beyond reason
| Enzyme | Substrate | ROS |
|---|---|---|
| (1) Acyl-CoA oxidases | ||
| (a) Palimtoyl-CoA oxidase | Long chain fatty acids | H2O2 |
| (b) Pristanoyl-CoA oxidase | Methyl branched chain fatty acids | H2O2 |
| (c) Trihydroxycoprostanoyl-CoA oxidase | Bile acid intermediates | H2O2 |
| (2) Urate oxidase | Uric acid | H2O2 |
| (3) Xanthine oxidase | Xanthine | H2O2, O2·− |
| (4) d-amino acid oxidase | d-Proline | H2O2 |
| (5) Pipecolic acid oxidase | l-pipecolic acid | H2O2 |
| (6) d-aspartate oxidase | d-aspartate, N-methyl-d-aspartate | H2O2 |
| (7) Sarosine oxidase | Sarcosine, pipecolate | H2O2 |
| (8) l-alpha-hydroxy acid oxidase | Glycolate, lactate | H2O2 |
| (9) Poly amine oxidase | N-Acetyl spermine/spermidine | H2O2 |
| (10) Nitric oxide synthase | l-Arginine | ·NO |
| (11) Plant sulfite oxidase [165] | Sulfite | H2O2 |
Enzymes in peroxisomes that degrade ROS
Enzyme Substrate Enzyme is also present in (1) Catalase H2O2 Cytoplasm (e. g., erythrocytes) and nucleus, mitochondria (rat heart only) (2) Glutathione peroxidase H2O2 All cell compartments (3) Mn SOD O2·− Mitochondria (4) Cu, Zn SOD O2·− Cytoplasm (5) Epoxide hydrolase Epoxides ER and cytoplasm (6) Peroxireodoxin 1 H2O2 Cytoplasm, nucleus, mitochondria (7) PMP 20 H2O2 Peroxisomes (8) Plant ascorbate–glutathione cycle H2O2 Peroxisomes, chloroplasts, cytoplasm, root nodule mitochondria (plants only) [27]
| Enzyme | Substrate | Enzyme is also present in |
|---|---|---|
| (1) Catalase | H2O2 | Cytoplasm (e. g., erythrocytes) and nucleus, mitochondria (rat heart only) |
| (2) Glutathione peroxidase | H2O2 | All cell compartments |
| (3) Mn SOD | O2·− | Mitochondria |
| (4) Cu, Zn SOD | O2·− | Cytoplasm |
| (5) Epoxide hydrolase | Epoxides | ER and cytoplasm |
| (6) Peroxireodoxin 1 | H2O2 | Cytoplasm, nucleus, mitochondria |
| (7) PMP 20 | H2O2 | Peroxisomes |
| (8) Plant ascorbate–glutathione cycle | H2O2 | Peroxisomes, chloroplasts, cytoplasm, root nodule mitochondria (plants only) [27] |
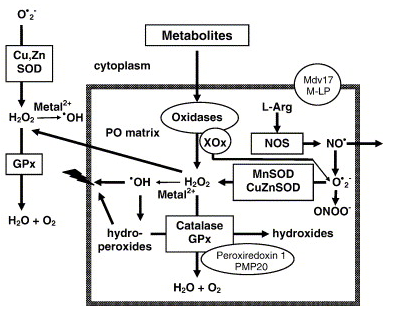
Fig. 2. Schematic overview of peroxisomal enzymes which produce or degrade ROS. H2O2 is mainly produced by several peroxisomal oxidases (e. g., acyl-CoA oxidase which is involved in the β-oxidation of fatty acids). H2O2 is decomposed by catalase and glutathione-peroxidase (GPx) or converted to hydroxyl radicals (·OH). Hydroxyl radicals can damage the peroxisomal membrane by lipid peroxidation of unsaturated fatty acids. Hydroperoxides formed in this process can be decomposed by catalase and glutathione-peroxidase. Superoxide anions (O2·−) generated by peroxisomal oxidases (for example, xanthine oxidase (XOx)) are scavanged by manganese superoxide-dismutase (MnSOD) and by copper-zink superoxide-dismutase (CuZnSOD). Nitric oxide synthase (NOS) catalyses the oxidation of l-arginine (l-Arg) to nitric oxide (NO·). NO· can react with O2·− radicals to form peroxynitrite (ONOO−), a powerful oxidant. H2O2 and NO· can penetrate the peroxisomal membrane and act in cellular signalling. Peroxiredoxin 1 and PMP20 are involved in the degradation of H2O2. Mpv17 and L-MP are implicated in the regulation of peroxisomal ROS metabolism.
ReviewPeroxisomes and oxidative stress
Fig. 2. Schematic overview of peroxisomal enzymes which produce or degrade ROS. H2O2 is mainly produced by several peroxisomal oxidases (e. g., acyl-CoA oxidase which is involved in the β-oxidation of fatty acids). H2O2 is decomposed by catalase and glutathione-peroxidase (GPx) or converted to hydroxyl radicals (·OH). Hydroxyl radicals can damage the peroxisomal membrane by lipid peroxidation of unsaturated fatty acids. Hydroperoxides formed in this process can be decomposed by catalase and glutathione-peroxidase. Superoxide anions (O2·−) generated by peroxisomal oxidases (for example, xanthine oxidase (XOx)) are scavanged by manganese superoxide-dismutase (MnSOD) and by copper-zink superoxide-dismutase (CuZnSOD). Nitric oxide synthase (NOS) catalyses the oxidation of l-arginine (l-Arg) to nitric oxide (NO·). NO· can react with O2·− radicals to form peroxynitrite (ONOO−), a powerful oxidant. H2O2 and NO· can penetrate the peroxisomal membrane and act in cellular signalling. Peroxiredoxin 1 and PMP20 are involved in the degradation of H2O2. Mpv17 and L-MP are implicated in the regulation of peroxisomal ROS metabolism.
