediated glutamine addiction in tumor cells induced PD-L1 expression and inhibited CD8(+) T-cell activity149. As a therapeutic option, combined treatment with asparaginase and an anti-PD-1 antibody could be useful because glutamine-addicted cells are sensitive to asparaginase149.
Fig. 3: T-cell-mediated immune responses to glutamine-targeted treatment in cancer cells.
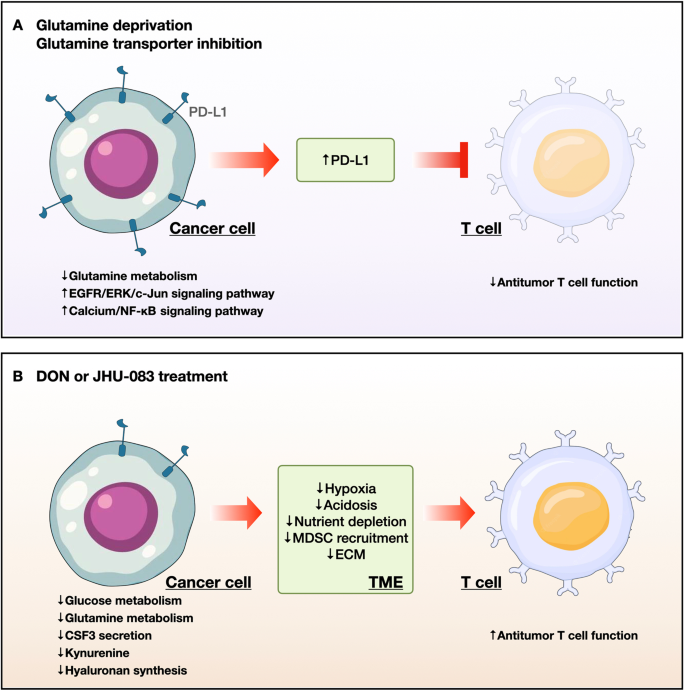
a Glutamine deprivation and transporter inhibition decrease glutamine metabolism, thereby boosting EGFR/ERK/c-Jun signaling and calcium/NF-kB signaling, leading to upregulation of PD-L1. PD-L1 suppresses antitumor immune responses by blocking T-cell activation in the tumor microenvironment. b Treatment with glutamine analogs, including DON and JHU-083, decreases glucose and glutamine metabolism, leading to inhibition of tumor growth via a decrease in hypoxia, acidosis, and nutrient depletion in the tumor microenvironment. Furthermore, DON decreases the recruitment of MDSCs by suppressing the secretion of CSF3 by tumor cells and blocking the production of the immunosuppressive metabolite kynurenine; this inhibits the synthesis of the hyaluronan-rich ECM, resulting in the activation and infiltration of T cells. PD-L1, programmed death-ligand 1; CSF3, colony stimulating factor 3; MDSC, myeloid-derived suppressor cell; ECM, extracellular matrix.
In contrast to PD-L1-mediated T-cell dysfunction induced by glutamine limitation, DON and its prodrug JHU-083 skew CD8(+) T cells toward a highly activated, persistent, and proliferative phenotype, thereby facilitating immunogenic cancer cell death58,149,150 (Fig. 3). DON treatment activates AMPK in cancer cells by increasing the AMP/ATP ratio and decreasing the expression of c-Myc proteins, thereby inhibiting glycolytic metabolism and suppressing cancer cell growth in vitro145. However, CD8(+) T cells in vitro overcome metabolic stress through glucose-dependent anaplerosis and acetate catabolism. Another study showed that in JHU-083-treated cancer cell allograft models, an increase in nutrient levels and oxygen and a decrease in the acidity of the TME resulted in T-cell-mediated tumor suppression145, whereas another study demonstrated the effects of JHU-083 on myeloid-derived suppressor cells (MDSCs) and TAMs150. JHU-083 markedly suppresses the recruitment of MDSCs by decreasing tumor-derived CSF3 levels via degradation of C/EBPβ, which in turn increases the numbers of proinflammatory TAMs and enhances antigen presentation to CD8(+) T cells, resulting in stronger T-cell responses150. In addition, JHU-083 reduces the expression of IDO (an enzyme that mediates tryptophan metabolism) by inhibiting the phosphorylation of STAT1 and STAT3 in tumor cells, MDSCs, and TAMs, thereby decreasing the kynurenine/tryptophan ratio and enhancing the functions of antitumor T cells150. Treatment of PDAC with DON decreases uridine diphosphate N-acetylglucosamine (UDP-GlcNAc) levels, which affects synthesis of the hyaluronan-rich extracellular matrix (ECM); this suggests that glutamine inhibitors such as DON could deplete the ECM and allow infiltration of tumors by CD8(+) T cells151.
Two compounds (V-9302 and JHU-083) that target SLC1A5 and glutamine metabolism in allograft models elicited different T cell-mediated immune responses; however, because these compounds do not reduce the activation or viability of CD8(+) T cells83,145, combined immunotherapy significantly improves their antitumor effects.
Conclusion
Glutamine metabolism plays a central role in regulating uncontrolled tumor growth by modulating bioenergetic and redox homeostasis and by serving as a precursor for the synthesis of biomass. Although targeting glutamine metabolism is a promising strategy for cancer therapy, there are many hurdles to be overcome before we develop a clinically effective drug. Metabolic flexibility or adaptation by cancer cells, as well as reduced antitumor immunity, may be unwanted consequences of inhibiting glutamine metabolism. A comprehensive understanding of the TME is of the utmost importance because it provides valuable insights into pathways that could be targeted by novel metabolic therapies for advanced or drug-resistant cancers.
References