대퇴부 통증
장경인대부위 통증
소리치는 자는 범인이 아니라 피해자다.



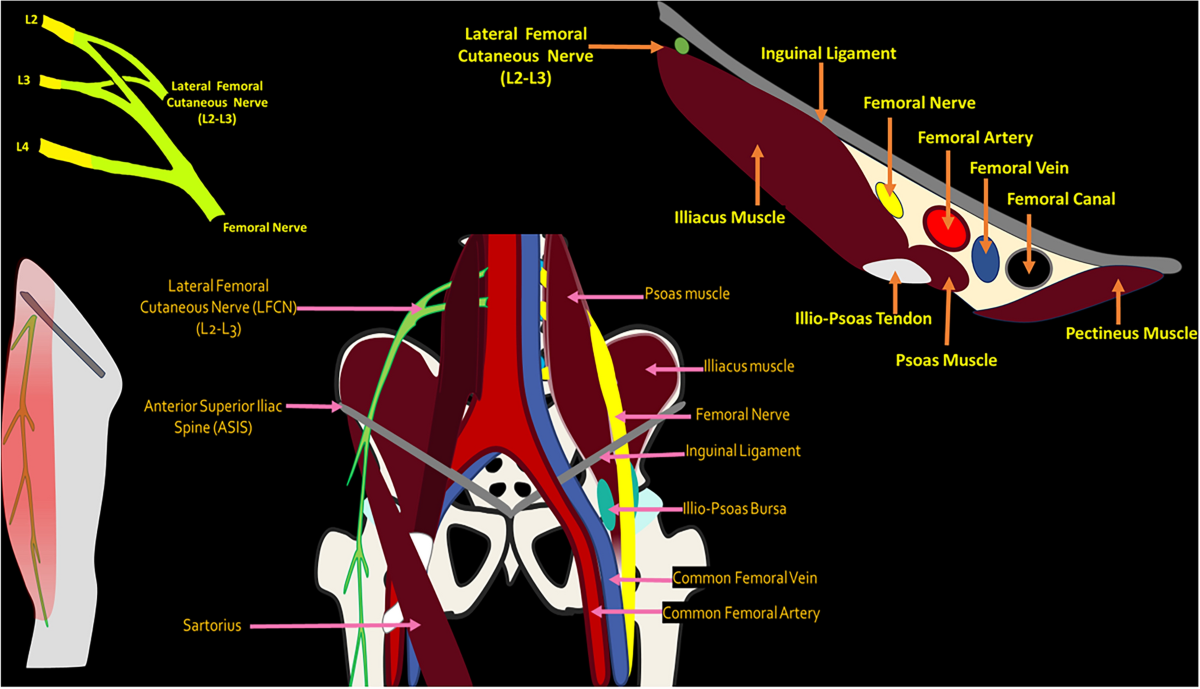
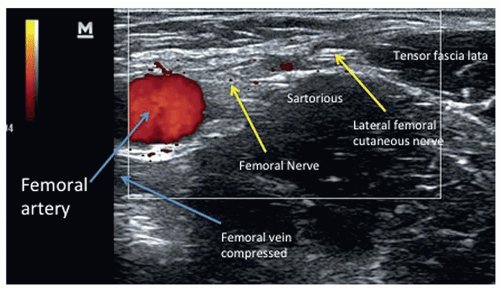
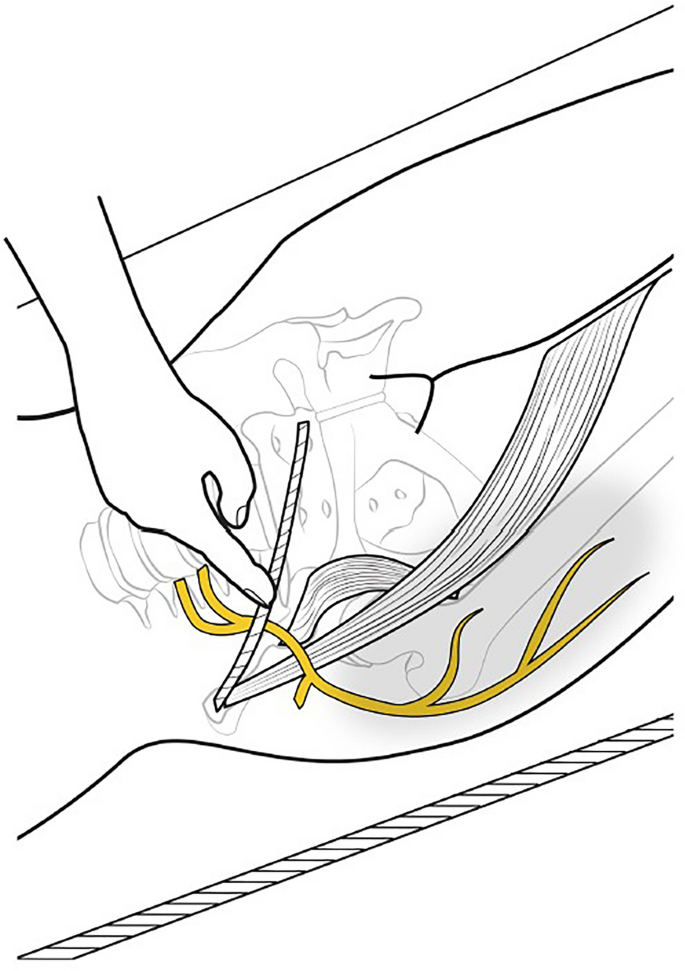
메랄지아 파레스테티카(Meralgia Paresthetica, MP)는 **측대퇴피부신경(Lateral Femoral Cutaneous Nerve, LFCN)**의 압박으로 인해 대퇴부 앞쪽-바깥쪽에 이상감각(저림, 화끈거림), 신경병증성 통증, 감각 변화가 발생하는 감각성 단일신경병증입니다. 주로 중년(특히 남성)에서 발생하며, 비만, 당뇨병, 임신, 꽉 조이는 옷, 외상, 수술 후 자세 등이 위험 요인입니다.
증상 및 진단
- 주요 증상: 대퇴부 바깥쪽의 타는 듯한 통증, 저림, 촉각 과민. 앉거나 서면 악화.
- 진단: 주로 임상 병력과 신체검사로 이루어지며, 배제 진단(exclusion diagnosis). 필요 시 초음파, MRI, 신경전도검사 등을 보조적으로 사용.
치료
치료는 단계적으로 진행됩니다:
- 보존적 치료 (초기 선택): 체중 감량, 꽉 조이는 옷 피하기, 진통제, 항우울제, 국소 캡사이신 크림, TENS 등. 많은 경우 자연 회복되거나 증상 완화.
- 주사 요법: 초음파 유도하 LFCN 차단술(스테로이드 + 국소마취제). 75~91%에서 효과적이며, 여러 번 주사가 필요할 수 있음.
- 펄스 고주파 치료(Pulsed Radiofrequency): 장기 완화 효과가 좋음 (63~100%).
- 수술: 보존적 치료 실패 시. 신경해방술(neurolysis) 또는 신경절제술(neurectomy). 절제술이 완전 통증 완화율이 높지만 (75~93%), 재발률 등에서 논란이 있음.