Abbreviations
Keywords
1. Oxidative stress and peroxisomes in brief
1.1. The “good” and the “evil” of ROS
Oxidative stress arises from a significant increase in the concentration of reactive oxygen species (ROS) and reactive nitrogen species (RNS), and/or a decrease in their detoxification mechanisms. There are many natural sources of oxidative stress, for example exposure to environmental oxidants, toxins like heavy metals, ionizing and UV irradiation, heat shock, and inflammation [1]. High levels of ROS exert a toxic effect on biomolecules such as DNA, proteins, and lipids (e. g., non-enzymatic lipoperoxidation), thus leading to the accumulation of oxidative damage in diverse cellular locations, to the deregulation of redox-sensitive metabolic and signalling pathways, and to pathological conditions. Much interest in oxidative stress comes from human pathologies, for example ischemia–reperfusion injury, atherosclerosis, hypertension, inflammation, cystic fibrosis, cancer, type-2 diabetes, or neurodegenerative diseases such as Parkinson's or Alzheimer's disease. Furthermore, oxidative stress has been linked to aging [2]. Besides their harmful role in clinical conditions, the importance of ROS (RNS) as mediators in various vital cellular processes and cell signalling pathways became apparent (reviewed in [3], [4]). A typical example is their function in apoptosis.
ROS include radical species (containing free, i. e. unpaired, electrons), such as the superoxide anion (O2·−), which is formed through one-electron reduction of O2 (O2 + e− → O2·−). Hydrogen peroxide (H2O2) is also ascribed to ROS, although it has no unpaired electrons, and thus is not a radical. It can, for example, be formed by the dismutation reaction of O2·− (catalyzed by superoxide dismutases) via the hydroperoxyl radical (O2·− + H+ → HO2·−; 2HO2·− → H2O2 + O2). Probably the most highly reactive and toxic form of oxygen, the hydroxyl radical (·OH), can be formed by the metal ion (e. g., iron or copper)-catalyzed decomposition of H2O2 (H2O2 + O2·− → O2 + OH− + ·OH). Similarly, RNS include radical species such as primary nitric oxide (·NO). ·NO and H2O2 are membrane permeable, diffusible molecules, which are less-reactive and longer-lived than ·OH, thus being best suited for intra- and even intercellular signalling. In particular H2O2, which is not harmful until converted to more reactive ROS, acts on cellular thiol-disulfide redox buffer systems such as glutathione, thioredoxin and peroxiredoxin (for review see [5], [6]).
1.2. Peroxisomes and ROS
Oxygen is consumed in various metabolic reactions in different cellular locations, with mitochondria, the ER, and peroxisomes being the major sites (Fig. 1A). De Duve and Baudhuin [7] first described a respiratory pathway in peroxisomes, in which electrons removed from various metabolites reduce O2 to H2O2, which is further reduced to H2O. The respiratory pathway in peroxisomes is not coupled to oxidative phosphorylation, and does not lead to the production of ATP. Free energy is released in the form of heat.
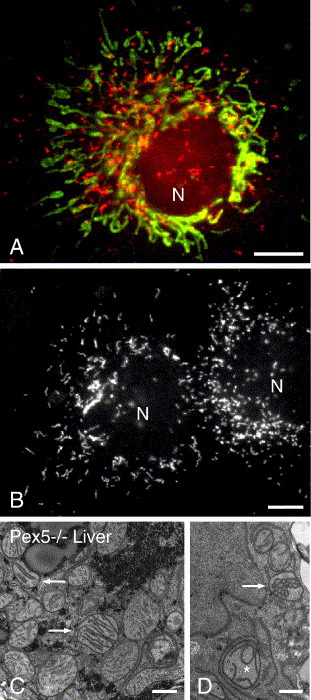
Fig. 1. (A) Fluorescence microscopy of peroxisomes and mitochondria, two major subcellular organelles involved in the metabolism of ROS, in COS-7 cells, a green monkey kidney cell line. Cells were stained with antibodies to peroxisomal catalase (red) and to mitochondrial MnSOD (green). (B) Mammalian cells can exhibit different peroxisomal morphologies under normal culture conditions. Peroxisomes in COS-7 cells were visualized by immunofluorescence using specific antibodies directed to PMP70, a peroxisomal membrane protein. Note the spherical shape of peroxisomes in the cell at the left, in contrast to their elongated, tubular morphology in the cell at right. The formation of elongated peroxisomes is induced after UV irradiation or exposure to H2O2 (see Section 4.2). (C, D) Electron micrographs of altered mitochondria in hepatocytes of PEX5−/− mice (reprinted from [91] with permission from Springer-Verlag) (C) Pleomorphic mitochondria with altered cristae. Some of the round, large mitochondria exhibit stacks of parallel cristae (arrows). (D) Mitochondria with circular cristae (asterisks) or mitochondrial ghosts (arrow). Scale bars, 10 μm (A, B), 500 nm (C, D).
The high peroxisomal consumption of O2, the demonstration of the production of H2O2, O2·−, ·OH, and recently of ·NO in peroxisomes [7], [8], [9], [10], as well as the discovery of several ROS-metabolizing enzymes in peroxisomes (see Tables 1 and 2) has supported the notion that these ubiquitous organelles play a key role in both the production and scavening of ROS in the cell, in particular H2O2 (Fig. 2).
