Re:Emerging Roles for Riboflavin in Functional Rescue of Mitochondrial beta-oxidation Flavoenzymes
작성자문형철작성시간20.04.22조회수518 목록 댓글 0beyond reason
https://www.peirsoncenter.com/uploads/6/0/5/5/6055321/10.2174_092986710793205462.pdf
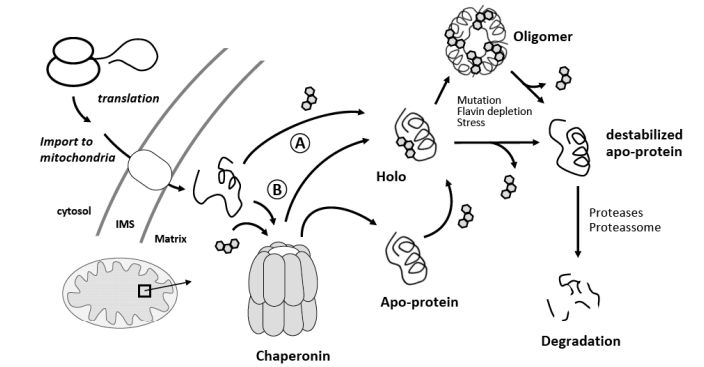
Cartoon representing different scenarios for pathways through which FAD may be inserted into proteins conferring structural and functional rescue. After translation and import into the mitochondria the apoprotein form may become flavinylated via a chaperonin-independent (A) or chaperonin-dependent (B) pathway. In both cases, steps involving FAD insertion may eventually be mediated by FAD-chaperone proteins. The chaperonin-dependent pathway may involve folding of the apo monomer which then gets flavinylated upon relase or immediately after release. Oligomerization into the functional forms (tetramers or dimers) is made starting from the holo-protein form. Upon an adverse cellular or patho-physiological condition such as a genetic mutation, stress (thermal, oxidative or other) or riboflavin and flavin depletion, cofactor lability may be enhanced thus resulting in an equilibrium of populations in which there is a significant amount of the enzyme in the apo-form. The latter is known to be more conformationally destabilized and susceptible to degradation or misfolding, resulting in loss of function. In some cases, restoring the intra-mitochondrial flavin levels as a result of riboflavin supplementation, results in an increase of the activity of the affected proteins. See text for details and key references.
Abstract
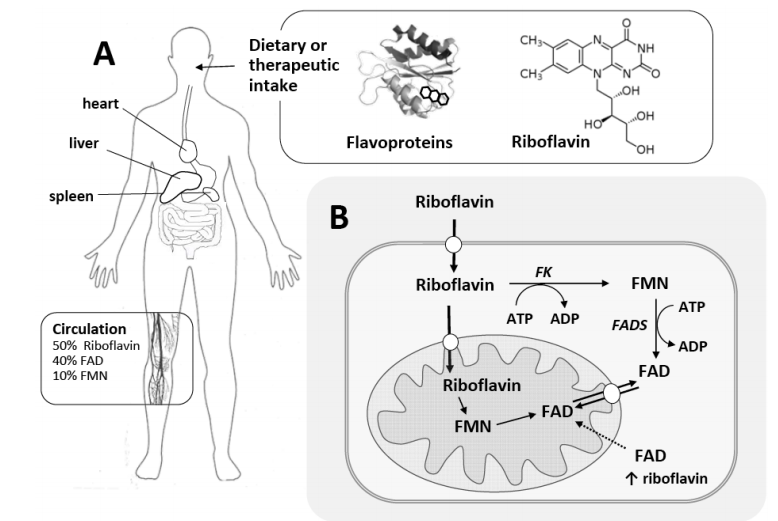
Riboflavin, commonly known as vitamin B2, is the precursor of flavin cofactors. It is present in our typical diet, and inside the cells it is metabolized to FMN and FAD. As a result of their rather unique and flexible chemical properties these flavins are among the most important redox cofactors present in a large series of different enzymes. A problem in riboflavin metabolism or a low intake of this vitamin will have consequences on the level of FAD and FMN in the cell, resulting in disorders associated with riboflavin deficiency.
In a few number of cases, riboflavin deficiency is associated
with impaired oxidative folding, cell damage and impaired heme biosynthesis. More relevant are several studies referring
reduced activity of enzymes such as dehydrogenases involved in oxidative reactions, respiratory complexes and enzymes
from the fatty acid -oxidation pathway. The role of this vitamin in mitochondrial metabolism, and in particular in fatty
acid oxidation, will be discussed in this review. The basic aspects concerning riboflavin and flavin metabolism and deficiency will be addressed, as well as an overview of the role of the different flavoenzymes and flavin chemistry in fatty
acid -oxidation, merging clinical, cellular and biochemical perspectives. A number of recent studies shedding new light
on the cellular processes and biological effects of riboflavin supplementation in metabolic disease will also be overviewed. Overall, a deeper understanding of these emerging roles of riboflavin intake is essential to design better therapies.