주요 결론 및 한계
대부분의 환자는 보존적 치료로 호전되며 예후가 좋습니다. 그러나 치료 효과에 대한 연구 결과가 상충되고, 대규모 무작위 대조 시험이 부족해 최적의 치료 프로토콜(주사 횟수, 약물 용량 등)이 명확하지 않습니다. 향후 더 많은 연구가 필요하다고 강조합니다.

Health Psychol Res
. 2023 Mar 15;11:71454. doi: 10.52965/001c.71454
Meralgia Paresthetica Review: Update on Presentation, Pathophysiology, and Treatment
Yanet de la Caridad Gomez 1, Edgar Remotti 2, Deandra Uju Momah 2, Emily Zhang 2,#, Daniel D Swanson 3, Rosa Kim 3, Ivan Urits 4, Alan D Kaye 5, Christopher L Robinson 2,✉
- Author information
- Article notes
- Copyright and License information
PMCID: PMC10019995 PMID: 36937080
Abstract
Purpose of Review
Meralgia paresthetica (MP) is a condition characterized by paresthesias, neuropathic pain, and alterations in sensorium of the anterolateral thigh secondary to impingement of the lateral femoral cutaneous nerve (LFCN). MP is generally diagnosed by clinical history and is often a diagnosis of exclusion. When diagnosis remains a challenge, diagnostic modalities such as ultrasound, MRI, electromyography, and nerve conduction studies have been utilized as an adjunct. This review summarizes the most recent medical literature regarding MP, its pathophysiology, presentation, and current treatment options.
Recent Findings
Treatment options for patients with MP range from lifestyle modifications and conservative management to surgical procedures. Initial management is often conservative with symptoms managed with medications. When conservative management fails, the next step is regional blocks followed by surgical management. The conflicting data for treatment options for MP highlight how the evidence available does not point to a single approach that’s universally effective for treating all patients with MP.
Summary
Despite the apparent success at treating MP with regional blocks and surgical interventions, much remains to be known about the dosing, frequency, and optimal interventions due to the inconclusive results of current studies. Further research including randomized controlled trials are needed to better understand the most optimal treatment options for MP including studies with a larger number of participants.
검토 목적
대퇴외측피부신경(LFCN)의 압박으로 인해 발생하는
대퇴부 전외측의 감각 이상, 신경병성 통증 및 감각 장애를 특징으로 하는 질환이
대퇴외측피부신경병증(MP)이다.
MP는
일반적으로 병력을 통해 진단되며
배제 진단을 통해 확인되는 경우가 많다.
진단이 어려운 경우 초음파, MRI, 근전도 검사, 신경전도 검사 등의 진단적 수단이 보조적으로 활용되어 왔다.
본 리뷰는 MP의 병태생리, 증상 양상 및 현재 치료 옵션에 관한 최신 의학 문헌을 종합한다.
최근 연구 결과
MP 환자의 치료 옵션은 생활습관 개선 및 보존적 치료부터 수술적 처치까지 다양하다. 초기 치료는 주로 보존적이며 약물로 증상을 관리한다. 보존적 치료가 실패할 경우, 다음 단계는 국소 신경차단술을 시행한 후 수술적 치료를 고려한다. MP 치료 옵션에 대한 상충되는 연구 결과는 현재 이용 가능한 증거가 모든 MP 환자에게 보편적으로 효과적인 단일 접근법을 제시하지 못함을 강조한다.
요약
국소 신경 차단 및 수술적 개입을 통한 MP 치료의 명백한 성공에도 불구하고, 현재 연구의 결정적이지 않은 결과로 인해 투여량, 빈도 및 최적의 개입 방법에 대해서는 아직 많은 부분이 알려지지 않았다. 더 많은 참가자를 대상으로 한 연구를 포함하여 무작위 대조 시험을 포함한 추가 연구가 MP에 대한 최적의 치료 옵션을 더 잘 이해하기 위해 필요하다.
Keywords: Meralgia Paresthetica, Lateral Femoral Cutaneous Nerve, Entrapment, Compression, Neurolysis, Neurectomy
Introduction
Meralgia paresthetica (MP) is a condition characterized by paresthesias, neuropathic pain, and alterations in the sensorium of the anterolateral thigh secondary to impingement of the lateral femoral cutaneous nerve (LFCN). It was first discovered in the late 1880s by Bernhardt and Roth who each independently published on the phenomenon.1 While initially referred to as Bernhardt-Roth syndrome, the name “meralgia paresthetica” termed by Roth was adopted soon thereafter, derived from the Greek words “meros” and “algos,” meaning “thigh” and “pain,” respectively.1,2
Although MP can present during any stage of life including childhood, it most often presents in the 5th decade of life and is more common in males.2,3 Patients with MP often present with subacute onset of pain of the anterolateral thigh which can be described as a burning, stinging or tingling sensation that is exacerbated even with light touch.4,5 Diagnosis often relies on clinically history alone but can be aided by imaging and nerve conduction studies.4,5 The majority of patients are managed conservatively with lifestyle modification, topical and oral analgesics, and injections with definitive surgical management reserved for refractory cases.4,5 In this review article, we explore MP with a focus on the clinical presentation, diagnosis, treat, and prognosis.
감각이상성 대퇴부 통증(MP)은
외측 대퇴 피부 신경(LFCN)의 압박으로 인해
대퇴부 전외측부의 감각 이상, 신경병성 통증 및 감각 이상이 나타나는 질환이다.
이 증후군은 1880년대 후반 베른하르트(Bernhardt)와 로트(Roth)가 각각 독립적으로 발표하며 처음 발견되었다.1
초기에는 베른하르트-로트 증후군(Bernhardt-Roth syndrome)으로 불렸으나,
로트가 명명한 “메랄지아 파레스테티카(meralgia paresthetica)”라는 명칭이 곧 채택되었으며,
이는 각각 “대퇴부”와 “통증”을 의미하는 그리스어 “메로스(meros)”와 “알고스(algos)”에서 유래하였다.1,2
MP는
유년기를 포함한 모든 연령대에서 발생할 수 있으나,
주로 50대에 발병하며 남성에서 더 흔하다.2,3
MP 환자는 대퇴부 전외측에 서서히 시작되는 통증을 호소하는 경우가 많으며,
이 통증은 화끈거림, 찌르는 듯한 통증 또는 저림으로 묘사되며 가벼운 접촉만으로도 악화된다. 4,5
진단은 주로 임상 병력만으로 이루어지지만 영상 검사와 신경전도 검사가 보조 수단이 될 수 있다.4,5 대부분의 환자는 생활습관 개선, 국소 및 경구 진통제, 주사 치료 등 보존적 방법으로 관리되며, 난치성 사례에만 수술적 치료가 시행된다.4,5 본 리뷰 논문에서는 임상 증상, 진단, 치료, 예후에 초점을 맞춰 MP를 탐구한다.