Table 1. Enzymes in peroxisomes that generate ROS
| Enzyme | Substrate | ROS |
|---|---|---|
| (1) Acyl-CoA oxidases | ||
| (a) Palimtoyl-CoA oxidase | Long chain fatty acids | H2O2 |
| (b) Pristanoyl-CoA oxidase | Methyl branched chain fatty acids | H2O2 |
| (c) Trihydroxycoprostanoyl-CoA oxidase | Bile acid intermediates | H2O2 |
| (2) Urate oxidase | Uric acid | H2O2 |
| (3) Xanthine oxidase | Xanthine | H2O2, O2·− |
| (4) d-amino acid oxidase | d-Proline | H2O2 |
| (5) Pipecolic acid oxidase | l-pipecolic acid | H2O2 |
| (6) d-aspartate oxidase | d-aspartate, N-methyl-d-aspartate | H2O2 |
| (7) Sarosine oxidase | Sarcosine, pipecolate | H2O2 |
| (8) l-alpha-hydroxy acid oxidase | Glycolate, lactate | H2O2 |
| (9) Poly amine oxidase | N-Acetyl spermine/spermidine | H2O2 |
| (10) Nitric oxide synthase | l-Arginine | ·NO |
| (11) Plant sulfite oxidase [165] | Sulfite | H2O2 |
Table 2. Enzymes in peroxisomes that degrade ROS
| Enzyme | Substrate | Enzyme is also present in |
|---|---|---|
| (1) Catalase | H2O2 | Cytoplasm (e. g., erythrocytes) and nucleus, mitochondria (rat heart only) |
| (2) Glutathione peroxidase | H2O2 | All cell compartments |
| (3) Mn SOD | O2·− | Mitochondria |
| (4) Cu, Zn SOD | O2·− | Cytoplasm |
| (5) Epoxide hydrolase | Epoxides | ER and cytoplasm |
| (6) Peroxireodoxin 1 | H2O2 | Cytoplasm, nucleus, mitochondria |
| (7) PMP 20 | H2O2 | Peroxisomes |
| (8) Plant ascorbate–glutathione cycle | H2O2 | Peroxisomes, chloroplasts, cytoplasm, root nodule mitochondria (plants only) [27] |
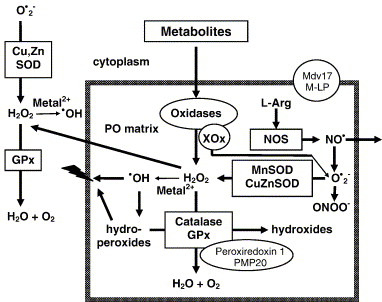
Fig. 2. Schematic overview of peroxisomal enzymes which produce or degrade ROS. H2O2 is mainly produced by several peroxisomal oxidases (e. g., acyl-CoA oxidase which is involved in the β-oxidation of fatty acids). H2O2 is decomposed by catalase and glutathione-peroxidase (GPx) or converted to hydroxyl radicals (·OH). Hydroxyl radicals can damage the peroxisomal membrane by lipid peroxidation of unsaturated fatty acids. Hydroperoxides formed in this process can be decomposed by catalase and glutathione-peroxidase. Superoxide anions (O2·−) generated by peroxisomal oxidases (for example, xanthine oxidase (XOx)) are scavanged by manganese superoxide-dismutase (MnSOD) and by copper-zink superoxide-dismutase (CuZnSOD). Nitric oxide synthase (NOS) catalyses the oxidation of l-arginine (l-Arg) to nitric oxide (NO·). NO· can react with O2·− radicals to form peroxynitrite (ONOO−), a powerful oxidant. H2O2 and NO· can penetrate the peroxisomal membrane and act in cellular signalling. Peroxiredoxin 1 and PMP20 are involved in the degradation of H2O2. Mpv17 and L-MP are implicated in the regulation of peroxisomal ROS metabolism.
Initially, it was assumed that the main function of peroxisomes was the decomposition of H2O2 generated by different peroxisomal oxidases (mainly flavoproteins) via catalase, the classical peroxisomal marker enzyme. However, it is now clear that peroxisomes are involved in a variety of important cellular functions in almost all eukaryotic cells (for details see articles about peroxisomal metabolism, this issue). The main metabolic processes contributing to the generation of H2O2 in peroxisomes are the β-oxidation of fatty acids, the enzymatic reactions of the flavin oxidases, the disproportionation of superoxide radicals, and in plant peroxisomes, the photorespiratory glycolate oxidase reaction. It has been estimated that about 35% of all H2O2 formed in rat liver derives from peroxisomal oxidases [11]. To degrade the ROS, which are produced due to their metabolic activity, and to maintain the equilibrium between production and scavenging of ROS, peroxisomes harbour several powerful defense mechanisms and antioxidant enzymes in addition to catalase (Table 2, Fig. 2).
An interesting feature of peroxisomes is their ability to proliferate and multiply, or be degraded in response to nutritional and environmental stimuli. In rodents, for example, the number and size of peroxisomes as well as the expression of peroxisomal β-oxidation enzymes are highly increased when activators of the peroxisome proliferator activated receptors (PPARs) like fibrates or free fatty acids are applied [12]. Such conditions are considered to generate peroxisome-induced oxidative stress, which may overwhelm the antioxidant capacity and lead to cancer. Furthermore, transition metal ions like iron and copper are abundant in peroxisomes mainly in a complexed form. Under certain conditions, these metal ions can be released (e. g., by xenobiotics) and catalyze the formation of ·OH in the Fenton reaction (Fe2+ + H2O2 → Fe3+ + OH− + ·OH), thus leading to lipid peroxidation, damage of the peroxisomal membrane and loss of peroxisomal functions [13], [14].
It is obvious that due to their oxidative metabolism, peroxisomes are considered a source of oxidative stress. However, peroxisomes can also respond to oxidative stress and ROS, which have been generated in other intra- or extracellular locations, most likely to protect the cell against an oxidative damage. Furthermore, certain peroxisome-generated ROS may act as mediators in intracellular signalling [15]. In this review, we will address recent findings on the production and decomposition of ROS in peroxisomes, the response of peroxisomes to oxidative stress, peroxisome proliferation and the induction of ROS, as well as a role of ROS in clinical conditions, peroxisomal disease, and mouse models. A function of peroxisomes in aging is addressed elsewhere [2]. We will mainly focus on animal peroxisomes, but findings in plant cells, where some of the peroxisomal functions in ROS metabolism were first discovered, are also discussed.
2. Peroxisomal enzymes that generate ROS
The discovery of the co-localization of several H2O2 producing oxidases together with catalase in peroxisomes was the first indication suggesting the participation of peroxisomes in the metabolism of oxygen metabolites. It also led de Duve to propose the term “peroxisome” for the designation of that organelle [7]. In mammalian peroxisomes the major sources of H2O2 production are the oxidases that transfer hydrogen from their respective substrates to molecular oxygen (Fig. 2). In rat liver, peroxisomes produce about 35% of all H2O2 [11] which accounts for about 20% of total oxygen consumption [16]. The occurrence of oxidases varies markedly in peroxisomes of different cells, tissues and organs contributing to the heterogeneity of peroxisomes. Table 1 shows a compilation of enzymes in peroxisomes producing H2O2 and O2·−. In addition, the enzyme NO-synthase is included which was first discovered in plant peroxisomes [17] with subsequent detection in animal peroxisomes [10].
2.1. Acyl-CoA oxidases
The β-oxidation of fatty acids is the most important metabolic process in peroxisomes contributing to the formation of H2O2 [18]. Although the peroxisomal lipid substrates have little significance as sources of metabolic fuel for the generation of energy, they serve important physiological functions. The different classes of lipids are metabolized by separate enzymes with distinct substrate specificities [19]. Three separate oxidases have been identified in rat liver for the degradation of (a) very long straight chain fatty acids and prostaglandins; (b) 2-methyl branched-chain fatty acids; and (c) bile acid intermediates. An important aspect of peroxisomal lipid metabolism is its inducibility by a variety of endogenous and exogenous substrates via the nuclear transcription factor PPARα which can in turn induce tumor formation [20].
2.2. Urate oxidase
This enzyme, which in most mammalian species is localized in the electron dense crystalline cores of hepatic peroxisomes [21], [22] catalyses the oxidation of urate yielding H2O2, allantoin, and CO2, which is the final step in the metabolic degradation of the purine ring in many mammalian species. In humans and primates the enzyme has lost its activity because of multiple mutations of the urate oxidase gene [23]. Because of the antioxidant properties of urate it has been postulated that the loss of the enzyme in humans may have a protective effect against conditions associated with ROS such as cancer and aging [24].
2.3. Xanthine oxidase
There are two functionally distinct forms of this enzyme: the NAD+ dependent ‘D’ or the dehydrogenase form, and the O2-dependent ‘O’ or the oxidase form. Under a variety of conditions such as proteolysis, heating and ischemia the D form is transformed to the O form [25] which can generate the toxic superoxide radical as well as H2O2. The oxidase activity was demonstrated in rat liver peroxisomes by the cerium technique [26]. In addition, the enzyme activity was found in the cytoplasm of hepatic endothelial cells as well as in plant peroxisomes [27].
2.4. d-amino acid oxidase
This flavoprotein exhibits a mosaic pattern in rat liver with strongly and weakly stained peroxisomes in hepatocytes [28]. The intensity of staining is stronger in periportal than in perivenous regions of the liver lobule. In addition, the enzyme is present in rat brain and kidney [29]. In kidney proximal tubules the enzyme occupies mostly the central part of the matrix of peroxisomes forming a distinct subcompartment [30], [31]. In marine mussel, Mytilus galloprovincialis, d-amino acid oxidase is present in peroxisomes of the digestive gland cells and is markedly induced after exposure to pollutants such as lubricant oil and phthalates [32]. In addition, the enzyme is found in digestive glands of terrestrial gastropods next to d-aspartate oxidase [33].
2.5. d-aspartate oxidase
This is another flavoenzyme that specifically oxidizes the d-amino acids with two carboxylic groups such as d-aspartate, d-glutamate and N-methyl-d-aspartate with important neuroregulatory functions in the central nervous system. The enzyme has been localized in rat and human brain with strong activity in the hypothalamic paraventricular nucleus [34] as well as in rat liver and kidney peroxisomes [35]. The rat liver enzyme has an inherent targeting signal (SKL) for import into peroxisomes [34].
2.6. Pipecolic acid oxidase
This enzyme catalyses the oxidation of l-pipecolic acid to piperidine-6-carboxylic acid generating H2O2 and was localized to renal and hepatic peroxisomes [36]. The cloning of both human and rat proteins revealed that the enzyme is related to the bacterial sarcosine oxidase rather than to d-amino acid oxidase [37]. In peroxisomal deficiency disorders the level of pipecolic acid is elevated, although this does not seem to be a specific diagnostic feature [38].
2.7. Sarcosine oxidase
This flavoprotein was cloned from rabbit kidney and was noted to oxidize not only sarcosine but also l-proline as well as l-pipecolic acid [39]. By cytochemistry the enzyme was localized to liver and kidney peroxisomes of several mammalian species including mouse and hamster [40]. In plants (Arabidopsis) the enzyme metabolizes both sarcosine and pipecolate with preferential utilization of the latter as an endogenous substrate [41].
2.8. l-alpha-hydroxy acid oxidase
There are two isoforms of this enzyme with the A-isoform oxidizing preferentially glycolate and being most prominent in liver, and the B-isoform occurring in kidney and utilizing dl-hydroxybutyrate as substrate [42], [43]. In kidney the enzyme is localized to marginal plates which are crystalline plate-like structures just below the peroxisomal membranes [44]. By genomic analysis three distinct human genes were identified with significant sequence similarity to plant glycolate oxidase [45]. Hydoxy acid oxidase 1 is a liver specific enzyme with a modified peroxisomal targeting signal consisting of SKI [46]. It oxidizes glycolate to glyoxylate and its mRNA is significantly reduced in rats subjected to oxidative stress induced by ischemia and reperfusion or by glutathione depletion [47].
2.9. Polyamine oxidase
The mammalian enzyme utilizes both spermine and spermidine as substrates [48] and exerts an important influence on the metabolism of polyamines which are significantly increased in tumors [49]. The murine and bovine enzymes were cloned and found to contain type 1 peroxisomal targeting signals at their C terminals [50].
2.10. Nitric oxide synthase
This enzyme generates nitric oxide (·NO), a molecule with important signalling functions in animals and plants. In plant cells the enzyme was localized to peroxisomes [17] and seems to be involved in various important physiological processes such as development, growth, senescence and innate immunity [51], [52], [53]. In animal cells, an inducible form of the enzyme (iNOS) is expressed under pathological conditions which can cause severe tissue injury. Stolz et al. [10] reported the detection of iNOS in peroxisomes of rat hepatocytes after exposure to tumor necrosis factor-alpha. Because of the concomitant reduction of catalase activity the possibility of the formation of the highly toxic peroxynitrite radical was suggested. Recently, however, it was noted that the peroxisomal pool of iNOS consists mostly of the inactive monomeric form, suggesting that the peroxisomal sequestration could prevent the formation of the more toxic dimeric form in the cytoplasm [54]. The exact mechanism of transport of iNOS into peroxisomes is not known, although variations of both PTS1 and PTS2 have been described [10].
3. Peroxisomal enzymes that scavange ROS
Table 2 summarizes the enzymes in peroxisomes that decompose ROS.
3.1. Catalase
This classical marker enzyme of peroxisomes metabolizes both H2O2 (catalatic function) and a variety of substrates such as ethanol, methanol, phenol and nitrites by peroxidatic activity [55]. In most mammalian cells it is targeted to peroxisomes via a modified PTS1 [56]. Catalase has an important protective function against the toxic effects of peroxides generated in peroxisomes and removes them with high efficiency [57] (Fig. 1A). Indeed, the inhibition of catalase in rat liver suppresses markedly the peroxisomal lipid β-oxidation activity [58] and in plant cells inhibits the glyoxylate cycle enzymes isocitrate lyase and malate synthase [59]. Whereas the lack of peroxisomal catalase in C. elegans causes developmental abnormalities and a progeric phenotype [60], its overexpression in transgenic mice induces an extension of the average lifespan of animals [61], [2]. The catalase activity and peroxisomes are significantly reduced in tumors of the liver [62] and other organs [63], as well as in a variety of pathological processes such as liver allograft rejection [64], and after ischemia–reperfusion injury [65].