Pathophysiology
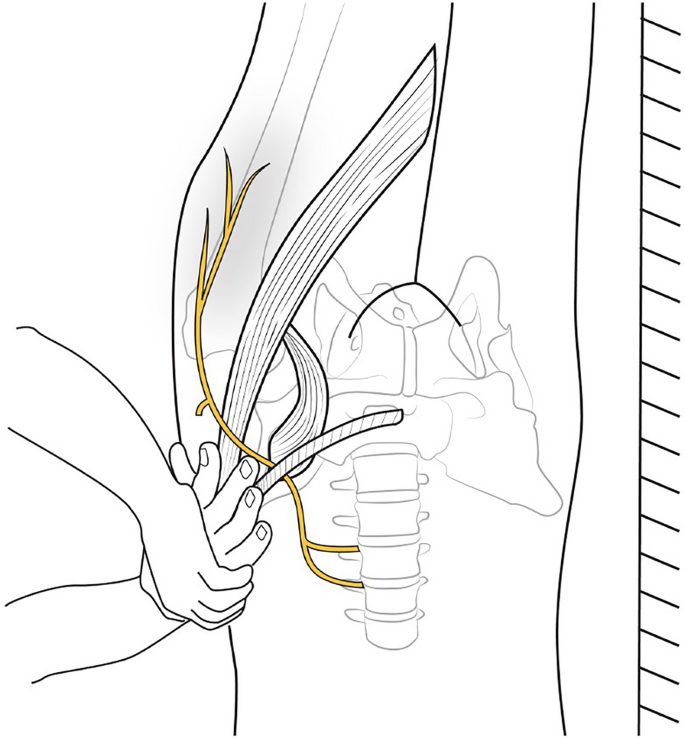
MP is attributed to the impingement of the lateral femoral cutaneous nerve (LFCN), a purely sensory nerve branch derived from the posterior divisions of the anterior rami of the L2 and L3 spinal nerves.6–8 As the nerve travels down the pelvis and lateral to the psoas muscle, it crosses the iliacus before passing beneath the inguinal ligament. The LFCN then traverses medial to the anterior superior iliac spine (ASIS), ultimately piercing the fascia lata before giving rise to its terminal cutaneous branches.6,7,9–11 Of note, the majority of cases are attributed to insults at the level of the inguinal ligament.10
Multiple diagnostic modalities have been investigated to evaluate pre- and post-evidence of pathologic changes secondary to MP; these pathological changes include polarized internodal swelling and endoneural vascular thickening, suggestive of a mechanistic injury pattern consistent with local demyelination and Wallerian degeneration.10 Ultrasound findings of MP include LFCN hypoechogenicity, anomalous perineurial borders, intraneural vascularity, enlarged neural cross sections, and diffuse focal thickening proximal to the anterior superior iliac spine (ASIS).12,13 Similar pathologic changes have been investigated with MRI neurography demonstrating persistent high signaling of the LFCN, nerve enlargement, and abnormal fat stranding all serving as indicators of potential injury.14
MP는
L2 및 L3 척추신경의 전지 분지에서 유래한
순수 감각 신경 가지인 외측 대퇴 피부 신경(LFCN)의 압박에 기인한다.6–8
이 신경은 골반을 따라 하강하며
요근의 외측을 지나 장골근을 가로질러 서혜 인대 아래로 통과한다.
이후 LFCN은 전상장골극(ASIS)의 내측을 가로지르며,
최종적으로 대퇴근막을 관통한 후 말단 피부 분지를 발생시킵니다.6,7,9–11
주목할 점은 대부분의 사례가
서혜 인대 수준에서의 손상에 기인한다는 것입니다.10
MP에 이차적인 병리학적 변화의 전후 증거를 평가하기 위해 다양한 진단 방법이 연구되었습니다; 이러한 병리학적 변화에는 극성 신경절간 부종 및 신경내 혈관 비후가 포함되며, 이는 국소 탈수초화와 월러 변성과 일치하는 기계적 손상 패턴을 시사한다.10
MP의 초음파 소견에는 LFCN 저음영화, 비정상적인 신경주위 경계, 신경내 혈관성, 신경 단면 확대, 그리고 전상장골극(ASIS) 근위부의 확산성 국소 비후가 포함된다. 12,13 유사한 병리학적 변화는 MRI 신경조영술을 통해 조사되었으며, LFCN의 지속적인 고신호 강도, 신경 비대, 비정상적인 지방 줄무늬가 모두 잠재적 손상의 지표로 작용함을 보여주었다.14
Epidemiology
Although cases of MP have been reported in all age groups, the condition primarily affects middle-aged adults in their fifth decade of life.15–19 Data are sparse and varied regarding the incidence of MP; for example, one study examined MP in a primary care cohort from 1990 to 1998 in the Netherlands found the incidence to be 4.3 per 10,000 person-years.20 Additionally, another population based study in Minnesota from 1990 to 1999 reported an incidence of 32.6 per 100,000 patient years.21 Finally, population and hospital-based studies from 2004-2011 reported an incidence of MP between 32.6-43/100,000 people.18,22 Interestingly, in the United States military, incidence rates are almost twice the average rate with the incidence being 62/100,000 service members.23 It is thought that this near doubling of incidence is due to prolonged load carriage, potentially leading to increased compression of the LFCN.23
While MP can occur secondary to stretching of the LFCN alone, many patients have predisposing risk factors that make them more susceptible.7 Common risk factors include obesity, diabetes, pregnancy, tight fitting clothing, toxins (lead and alcohol), infectious diseases (acquired immunodeficiency syndrome and leprosy), trauma, iatrogenic (direct surgical injury, prolonged prone positioning, and inadequate padding), and mass effect causing direct impingement on the nerve.6–8,10,11 Furthermore, anatomical variations have been described in the literature, with certain patterns, including an anterior or posterior relation of the LFCN to the ASIS, conferring higher rates of developing MP with an estimated 25% of patients having an anatomical variant of the LCFN.10,11
Patients with an elevated body mass index (BMI) show an increased odds ratio of developing MP ranging from 1.2-1.83 (OR 1.2, 95% CI 1.04–1.44, OR 1.83, 95% CI 1.02–3.29).18,19 The average BMI in patients diagnosed with MP is 28.0-33.8 kg/m2, and in patients with a BMI ≥ 30 kg/m2, there is nearly a doubling of the risk of developing MP compared to age and sex-matched controls, likely due to increased compression along the LFCN.18,24 Additionally, obese individuals may be more vulnerable to occupational factors affecting the LCFN, such as jobs where patients wear a seatbelt, tight clothing, or are standing for long periods of time.17,23 The co-occurrence of diabetes mellitus (DM) has also been investigated as a risk factor. A population-based study in Olmsted County, Minnesota examined 262 patients diagnosed with MP and found that patients with DM have almost 7.5 times higher rates of MP than the general population.18 Other studies that have found no association between DM and MP, but it may be due to differences in study populations and sources of data.22 Moreover, age is a risk factor with the average age of diagnosis being 49.8 ±12.8 years.17 Peak incidences for each gender differ and are between 41-50 years old for men and 51-60 years old for women.17,25,26 Despite studies reporting an increased prevalence of MP among males, the literature surrounding sex as a risk factor is inconclusive.17,22,27–29 Unfortunately, there have only been a limited number of cases of adolescent MP that have been documented, and adolescents may have different risk factors than those in middle-aged adults. About 39% of adolescents being treated for osteoid osteoma lesions at the hip subsequently develop MP with BMI being an unrelated factor.30,31