3.2. Glutathione peroxidase
This is primarily a cytosolic enzyme which was also detected in peroxisomes in rat liver [66]. It catalyses the destruction of H2O2 with concomitant conversion of reduced glutathione (GSH) to glutathione disulfide (GSSG). It can also oxidize peroxide substrates such as cumene hydroperoxide.
3.3. Manganese superoxide dismutase (MnSOD)
In most eukaryotic cells this enzyme is primarily localized in mitochondria (Fig. 1A) but in addition it was reported in rat liver peroxisomal membranes [67]. In higher plants the enzyme was first localized in peroxisomes [68]. More recently, the involvement of MnSOD in plant senescence was reported, and it was found that the mitochondrial and peroxisomal enzyme are differentially regulated [69]. A novel peroxisomal membrane protein (M-LP) has been reported to up-regulate the expression of MnSOD in COS-7 cells [70].
3.4. Copper Zinc superoxide dismutase (Cu, Zn SOD)
The localization of this enzyme in peroxisomes was first detected by immuno-electron microscopy [71] and was subsequently confirmed by cell fractionation [72]. The expression of the gene can be induced by ciprofibrate as well as by arachidonic acid and is mediated by a peroxisome proliferator response element (PPRE) [73].
3.5. Epoxide hydrolase
This enzyme is present in peroxisomes and cytoplasm in addition to the ER [74]. The rat liver enzyme contains a modified PTS1 consisting of Ser–Lys–Ile [75]. The enzyme in peroxisomes can catabolize fatty acids with an oxirane ring which is formed in sterol metabolism and is inducible by ciprofibrate [76].
3.6. Peroxiredoxin I
This antoxidant protein has thioredoxin-dependent peroxidase activity with strong affinity for the pro-oxidant heme [77]. The presence of peroxiredoxin 1 in rat liver peroxisomes, in addition to cytoplasm, mitochondria and nucleus was reported recently [78]. The peroxiredoxins play an important role in cellular protection against oxidative stress and in cell signalling [79]. The expression of thioredoxins is induced by the ligand activation of PPARα while overexpression of thioredoxins inhibits the PPARα activity suggesting an auto-regulatory mechanism [80].
3.7. Peroxisomal membrane protein 20 (PMP 20)
In addition to the above mentioned enzymes, a human and mouse protein with peroxisomal targeting signal SQL, which is similar to the PTS1, has been isolated exhibiting thiol-specific antioxidant activity [81]. This PMP 20 is capable to remove H2O2 by its thiol-peroxidase activity and has been suggested to protect peroxisomal proteins against oxidative stress.
4. The response of peroxisomes to oxidative stress
4.1. Alterations of ROS-metabolizing enzymes
An elevation of the environmental oxygen concentration has been shown to induce a moderate increase in the volume density of peroxisomes and their enzymes involved in the scavenging of ROS. Chinese hamster ovary (CHO) cells exposed to 99% O2 exhibit a two-fold increase of the volume fraction of peroxisomes and two–four fold elevations of catalase, glutathione peroxidase as well as manganese- and copper zinc-SODs [82]. On the other hand, more pronounced proliferation of peroxisomes and many-fold induction of H2O2-generating fatty acyl-CoA oxidase activity is observed after treatment with peroxisome proliferating chemicals [12], [83] (see Section 5). Whereas the mechanism of induction of oxidative enzymes in peroxisomes via the nuclear transcription factor PPARα and its co-activators [20] is now better understood, their response to ROS is much more complex requiring further research. The regulation of anti-oxidant enzymes is closely related to apoptotic signalling because the oxidative state can determine the cell fate to enter either the cell cycle or to undergo apoptosis [84]. Indeed, elevation of anti-oxidative enzymes can suppress apoptosis but this can also promote carcinogenesis [85]. On the other hand, low levels of anti-oxidant enzymes such as catalase, glutathione peroxidase and MnSOD are a typical hallmark of malignant cells [86], [62]. Thus, it seems that the regulation of anti-oxidant enzymes is a more complex process in comparison to that of oxidative enzymes which are mainly regulated by PPARα. One of the forkhead transcription factors that protects quiescent cells from oxidative stress is FOXO3a (also known as FKHR-L1) which induces MnSOD [87]. Recently, it was reported that the same transcription factor also regulates the expression of the peroxisomal sterol carrier protein 2 (SCP2) which is supposed to protect fatty acids from lipid peroxidation [88]. Furthermore, the peroxisomal membrane proteins Mpv17 and M-LP (Mpv17-like protein), have been implicated in regulating the expression of antioxidant enzymes in mammals [9], [70], [89] (see Section 6).
4.2. Peroxisome elongation and protection against ROS
In cultured cells, oxidative stress has been shown to induce morphological changes of the peroxisomal compartment. In mammalian and plant cells a pronounced elongation of peroxisomes is observed after UV irradiation, direct exposure to H2O2, or even during live cell imaging of GFP-PTS1 labeled peroxisomes by regular fluorescent light [90], [91], [92], [93]. Antioxidant treatment blocked the elongation of peroxisomes, thus supporting the role of ROS in UV-mediated peroxisomal growth [90]. Peroxisomes are highly dynamic organelles with large plasticity, and spherical as well as elongated-tubular or tubulo-reticular peroxisomal structures have been frequently described in ultrastructural and light microscopic studies, even in live cells (Fig. 1B). Evidence has been presented in the last years that peroxisome elongation appears to be an important prerequisite of peroxisome division, and that tubulation and fission of elongated peroxisomes represent processes of peroxisomal growth and division, which contribute to peroxisome proliferation. The molecular machinery involved in these processes is just beginning to emerge. Recently, a role for the peroxisomal membrane protein Pex11p, the dynamin-like protein DLP1/Drp1, and Fis1, a putative membrane receptor/adaptor for DLP1/Drp1 at the peroxisomal membrane has been demonstrated [94], [95], [96], [97], [98], [99]. Elongated/tubular peroxisomes can also be induced by partial hepatectomy [100], stimulation of cultured cells with defined growth factors or polyunsaturated fatty acids (PUFAs), particularly arachidonic acid [90], microtubule depolymerization [101], Pex11p overexpression [92], [102], or by inhibition of DLP1/Drp1 or hFis1 function [94], [95], [96], [97], [98]. Whereas the latter manipulations appear to act directly on the peroxisomal membrane, the other conditions require intracellular signalling to induce peroxisome elongation/growth [103]. Arachidonic acid, for example, is a substrate for peroxisomal β-oxidation, but has also been shown to stimulate ROS production by cultured cells [104]. In plant cells (but also in mammals) it is suggested that diverse types of stress (e. g., wounding, pathogen attack, drought, osmotic stress, excess light) that generate H2O2 as a signalling molecule result in peroxisome proliferation via the up-regulation of PEX genes required for peroxisome biogenesis and import of proteins [27], [105], [106]. An elevation of peroxisomal SOD and glutathione peroxidase has been reported after induction of oxidative stress by endotoxin treatment of rat liver [107]. In plant cells, ROS and NO that are generated and released to the cytosol by peroxisomes, are suggested to target the nuclear proteins DET1 and COP1, which are involved in photomorphogenesis [108]. They negatively regulate the transcription of many light responsive genes involved in peroxisomal functions, in light signalling, photosynthesis and in stress response [108], [109]. However, most of the UV- or ROS-mediated signalling pathways and the gene responses leading to peroxisome elongation/proliferation are still largely unknown. Although information on the exact functions of elongated/tubular or complex peroxisomal structures is scarce, the evidence of ROS-mediated peroxisome elongation/proliferation and induction of peroxisomal genes supports an involvement in cellular rescue from ROS.
In Arabidopsis cells, the active sites for the antioxidant enzymes ascorbate peroxidase and monodehydroascorbate oxidase, are located on opposite sides of the peroxisomal membrane, thus requiring diffusion of ROS across the peroxisomal membrane for interaction [110]. The increased membrane to matrix surface area ratio of elongated peroxisomes may increase the accessibility of H2O2 or ascorbate free radical to these enzymes [92]. Peroxisome elongation induced by ROS must therefore not necessarily be accompanied by an elevation of the antioxidative peroxisomal enzymes to allow efficient decomposition of ROS. Furthermore, a CHO cell mutant with an inactivating point mutation in DLP1/Drp1, which exhibited permanently elongated peroxisomes and mitochondria, showed resistance to oxidative stress and apoptotic stimulation [98]. On the other hand, peroxisome elongation can lead to an increase in the number of peroxisomes/proliferation by fission. Along the same line, Wy-14463 induced peroxisome proliferation (accompanied by an increase in catalase activity) has been shown to prevent ROS production in hippocampal neurons and to protect from β-amyloid neurodegeneration [111]. Finally, ether phospholipid (plasmalogen) synthesis might be promoted by peroxisomal elongation/proliferation after exposure to UV light or H2O2. Plasmalogens have been implicated in the protection against oxidative stress and in an important anti-oxidative function in biomembranes [112], [133]. An important role of peroxisomes in defense against ROS is demonstrated by the high sensitivity to UV-irradiation of mutant CHO cells defective in plasmalogen synthesis [114], and cells from patients with peroxisome biogenesis disorders (PBDs) with low levels of plasmalogens [115], [116], [117]. It has to be noted, that certain signalling lipids are derived from ether phospholipids (e. g., platelet activating factor), which act on several cellular signalling cascades and can influence ROS equilibrium indirectly [118].
4.3. Peroxisomes in inflammation, ischemia–reperfusion injury and related conditions
Hyperlipidemia is a hallmark of bacterial infections and it was suggested to be related to the elevation of the cytokine TNFα [119]. In rat liver TNFα suppresses markedly the expressions of catalase and other peroxisomal proteins [120] with simultaneous reduction of the mRNAs for peroxisomal lipid β-oxidation enzymes as well as for PPARα [121]. Significant reduction of the volume density of peroxisomes and several of their enzymes in liver was also reported in rats with pneumococcal sepsis [122]. Endotoxin treatment also reduced significantly the peroxisomal metabolism of lipids and fatty acids [123].
Another closely related pathological process associated with severe oxidative stress is the ischemia and reperfusion injury [124]. Peroxisomes from rat kidney [125] and liver [126] exposed to ischemia and reperfusion exhibited significant loss of catalase and lipid β-oxidation enzymes. Marked reduction of catalase and fatty acyl-CoA oxidase activities with structural alterations of peroxisomes were also reported in acute rejection of rat liver allografts [64]. Similar impairment of peroxisomal functions have also been reported in the central nervous system of rats with an experimental autoimmune encephalomyelitis [127]. The loss of peroxisomal functions in all above mentioned models could lead to the accumulation of harmful metabolites of arachidonic acid further intensifying the cell injury and inducing an inflammatory response.
Because of the central role of peroxisomes in the catabolism of inflammatory lipid mediators like leukotriene B4 [128], the impairment of peroxisomal function can contribute significantly to the prolongation and intensification of the inflammatory process. This is supported also by the observation of chronic inflammation with fibrosis/cirrhosis in the liver of children with Zellweger syndrome [129]. In this respect the activation of PPARα by leukotriene B4 [130] serves to limit the inflammatory process, providing a physiological mechanism to stop the damaging effects associated with inflammation [131]. This notion is confirmed by the demonstration of the important role of PPARα in the regulation of the inflammatory response in ischemia–reperfusion injury of liver [132]. In mice nullizygous for PPARα the extent of liver damage is significantly greater than in wild type littermates, and the expression of iNOS after ischemia–reperfusion is much less. Moreover, the treatment of normal mice with the PPARα agonist, WY-14643, reduces significantly the severity of liver pathology. The same PPARα agonist also is highly effective in treatment of dietary steatohepatitis in mice which results from the action of ROS on accumulated lipids and excessive formation of lipoperoxides in the liver [133]. In brain also, Wy-14643 prevents the β-amyloid related neuronal degeneration induced by ROS [111].
The above observations demonstrate clearly the participation of peroxisomes in the pathophysiology of inflammation and the protective role of their induction via the PPARs in limiting the damaging effects of this important pathologic process. It is almost impossible to dissect out the role of peroxisomes alone in the pathophysiology of the inflammatory process since PPARα induces not only several peroxisomal proteins but also affects the transcription of other genes such as those of mitochondrial and microsomal enzymes as well. Although the details of the cellular responses to activation of PPARs by their internal ligands, particularly the lipid mediators, and their effects upon cell and tissue injury and repair are just beginning to emerge [134], that information shall have a great impact on the development of novel therapeutic concepts in inflammation and repair processes [135].