Iatrogenic MP can also be seen in patients who underwent prone positioning during surgery or during a stay in the intensive care unit (ICU), with the incidence ranging from 10.3-23.8%.15,19,28,32–36 Spinal surgeries such as thoracic and lumbar laminectomies have higher incidences of MP than other prone positioning surgeries, with risk factors including surgical times longer than 3.5 hours and an elevated BMI.19,28,32
MP 사례는 모든 연령대에서 보고되었으나,
이 질환은 주로 50대 중년 성인에게 영향을 미친다.15–19
MP의 발생률에 관한 데이터는 부족하고 다양하다;
예를 들어,
네덜란드에서 1990년부터 1998년까지 1차 진료 코호트를 대상으로 한 한 연구에서는
MP 발생률이 10,000인 년당 4.3건으로 나타났다.20
또한,
1990년부터 1999년까지 미네소타에서 수행된 또 다른 인구 기반 연구에서는
100,000환자 년당 32.6건의 발생률을 보고하였다. 21
마지막으로,
2004년부터 2011년까지의 인구 기반 및 병원 기반 연구에서는
MP 발생률이 인구 10만 명당 32.6~43명 사이로 보고되었다.18,22
흥미롭게도, 미국 군대에서는 발생률이 평균의 거의 두 배에 달하며,
군인 10만 명당 62명의 발생률을 보인다. 23
이러한 발생률의 거의 두 배 증가 현상은
장기간의 무게 운반으로 인해 좌측 복신경(LFCN)의 압박이 증가할 가능성이 있기 때문으로 여겨집니다.23
MP는 LFCN의 신장만으로 이차적으로 발생할 수 있지만,
많은 환자들은 이를 더 취약하게 만드는 선행 위험 요인을 가지고 있습니다.7
일반적인 위험 요인에는 비만, 당뇨병, 임신, 꽉 끼는 옷, 독소(납 및 알코올), 감염성 질환(후천성 면역 결핍 증후군 및 한센병), 외상, 의인성(직접적인 수술 손상, 장기간의 엎드린 자세, 부적절한 패딩), 및 신경에 직접적인 압박을 유발하는 종괴 효과 등이 있습니다.6–8,10,11 또한 문헌에는 해부학적 변이가 기술되어 있으며, 특히 LFCN이 ASIS에 대해 전방 또는 후방 위치에 있는 특정 패턴은 MP 발생률을 높이는 것으로 알려져 있습니다. 추정되기로는 환자의 약 25%가 LCFN의 해부학적 변이를 가지고 있습니다.10,11
체질량지수(BMI)가 높은 환자는 MP 발생 위험비(OR)가 1.2~1.83배 증가한다(OR 1.2, 95% CI 1.04–1.44; OR 1.83, 95% CI 1.02–3.29). 18,19 MP로 진단된 환자의 평균 BMI는 28.0-33.8 kg/m²이며, BMI ≥ 30 kg/m²인 환자는 연령 및 성별을 매칭한 대조군에 비해 MP 발병 위험이 거의 두 배에 달합니다. 이는 LFCN을 따라 압박이 증가하기 때문일 가능성이 높습니다.18,24 또한, 비만인 개인은 안전벨트 착용, 꽉 끼는 옷 착용, 장시간 서 있는 작업과 같이 좌측 쇄골하 신경(LCFN)에 영향을 미치는 직업적 요인에 더 취약할 수 있다.17,23 당뇨병(DM)의 동반 발생도 위험 요인으로 조사되었다. 미네소타주 올름스테드 카운티에서 실시된 인구 기반 연구에서는 MP로 진단받은 262명의 환자를 조사한 결과, 당뇨병 환자의 MP 발생률이 일반 인구 대비 약 7.5배 높은 것으로 나타났다.18 당뇨병과 MP 간 연관성을 발견하지 못한 다른 연구들도 있으나, 이는 연구 대상 집단과 데이터 출처의 차이에 기인할 수 있다. 22 또한 연령은 위험 요인으로, 평균 진단 연령은 49.8 ±12.8세이다.17 성별별 최고 발병률은 남성이 41-50세, 여성이 51-60세 사이이다.17,25,26 남성에서 MP 유병률이 증가한다는 연구 보고에도 불구하고, 성별을 위험 요인으로 보는 문헌은 결론적이지 않다.17,22, 27–29 안타깝게도 청소년기 MP 사례는 제한적으로만 기록되었으며, 청소년은 중년 성인과는 다른 위험 요인을 가질 수 있다. 고관절 골모세포종 병변으로 치료받는 청소년의 약 39%가 이후 MP를 발병하며, 체질량지수(BMI)는 관련 없는 요인으로 나타났다.30,31
의학적 MP는
수술 중 또는 중환자실(ICU) 입원 중 엎드린 자세를 취한 환자에서도 관찰되며,
발생률은 10.3~23.8%이다.15,19,28, 32–36
흉추 및 요추 척추판절제술과 같은 척추 수술은
다른 엎드린 자세 수술보다 MP 발생률이 높으며,
3.5시간 이상의 수술 시간 및 높은 BMI가 위험 요인으로 알려져 있다.19,28,32
Clinical Features
Patients with MP commonly present with persistent or intermittent dysesthesia or paresthesia. Common sensations include numbness, burning, aching or pain in the anterolateral aspect of the thigh.27 Patients often describe a “shooting” pain or a “troublesome tingling and itch-like sensation” that is localized to one thigh.25,37 This pain typically worsens with prolonged sitting, standing, or extending the hips.27,38 Patients describe an increased sensitivity to clothing and are unable to tolerate tight clothing or carrying keys in the pockets on the side of dysesthesia.39–41 A comparative study that examined 120 cases of MP found no difference in symptoms based on right or left thigh localization.42
The duration of MP can vary drastically, as symptoms can range from 0.5 months to 20 years.42 The mean duration of MP is 34 months with 36% of cases experiencing symptoms lasting from 12 months to 5 years.42 Of the cases evaluated, nearly 73% involved the lateral aspect of the thigh, and 26% of cases involved the anterior aspect of the thigh.42 The area of paresthesia typically does not extend past the medial line of the anterior thigh, and some patients may report lower lateral pain that is distinct from their thigh paresthesia.40,42 While most patients present with unilateral symptoms, bilateral presentation has been documented in about 10-18% of cases.33,42,43 Although patients with bilateral MP commonly present with the same clinical symptoms, there is the possibility of distinct etiology.33,36
Patients with iatrogenic MP present similarly to those with spontaneous MP.15,42 These patients can have symptoms that emerge on the same day as the surgery or even several months later with 92% of patients reporting initial symptoms within the first 12 hours.15,33
임상적 특징
대퇴부 전외측부 신경병증(MP) 환자는
일반적으로 지속적이거나 간헐적인 이감증 또는 감각 이상을 호소합니다.