5. Peroxisome proliferation and induction of oxidative stress
The massive proliferation of peroxisomes in the liver of rats treated with the hypolipidemic drug clofibrate was the first indication of a possible involvement of this organelle in the lipid metabolism in mammalian liver [136]. Since the central role of peroxisomes in β-oxidation of fatty acids in plant cells was at that time quite well known [137], the observations with clofibrate in rat liver led to the discovery of lipid β-oxidation in animal peroxisomes [138]. The list of compounds inducing peroxisome proliferation is quite long and includes hypolipidemic drugs, industrial chemicals such as plasticizers and lubricants as well as agrochemicals and many toxic environmental pollutants [139]. Selective transcription of peroxisomal genes by those chemicals is mediated by PPARα, which belongs to the family of nuclear transcription factors [140]. Whereas the expression of the genes of the lipid β-oxidation, particularly of acyl-CoA oxidase, is induced very strongly (10–30 fold depending on compound and dosage), the maximal induction of catalase does not exceed 1–2 fold [12], [141]. The disproportionate increase of H2O2-generating oxidases, particularly of fatty acid oxidase, in comparison to H2O2-scavenging catalase was suggested to be responsible for oxidative stress leading to the development of hepatic tumors in rodents treated with peroxisome proliferating compounds [83]. This notion was supported by in vitro studies using cultured cells transfected with cDNA for acyl-CoA oxidase and exposed subsequently to a fatty acid substrate. Indeed, the prolonged exposure to oxidative stress induced malignant transformation of cells which formed distinct tumors after injection to nude mice [142]. The oxidative stress however, does not seem to be the sole factor responsible for the development of tumors in rodents exposed to peroxisome proliferators as additional parameters such as potency to induce cell proliferation or to promote initiated cells are also of great importance [143]. Moreover, other factors such as suppression of apoptosis [144], perturbation of cell proliferation, and release of superoxide radicals from Kupffer cells [145] have also been suggested to play roles in the pathogenesis of tumors associated with peroxisome proliferation [146]. The central event in the carcinogenesis, however, seems to be the activation of PPARα [147], and indeed, the PPARα−/− mice are refractory not only to the peroxisome proliferating effect but are also resistant to hepato-carcinogenesis when fed a diet containing a potent non-genotoxic carcinogen such as WY-14643 [148]. Along the same line, the low level of PPARα in livers of primates and humans seems to be responsible for the resistance of those species to the carcinogenic effect of peroxisome proliferating compounds [149], although their lipid lowering effect is not affected by that [150], [151].
6. Mouse models, peroxisomal disorders, and ROS
In recent years, several PEX gene knockout-mouse models (e. g., PEX2, 5, 7, 11α, 11β, 13) have been developed to study peroxin loss/function and the pathogenetic mechanisms of PBDs [152]. PEX5−/− mice, which have been generated by disrupting the PEX5 gene encoding the PTS1 receptor for most peroxisomal matrix proteins, show a severe peroxisomal import defect, lack morphologically identifiable peroxisomes, and exhibit the typical biochemical abnormalities and pathological defects of Zellweger patients [153]. In addition, marked mitochondrial abnormalities were observed in various organs of the PEX5−/− mice (e. g., liver, kidney, adrenal cortex, heart) and specific cell types (skeletal and smooth muscle cells, neutrophils) [154]. Ultrastructural studies revealed the proliferation of pleomorphic mitochondria with alterations of the cristae and the outer mitochondrial membrane (Fig. 1C, D). Similar mitochondrial alterations, especially of the inner mitochondrial membrane, were also observed in hepatocytes using a mouse model with hepatocyte-selective elimination of peroxisomes (by inbreeding Pex5–loxP and albumin–Cre mice) (L-PEX5−/−) [155]. Such ultrastructural alterations of mitochondria have also been found in respiratory chain disorders [156], and other diseases associated with oxidative stress [157]. Biochemically, changes in the expression and activities of mitochondrial respiratory chain complexes, and a marked increase of mitochondrial MnSOD were described in PEX5−/− mice [154]. Furthermore, apoptosis of neuronal cells was highly increased in the brain of the PEX5−/− mouse [153]. In L-PEX5−/− mice, severely reduced activities of complex I, III, an V and a collapse of the mitochondrial inner membrane potential were reported [155]. No disturbance of the ATP levels and redox state of the liver but a compensatory increase of glycolysis and mitochondrial proliferation were detected [155]. It has been suggested that an increased production of ROS in altered mitochondria in combination with defective peroxisomal antioxidant mechanisms and the accumulation of lipid intermediates of the peroxisomal β-oxidation system, might contribute to the pathogenesis of Zellweger syndrome [154]. However, neither increased peroxide production, oxidative damage to proteins or lipids, nor elevation of oxidative stress defense mechanisms were found in L-PEX5−/− hepatocytes [155]. In line with this, evidence for increased oxidative stress was reported in fibroblasts of patients with multifunctional protein-2 deficiency, a peroxisomal β-oxidation defect, but not in patients with a PBD [158]. Furthermore, mitochondria of the PEX2−/− mouse, which has a remarkably similar clinical, biochemical and cellular phenotype as the PEX5−/− mouse, appear to be normal [159]. Another model, which links peroxisomal ROS production to kidney disease, is the MPV17−/− mouse. MPV17−/− mice have been reported to develop age-dependent hearing loss and severe renal failure owing to glomerular sclerosis [9]. Mpv17 has been localized to the peroxisomal membrane, and implicated in the regulation of peroxisomal ROS metabolism. Loss of the Mpv17 protein does not impair peroxisome biogenesis but instead leads to a reduced ability to produce ROS, whereas overexpression results in dramatically enhanced levels of intracellular ROS [9]. These findings have been challenged by a recent report, where Mpv17 has been localized to the inner mitochondrial membrane, and its absence or malfunction has been linked to oxidative phosphorylation failure and mtDNA depletion [160]. If this discrepancy is related to different Mpv17 isoforms, splice variant, or dual targeting, has to be elucidated.