흔한 증상으로는
대퇴부 전외측부의 저림, 화끈거림, 쑤시는 통증 또는 통증이 포함됩니다.27
환자들은 종종 한쪽 대퇴부에 국한된
“찌르는 듯한 통증” 또는 “괴로운 따끔거림과 가려움 같은 감각”을 묘사합니다.25, 37
이 통증은
장시간 앉아 있거나 서 있거나 고관절을 펴는 동작 시 악화되는 경향이 있습니다.27,38
환자들은 의복에 대한 민감도가 증가하여
불편감 부위의 주머니에 열쇠를 넣고 다니거나 꽉 끼는 옷을 입는 것을 견디지 못한다고 보고합니다.39–41
120명의 MP 환자를 대상으로 한 비교 연구에서는
통증이 오른쪽 또는 왼쪽 허벅지에 국한되는지에 따른 증상 차이는 발견되지 않았습니다.42
MP의 지속 기간은
0.5개월에서 20년까지 다양하여 극심한 차이를 보일 수 있다.42
MP의 평균 지속 기간은 34개월이며,
36%의 사례에서 12개월에서 5년까지 증상이 지속되었다. 42
평가된 사례 중
약 73%는 대퇴부 외측을,
26%는 대퇴부 전면을 침범하였다.42
감각이상 부위는 일반적으로 대퇴부 전면의 내측선을 넘어서지 않으며, 일부 환자는 대퇴부 감각이상과 별개로 하부 외측 통증을 호소할 수 있다.40, 42 대부분의 환자는 일측성 증상을 보이지만, 약 10-18%의 사례에서 양측성 증상이 보고되었습니다.33,42,43 양측성 MP 환자는 일반적으로 동일한 임상 증상을 보이지만, 원인이 다를 가능성도 있습니다.33,36
의인성 MP 환자는 자연발생성 MP 환자와 유사한 양상을 보인다.15,42 이 환자들은 수술 당일 또는 수개월 후에 증상이 나타날 수 있으며, 92%의 환자가 초기 증상을 수술 후 12시간 이내에 보고한다.15,33
Diagnosis
Diagnosis of MP is often clinical based on symptomatology with evidence of sensory nerve changes but preservation of motor nerve function. The neuropathy is typically unilateral in nature and more commonly observed in males as compared to females.44 Patients commonly present with pain, paresthesias and burning in the affected region. They can also experience diminished sensation of pain, heat, and touch.44 Symptoms can be exacerbated by light touch (e.g., from tight clothing) or sleeping on the affected side and alleviated with thigh flexion.44 If severe enough, patients can experience limitations on their mobility and walk with a limp secondary to pain.44
In the presence of symptoms, particularly atypical symptoms, diagnosis can be made using sensory nerve velocity studies. Testing will show evidence of nerve compression when compared against the unaffected side. However, even in patients of average body mass, performing such studies can be difficult.45 Electromyographic studies can also be used to rule out other causes of neuropathy. Ultrasound imaging, as well as CT or MRI can also be utilized to visualize nerve compression. Clinical suspicion for the condition should be high in patients with chronic metabolic disease (e.g. ,obesity and diabetes) who present with chronic neuropathy that is unresponsive to conservative management.46
진단
MP 진단은 주로 증상학적 근거에 기반하며,
감각 신경 변화의 증거가 있으나
운동 신경 기능은 보존된 상태입니다.
이 신경병증은 일반적으로 일측성이며
여성에 비해 남성에서 더 흔히 관찰됩니다.44
환자들은
주로 통증, 감각 이상, 그리고 해당 부위의 화끈거림을 호소합니다.
또한 통증, 열감, 촉각에 대한 감각이 감소할 수 있습니다.44
증상은 가벼운 접촉(예: 꽉 끼는 옷)이나 환측으로 잠을 자는 경우 악화되고,
대퇴부 굴곡 시 완화될 수 있습니다.44
증상이 심한 경우 환자는
통증으로 인해 이동이 제한되고 절뚝거리며 걸을 수 있습니다.44
증상이 존재할 경우, 특히 비정형 증상이 있을 때 감각 신경 속도 검사를 통해 진단할 수 있습니다. 검사 결과는 비영향 측과 비교 시 신경 압박 증거를 보여줍니다. 그러나 평균 체중의 환자에서도 이러한 검사를 수행하기 어려울 수 있습니다.45 근전도 검사는 신경병증의 다른 원인을 배제하는 데에도 활용될 수 있습니다. 초음파 영상 및 CT 또는 MRI도 신경 압박을 시각화하는 데 사용될 수 있습니다. 만성 대사 질환(예: 비만 및 당뇨병)을 가진 환자에서 보존적 치료에 반응하지 않는 만성 신경병증이 나타날 경우 이 질환에 대한 임상적 의심이 높아야 합니다.46
Differential Diagnosis
MP can present similarly to other neuropathies or radiculopathies depending on its severity. For this reason, diagnosis can be delayed by several months to years. The differential diagnosis for MP includes neuropathies of the upper lumbar plexus, as well as femoral neuropathy, pelvic tumors and metastases, and chronic appendicitis.44,47 Like MP, radiculopathy of the lumbar plexus presents with nerve pain; however, it is typically associated with back pain of the affected region (e.g., secondary to disk herniation) with paresthesia radiating along the associated nerve fibers. Lumbar pathology can be cause both sensory and motor deficits depending on the nerve fibers affected, while MP causes only sensory deficits.
Femoral neuropathy presents similarly to MP with nerve pain. However, its etiology differs, as it can arise secondary to surgery of the abdomen, hip and pelvis from iatrogenic injury, lithotomy positioning, local ischemia, or trochar insertion.45 Procedures requiring femoral catheterization can also increase the risk of hematoma formation and neuropathy development. Patients can present with unilateral knee pain, weakness, loss of knee extension, and loss of the patellar reflex.45 Similar to the aforementioned conditions, pelvic tumors and metastases can also cause neuropathic pain, depending on their size and location. However, lesions can cause abdominal pain, pressure and distention, as well as nausea and vomiting. In the context of the associated malignancy, patients can develop weight loss, fatigue, and generalized weakness. Finally, chronic appendicitis is a rare condition and an uncommon cause of nerve pain. It occurs secondary to appendiceal obstruction (e.g., fecalith, lymphoid hyperplasia, foreign bodies, and tumors).48 Patients may present with right lower quadrant abdominal pain.
감별 진단
MP는 중증도에 따라 다른 신경병증이나 신경근병증과 유사하게 나타날 수 있습니다.
이로 인해 진단이 수개월에서 수년 지연될 수 있습니다.
MP의 감별진단에는
상부 요추 신경총 신경병증, 대퇴신경병증, 골반 종양 및 전이, 만성 충수염 등이 포함됩니다.44,47
MP와 마찬가지로 요추 신경총 신경근병증도 신경통을 보이지만, 그러나 일반적으로 해당 부위의 요통(예: 추간판 탈출증에 이차적)과 연관된 감각 이상이 관련 신경 섬유를 따라 방사되는 특징이 있습니다. 요추 병변은 영향을 받는 신경 섬유에 따라 감각 및 운동 장애를 모두 유발할 수 있는 반면, MP는 감각 장애만 유발합니다.
대퇴신경병증은 신경통으로 MP와 유사하게 나타납니다.
그러나 그 원인은 다르며,
복부, 고관절 및 골반 수술 시 의학적 손상, 석고위 자세, 국소 허혈 또는 트로카 삽입으로 인해 이
차적으로 발생할 수 있습니다.45
대퇴동맥 카테터 삽입이 필요한 시술도 혈종 형성 및 신경병증 발생 위험을 증가시킬 수 있습니다.
환자는 일측성 무릎 통증, 근력 약화, 무릎 신전 장애, 슬개 반사 소실을 호소할 수 있다.45
앞서 언급한 질환들과 유사하게, 골반 종양 및 전이도 크기 및 위치에 따라 신경병성 통증을 유발할 수 있다. 그러나 병변은 복통, 압박감 및 팽만감, 메스꺼움 및 구토를 동반할 수 있다. 관련 악성 종양의 맥락에서 환자는 체중 감소, 피로, 전신 쇠약이 발생할 수 있다. 마지막으로 만성 충수염은 드문 질환이며 신경통의 흔하지 않은 원인이다. 이는 충수 폐쇄(예: 분변석, 림프구 증식, 이물질, 종양)에 이차적으로 발생한다.48 환자는 우측 하복부 통증을 호소할 수 있다.
Treatment
Treatment options for patients with MP range from lifestyle modifications and conservative management to surgical procedures (Table 1). Since most patients experience spontaneous recovery, conservative management is often the first-line treatment.49,50 Initial recommendations include lifestyle modifications such as avoidance of tight-fitting garments that compress the area around the waist and discussions about weight loss when appropriate.51 Symptoms are managed conservatively with medications including analgesics and nonsteroidal anti-inflammatory agents.51,52 An observational study including 277 patients reported that 50% of patients experienced satisfactory relief with these lifestyle modifications and conservative measures alone.52 Tricyclic antidepressants and anticonvulsive agents have also been documented as pharmacologic treatments used for symptom relief although the efficacy of these have not been systematically studied in MP.53 Topical capsaicin has been used to treat surface hypersensitivity.54 Transcutaneous electrical nerve stimulation (TENS) has been employed in patients with more disabling symptoms.55
Table 1. Clinical Efficacy and Safety of Treatments for Meralgia Paresthetica.
Author and YearGroups Studied and InterventionResults and FindingsConclusions
| Williams et al. 1991 | 277 patients were treated with a combination of conservative treatments, steroid injections, and surgical procedures. | Conservative treatments combined with injections successfully relieved symptoms in 91% of 277 patients, while the remaining 24 patients required surgical treatment. | Most patients with MP will not require surgical intervention. |
| Puig et al. 1995 | One patient was treated with topical 0.025% capsaicin cream 5 times daily. | Symptoms were markedly relieved after five days but returned after medication was stopped. | Topical capsaicin can provide symptomatic relief; however larger trials would be beneficial to confirm effect. |
| Fisher et al. 1987 | Three pregnant patients were treated by TENS. | All three patients had complete resolution of symptoms postpartum. | TENS may be an effective treatment for MP; however, in this study the cessation of pregnancy may have also helped with symptom resolution. |
| Dureja et al. 1995 | 40 patients were treated with a minimum of 5 LFCN blocks and diphenhyldantoin 100 -300 mg daily for 10-12 weeks. | 85% of patients experienced complete relief within 10 weeks, 5% had partial relief, and 10% showed no relief with up to six months of treatment. | Most patients with MP were successfully treated with a combination of conservative management and local injections. |
| Klauser et al. 2016 | 20 patients were treated with US-guided injection of steroids along the LFCN at three different levels in a mean of 2.25 sessions. | 75% of patients had complete resolution of symptoms and the remaining 25% had partial resolution of symptoms. | US-guided injection of corticosteroids at multiple levels of the LFCN can lead to sustained symptom resolution. |
| Kiliç et al. 2020 | Three groups were compared: single-dose US-guided LFCN injection (n=17) vs. TENS (n=16) vs. sham TENS (n=21). | Patients treated with US-guided LFCN injection experienced more pain relief compared to the other groups. | Both US-guided injections and TENS of the LFCN can have a therapeutic effect; however, US-guided injections show greater benefits. |
| Tagliafico et al. 2011 | 20 patients were treated with one or two US-guided injections of the LFCN. | 80% of patients experienced diminishing symptoms after a single injection while 20% of patients required a second injection for symptom relief. | Some patients may experience sustained symptom relief after a single nerve block while other require successive nerve blocks for sustained relief. |
| Haim et al. 2006 | 79 patients were treated with conservative management, nerve blocks, or surgery. | 21 out of 79 patients responded to conservative management. Out of the 58 treated with nerve blocks, 48 had complete symptom resolution after up to three injections. | Patients may require repeated nerve blocks for symptom relief. |
| Su et al. 2020 | One patient was treated with US-guided nerve hydrodissection of the LFCN using D5W during seven sessions of injections for two months. | Patient successfully recovered after treatment. | US-guided nerve hydrodissection is an emerging treatment; however, larger studies to confirm its efficacy are needed. |
| Philip et al. 2009 | One patient was treated with pulsed radiofrequency (PRF) of the LFCN at 42oC for 120 seconds. | The patient experienced complete and sustained cessation of pain without evident side effects. | PRF of the LFCN may be an effective and safe treatment in refractory cases. |
| Dalmau-Carolà 2009 | Two patients were treated with PRF of the LFCN. | One patient received two PRF treatments which resulted in total relief sustained at two years’ follow-up. The second patient was pain free after one PRF treatment. | PRF of the LFCN can result in sustained pain relief in patients. |
| Lee et al. 2016 | 11 patients underwent PRF of the LFCN. | 63.6% of the patient reported complete pain relief while the remaining patients experienced >50% pain reduction. No complications were observed. | PRF of the LFCN can result in immediate and sustained pain relief without complications. |
| Perryman et al. 2018 | A patient underwent placement of a wireless peripheral nerve stimulator along the LFCN. | The patient reported immediate pain relief that was sustained at a three month follow-up evaluation. | Wireless PNS may be an effective intervention; however, larger studies are required to better understand its efficacy and safety. |
| Barna et al. 2005 | A patient is treated with an implantable spinal cord stimulator. | Patient reported 100% pain relief and improved functional status. | An implantable spinal cord stimulator led to complete symptom resolution. |
| Ruiter et al. 2012 | 16 patients were treated with neurolysis or neurectomy. | 60% of patients who underwent neurolysis had complete pain relief while 75% of patients who underwent neurectomy had complete pain relief. | Neurectomy was slightly more effective than neurolysis in the treatment of MP. |
| Ruiter et al. 2015 | 22 patients were followed prospectively and assessed six weeks after neurolysis or neurectomy. | Complete recovery or almost complete recovery was seen in 37.5% of neurolysis cases and in 93.3% of neurectomy cases. | Neurectomy was more effective in treating MP than neurolysis. |
| Emamhadi 2012 | 14 patients were treated with neurolysis (5) or neurectomy (9). | All patients who underwent neurectomy had complete pain relief without recurrence while those who underwent neurolysis had recurrence of symptoms within nine months. | Patients treated with neurectomy have a lower incidence of symptom recurrence than patients treated with neurolysis. |
| Benezis et al. 2007 | 167 cases were treated with either neurolysis (153) or neurectomy (14). | 78% of patients treated with neurolysis reported complete or partial improvement while only 35.7% of patients treated with neurectomy reported improvement. | Neurolysis was more effective in treating MP than neurectomy; however, differences between the two treatment groups make the comparison difficult. |
| Morimoto et al. 2018 | 12 patients were treated with microsurgical deep decompression of the LFCN. | All patients reported symptom improvement with complete relief in nine (75%) patients and partial relief in three (25%) patients. | Microsurgical deep decompression can effectively treat MP. |
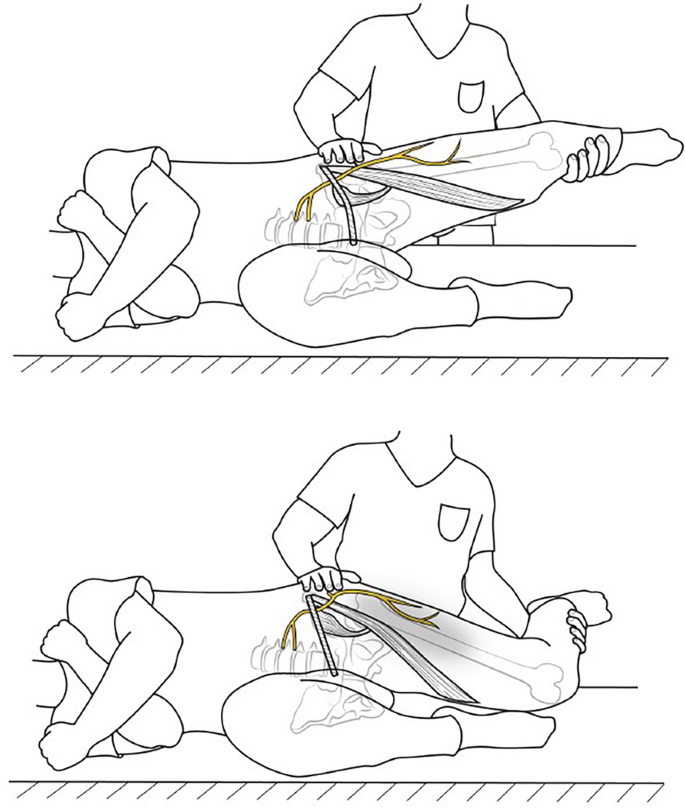
When conservative management fails, patients commonly transition to treatment with regional nerve blocks. Multimodal approaches combining corticosteroid injections of the LFCN with conservative pharmacologic treatments have been reported to have success rates between 85-91%.52,56 Ultrasound (US) technology is useful to correctly localize the LFCN and accurately guide the injection.57 This is important because blind injection can have a failure rate of up to 60% due to the anatomic variability of the LFCN.58
US-guided injections of the LFCN are most commonly a combination of a steroid and a local anesthetic.56,57,59,60 In a prospective study from 2020, 14 (82%) out of 17 patients treated with a single US-guided injection of the LFCN had significant decrease in pain symptoms.59 Another study followed 20 patients treated with US-guided injections of the LFCN at three different levels for 12 months and found that 15 (75%) patients had complete resolution of symptoms and the remaining 5 (25%) had partial resolution of symptoms after a mean of 2.25 injections.57 Other studies similarly report requiring multiple injections to achieve complete symptom resolution.56,60,61 Despite the apparent overall high success of local injections in treating patients with MP, there has yet to be a consensus in the literature on the frequency of injections and the dosage of the medications used.