7. Perspectives
The central role of peroxisomes in the generation and scavenging of H2O2 has been well-known ever since their discovery almost five decades ago. Recent studies, particularly in plant cells, have now revealed their involvement in the metabolism of ROS and nitric oxide that have important functions in intra- and inter-cellular signalling. Apparently, peroxisomes can have two ambivalent roles in the cell, as generators of oxidative stress and as a source of ROS signal mediators. It will be a great challenge for the future to dissect the UV- or ROS-mediated signalling pathways and the gene responses regulating peroxisomal metabolism, biogenesis and disease. These approaches require novel techniques and improved tools, which are partially available. For example, a new chemiluminescence method for a sensitive and real-time determination of H2O2 metabolism in suspensions of intact peroxisomes has been developed [161]. Furthermore, studies to assess the pH or the hydroxyl-radical formation in peroxisomes of living cells by targeting peptide-conjugated fluorophores carrying a PTS1 have been reported [162], [163], [164].
There is growing evidence for a role of peroxisomes in a variety of physiological and pathological processes involving ROS, particularly the participation of peroxisomes in the pathophysiology of inflammation and the protective role of their induction via the PPARs in limiting oxidative injury. Molecular details on the cellular responses to the activation of PPARs by their internal ligands, particularly the lipid mediators, and their effects upon cell and tissue injury and repair processes are just beginning to emerge [134]. In this respect, a novel area of medical research with prospects of vast therapeutic applications is developing [135], that deserves a great deal of attention.
References
- [1]
- G. Ermak, K.J. DaviesCalcium and oxidative stress: from cell signaling to cell deathMol. Immunol., 38 (2002), pp. 713-721
- [2]
- S.R. Terlecky, P.A. WaltonPeroxisomes and AgingBiochim. Biophys. Acta, 1763 (2006), pp. 1749-1754, 10.1016/j.bbamcr.2006.08.017(this issue)
- [3]
- W. DrogeOxidative stress and agingAdv. Exp. Med. Biol., 543 (2003), pp. 191-200
- [4]
- M. SaranTo what end does nature produce superoxide? NADPH oxidase as an autocrine modifier of membrane phospholipids generating paracrine lipid messengersFree Radical Res., 37 (2003), pp. 1045-1059
- [5]
- L. Moldovan, N.I. MoldovanOxygen free radicals and redox biology of organellesHistochem. Cell Biol., 122 (2004), pp. 395-412
- [6]
- P. Jezek, L. HlavataMitochondria in homeostasis of reactive oxygen species in cell, tissues, and organismInt. J. Biochem. Cell Biol., 37 (2005), pp. 2478-2503
- [7]
- C. De Duve, P. BaudhuinPeroxisomes (microbodies and related particles)Physiol. Rev., 46 (1966), pp. 323-357
- [8]
- B.M. Elliott, N.J. Dodd, C.R. ElcombeIncreased hydroxyl radical production in liver peroxisomal fractions from rats treated with peroxisome proliferatorsCarcinogenesis, 7 (1986), pp. 795-799
- [9]
- R.M. Zwacka, A. Reuter, E. Pfaff, J. Moll, K. Gorgas, M. Karasawa, H. WeiherThe glomerulosclerosis gene Mpv17 encodes a peroxisomal protein producing reactive oxygen speciesEMBO J., 13 (1994), pp. 5129-5134
- [10]
- D.B. Stolz, R. Zamora, Y. Vodovotz, P.A. Loughran, T.R. Billiar, Y.M. Kim, R.L. Simmons, S.C. WatkinsPeroxisomal localization of inducible nitric oxide synthase in hepatocytesHepatology, 36 (2002), pp. 81-93
- [11]
- A. Boveris, N. Oshino, B. ChanceThe cellular production of hydrogen peroxideBiochem. J., 128 (1972), pp. 617-630
- [12]
- H.D. Fahimi, A. Reinicke, M. Sujatta, S. Yokota, M. Ozel, F. Hartig, K. StegmeierThe short- and long-term effects of bezafibrate in the ratAnn. N. Y. Acad. Sci., 386 (1982), pp. 111-135
- [13]
- B.R. Bacon, R.S. BrittonHepatic injury in chronic iron overload. Role of lipid peroxidationChem. Biol. Interact., 70 (1989), pp. 183-226
- [14]
- S. Yokota, T. Oda, H.D. FahimiThe role of 15-lipoxygenase in disruption of the peroxisomal membrane and in programmed degradation of peroxisomes in normal rat liverJ. Histochem. Cytochem., 49 (2001), pp. 613-622
- [15]
- C.J. MastersCellular signalling: the role of the peroxisomeCell Signal, 8 (1996), pp. 197-208
- [16]
- J.K. Reddy, G.P. MannaertsPeroxisomal lipid metabolismAnnu. Rev. Nutr., 14 (1994), pp. 343-370
- [17]
- J.B. Barroso, F.J. Corpas, A. Carreras, L.M. Sandalio, R. Valderrama, J.M. Palma, J.A. Lupianez, L.A. del RioLocalization of nitric-oxide synthase in plant peroxisomesJ. Biol. Chem., 274 (1999), pp. 36729-36733
- [18]
- J.K. Hiltunen, Y. Poirier, Peroxisomal β-oxidation—A metabolic pathway with multiple functions, Biochim. Biophys. Acta (2006) (this issue), doi:10.1016/j.bbamcr.2006.08.034.
- [19]
- P.P. Van Veldhoven, G.P. MannaertsRole and organization of peroxisomal beta-oxidationAdv. Exp. Med. Biol., 466 (1999), pp. 261-272
- [20]
- J.K. ReddyPeroxisome proliferators and peroxisome proliferator-activated receptor alpha: biotic and xenobiotic sensingAm. J. Pathol., 164 (2004), pp. 2305-2321
- [21]
- S. Angermuller, H.D. FahimiUltrastructural cytochemical localization of uricase in peroxisomes of rat liverJ. Histochem. Cytochem., 34 (1986), pp. 159-165
- [22]
- A. Volkl, E. Baumgart, H.D. FahimiLocalization of urate oxidase in the crystalline cores of rat liver peroxisomes by immunocytochemistry and immunoblottingJ. Histochem. Cytochem., 36 (1988), pp. 329-336
- [23]
- A.V. Yeldandi, V. Yeldandi, S. Kumar, C.V. Murthy, X.D. Wang, K. Alvares, M.S. Rao, J.K. ReddyMolecular evolution of the urate oxidase-encoding gene in hominoid primates: nonsense mutationsGene, 109 (1991), pp. 281-284
- [24]
- B.N. Ames, R. Cathcart, E. Schwiers, P. HochsteinUric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: a hypothesisProc. Natl. Acad. Sci. U. S. A., 78 (1981), pp. 6858-6862