The first case reporting the successful use of pulsed radiofrequency (PRF) neuromodulation of the LFCN in MP was in 2009.62 A single treatment with PRF resulted in complete pain relief that was sustained after 6 months without any evident side effects.62 A case report of a single patient who experienced complete resolution of symptoms after a second PRF procedure points to the possible benefit of repeating the procedure to achieve long-term relief.63 A retrospective study reviewed clinical outcomes of PRF in 11 patients with intractable meralgia paresthetica and reported complete pain relief in 63.6% of patients, with the rest experiencing >50% pain reduction.64
The available evidence in support of interventional pain procedures including peripheral nerve stimulation (PNS) and spinal cord stimulation (SCS) in the treatment of meralgia paresthetica is scarce. In a retrospective study of patients treated with implantable PNS systems over 30 years for the diagnosis of complex regional pain syndrome (CRPS), only one out of 165 patients had a PNS implant targeting the LFCN.65 More recently, a case report described the novel use of a PNS implanted along the course of the LFCN in a patient with severe refractory MP.66 The implanted device resulted in immediate pain relief that was sustained at a 3-month follow-up evaluation.66 The use of an SCS for the treatment of meralgia paresthetica has only been reported once in the literature.67,68 After the implantation of the device, the patient reported nearly complete pain relief at 8 months with no reported adverse effects.67 Although further studies are needed to firmly elucidate the long-term effectiveness and safety of these treatments, PNS and SCS are promising minimally invasive alternatives to surgery.
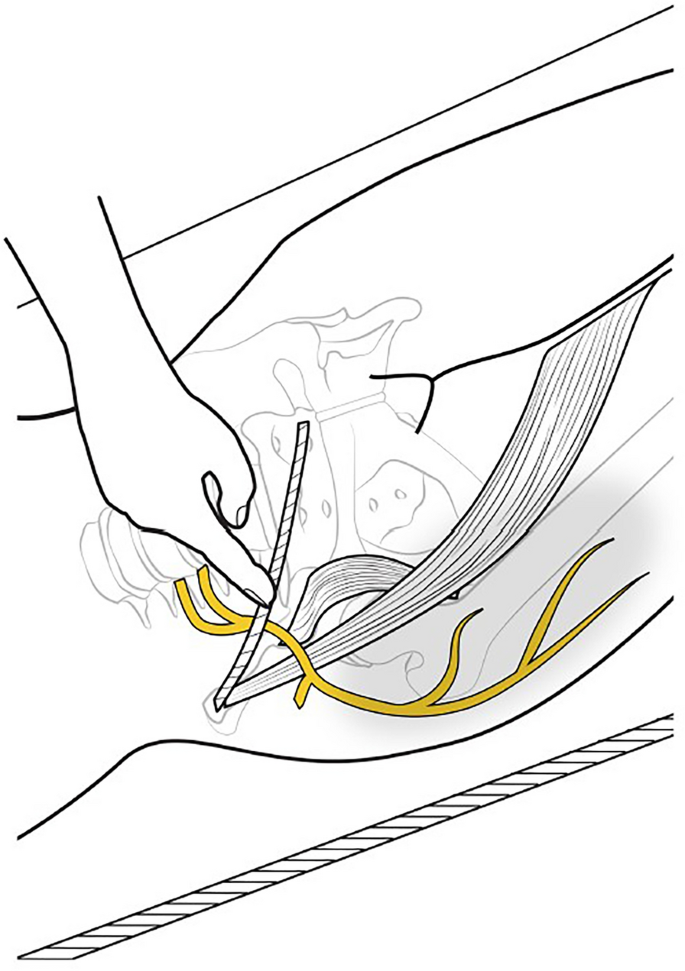
The main two surgical procedures performed in patients with MP are neurolysis and neurectomy. A retrospective cohort study of 16 patients reported complete pain relief in 75% of patients who underwent primary neurectomy compared to 60% of those who underwent primary neurolysis.69 A higher success rate in patients undergoing neurectomy was also reported in a prospective cohort study on 22 patients.70 In this study, complete or almost complete recovery was reported in 93.3% of neurectomy cases while only 37.5% of neurolysis cases achieved the same result.70 In a study evaluating 14 patients after neurectomy or neurolysis, all patients treated with neurectomy had complete pain relief without recurrence while those who were treated with neurolysis had recurrence of symptoms within 9 months.71 Symptom recurrence was not observed 19 months postoperatively in patients treated with microsurgical deep decompression of the LFCN.72
Neurolysis was highly favored by one retrospective study which reported complete or partial improvement in 78% of patients treated with neurolysis while only 35.7% of patients treated with neurectomy reported improvement.73 This study reserved neurectomy for patients who had failed neurolysis in the past, had neuroma formation, or had extensive deformation of the nerve.73 The higher complexity in these cases may have played a role in the lower success rate seen with neurectomy.74 While the majority of available studies seem to favor neurectomy as a treatment, the evidence is still insufficient to firmly recommend one treatment over the other.
A recent meta-analysis compared outcomes in patients treated with injection, neurolysis, or neurectomy.75 The study found the incidence of complete pain relief was significantly higher after neurectomy, followed by neurolysis and then injection.75 Importantly, no statistically significant differences were found regarding incidence of complications between the three modalities.75 Although this study suggests neurectomy is the most effective procedure, a different meta-analysis compared the outcomes of US-guided injections and surgery and found no statistically significant difference between the two modalities.76 The conflicting results in these studies highlight how the evidence available does not point to a single approach that’s universally effective for all patients with MP. Given the lack of consensus over the most effective treatment modality, patient preference should be a key consideration when selecting a treatment.
Prognosis
Complications of MP can include permanent, debilitating anterolateral thigh nerve damage that can limit functional capacity and mobility due to pain. In many cases, MP can be improved with conservative management. For patients with severe refractory symptoms that are not improved with the aforementioned treatments, conditions can be improved with surgical nerve decompression, although success rates can vary. Cases caused by pregnancy typically resolve after delivery.
Conclusion
MP is a sensory mononeuropathy of the anterolateral thigh, that is characterized by paresthesias, numbness, altered sensorium and burning pain. MP has been associated with numerous risk factors and inciting triggers, many of which are modifiable, that can contribute to the development of these debilitating symptoms. MP is generally diagnosed by clinical history and is often a diagnosis of exclusion. When diagnosis remains a challenge, diagnostic modalities such as ultrasound, MRI, electromyography, and nerve conduction studies have been utilized as an adjunct. MP can sometimes be misdiagnosed with other pathologic processes such femoral and lumbar neuropathies, trochanteric bursitis, herpes zoster, and osteoarthritis of the hip.
The prognosis of MP is generally good as most cases resolve spontaneously or respond to conservative management (lifestyle modification, medications, physical therapy, and injections). When surgical intervention is warranted, outcomes are generally favorable with most patients experiencing complete resolution of their symptoms. Despite the inherent risks associated with undergoing LFCN neurolysis and decompression, complications are low, making this a favorable option for those who have failed conventional treatment. While there are currently several effective means for management of MP, further efforts should be aimed at identifying individuals at high risk of developing MP and addressing any modifiable risk factors. Further studies are required to better evaluate the optimal treatment and dosing for the management of MP.
Author Contributions
All authors were involved in the writing and editing of the manuscript.
Disclosures
There are no conflict of interests with the authors.
References
- Meralgia Paresthetica, The Elusive Diagnosis: Clinical Experience With 14 Adult Patients. Ivins Gregory K. Aug;2000 Annals of Surgery. 232(2):281–286. doi: 10.1097/00000658-200008000-00019. doi: 10.1097/00000658-200008000-00019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MERALGIA PARESTHETIGA A REVIEW OF 67 PATIENTS. Kitchen CHARLES, Simpson JOHN. Dec;1972 Acta Neurologica Scandinavica. 48(5):547–555. doi: 10.1111/j.1600-0404.1972.tb07575.x. doi: 10.1111/j.1600-0404.1972.tb07575.x. [DOI] [PubMed] [Google Scholar]
- Clinical and Electrophysiological Characteristics of Meralgia Paresthetica. Choi M.H., Park H., Eom Y.I., Joo I.S. 2013Korean Journal of Community Nutrition. 15:48–52. [Google Scholar]
- MERALGIA PARESTHETICA: A REVIEW OF THE LITERATURE. Cheatham S.W., Kolber M.J., Salamh P.A. 2013Int J Sports Phys Ther. 8:883. [PMC free article] [PubMed] [Google Scholar]
- Review Article RAD 547. Glasović D.M., Šlaus N., Šitum M., Pećina M. 2021Medical Sciences. 547:54–55. [Google Scholar]
- Treatment for meralgia paraesthetica. Khalil N., Nicotra A., Rakowicz W. 2012Cochrane Database Syst Rev. 2012 doi: 10.1002/14651858.CD004159.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Review Article RAD 547. Glasović D.M., Šlaus N., Šitum M., Pećina M. 2021Medical Sciences. 547:54–55. [Google Scholar]
- Yoshida Shinsuke, Oya Soichi, Matsui Toru. Journal of Clinical Neuroscience. Vol. 89. Elsevier BV; Risk factors of meralgia paresthetica after prone position surgery: Possible influence of operating position, laminectomy level, and preoperative thoracic kyphosis; pp. 292–296. [DOI] [PubMed] [Google Scholar]
- Meralgia Paresthetica. Patijn Jacob, Mekhail Nagy, Hayek Salim, Lataster Arno, van Kleef Maarten, Van Zundert Jan. Mar 25;2011 Pain Practice. 11(3):302–308. doi: 10.1111/j.1533-2500.2011.00458.x. doi: 10.1111/j.1533-2500.2011.00458.x. [DOI] [PubMed] [Google Scholar]
- Harney Donal, Patijn Jacob. Pain Medicine. 8. Vol. 8. Oxford University Press (OUP); Meralgia paresthetica: Diagnosis and management strategies; pp. 669–677. [DOI] [PubMed] [Google Scholar]
- MERALGIA PARESTHETICA: A REVIEW OF THE LITERATURE. Cheatham S.W., Kolber M.J., Salamh P.A. 2013Int J Sports Phys Ther. 8:883. [PMC free article] [PubMed] [Google Scholar]
- Shi Xiaochen, Liu Feifei, Liu Fang, Chen Zheng, Zhu Jiaan. Quantitative Imaging in Medicine and Surgery. 10. Vol. 11. AME Publishing Company; Sonographic features of the lateral femoral cutaneous nerve in meralgia paresthetica; pp. 4269–4274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powell G. M., Baffour F. I., Erie A. J., Puffer R. C., Spinner R. J., Glazebrook K. N. Skeletal Radiology. 7. Vol. 49. Springer Science and Business Media LLC; Sonographic evaluation of the lateral femoral cutaneous nerve in meralgia paresthetica; pp. 1135–1140. [DOI] [PubMed] [Google Scholar]
- Meralgia paresthetica: Now showing on 3T magnetic resonance neurography. Ally Raihaan M., Velleman Mark D., Suleman Farhana E. Aug 21;2019 South African Journal of Radiology. 23(1) doi: 10.4102/sajr.v23i1.1745. doi: 10.4102/sajr.v23i1.1745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meralgia paraesthetica following lumbar spine surgery: A study in 110 consecutive surgically treated cases. Gupta A., Muzumdar D., Ramani P.S. 2004Neurol India. 52:66. [PubMed] [Google Scholar]
- Meralgia paraesthetica in a child. Massey E. Wayne, Pellock John M. Aug;1978 The Journal of Pediatrics. 93(2):325–326. doi: 10.1016/s0022-3476(78)80566-6. doi: 10.1016/s0022-3476(78)80566-6. [DOI] [PubMed] [Google Scholar]
- Risk factor analysis for meralgia paresthetica: A hospital-based study in Taiwan. Weng Wei-Chieh, Wei Yi-Chia, Huang Wen-Yi, Chien Yu-Yi, Peng Tsung-I, Wu Chia-Lun. Sep;2017 Journal of Clinical Neuroscience. 43:192–195. doi: 10.1016/j.jocn.2017.04.024. doi: 10.1016/j.jocn.2017.04.024. [DOI] [PubMed] [Google Scholar]
- Meralgia paresthetica. Parisi T. J., Mandrekar J., Dyck P. J. B., Klein C. J. Oct 5;2011 Neurology. 77(16):1538–1542. doi: 10.1212/wnl.0b013e318233b356. doi: 10.1212/wnl.0b013e318233b356. [DOI] [PMC free article] [PubMed] [Google Scholar]