Re:Re: First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX)
작성자문형철작성시간20.05.20조회수2,241 목록 댓글 0beyond reason
First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid
Keywords
1. Antioxidants and free radicals: friend and foe of man
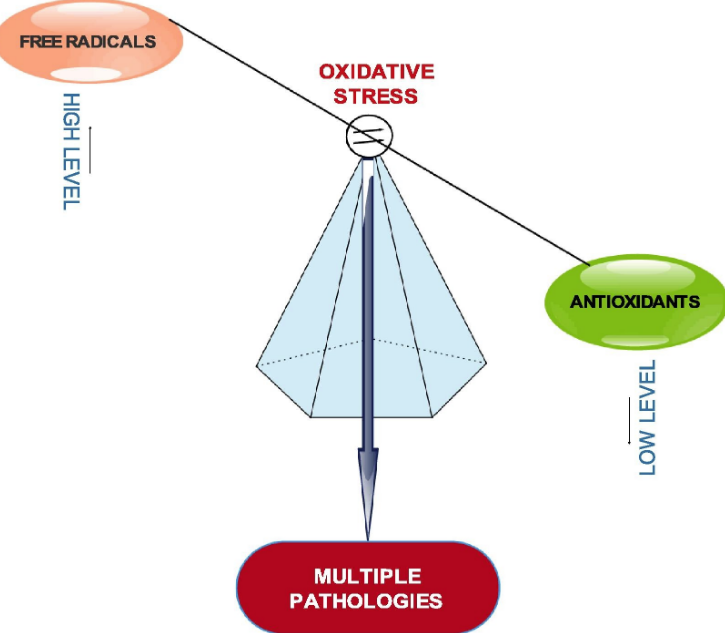
For over two and half decades, numerous studies have highlighted the nutritional and health benefits of antioxidants as well as the inverse correlation that exists between them and free radicals. Free radicals or reactive species through oxidative stress have been evidently implicated in the incidence and progression of several health conditions such as atherosclerosis, diabetes, cancer, neurodegenerative disorders, cardiovascular disorders and other chronic conditions.1 Oxidative stress is a cellular phenomenon or condition which occurs as a result of physiological imbalance between the levels of antioxidants and oxidants (free radicals or reactive species) in favour of oxidants (Fig. 1).
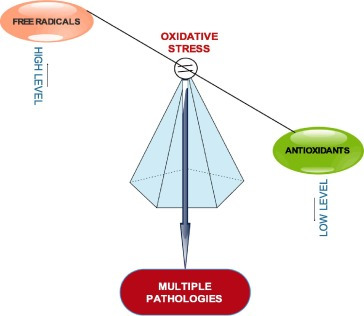
Fig. 1. Oxidative stress: Imbalance between free radicals and antioxidants.
The antioxidants are said to have been overwhelmed by the free radicals. This happens when the production of free radicals exceeds the level which the body’s natural antioxidant defense mechanisms can cope with; consequently creating a cellular oxidative environment which triggers the oxidation of essential biomolecules like DNA, protein and lipids, leading to multiple disease conditions. The interplay between the trio of free radicals antioxidants, and diseases is important in maintaining health, aging and age-related diseases.2 Input of exogenous antioxidants to human wellness has also been publicized. In fact, it has been suggested that reduced exposure to free radicals and increased intake of antioxidant rich foods or antioxidant supplements will enhance the body’s potential to minimize the risk of free radical related health problems.3
Antioxidants such as polyphenols, ascorbic acid, vitamin A, alpha-lipoic acid, thioredoxin, glutathione, melatonin, coenzyme Q, beta carotenoids, alpha-tocopherols as well as antioxidant enzymes including superoxide dismutase, catalase, glutathione-peroxidases, glutathione reductases and glutathione-s-transferases have been widely investigated for the prevention and treatment of diseases resulting from oxidative damage.4, 5, 6, 7
2. Levels of action of antioxidants
The antioxidants molecules that constitute the antioxidant defense grid in living systems act at different levels. These levels may be radical preventive, radical scavenging and radical-induced damage repair. On the basis of line of defense, antioxidants can be categorized as first line defense antioxidants, second line defense antioxidants, third line defense antioxidants and fourth line defense antioxidants.
2.1. First line defense antioxidants
These are a collection of antioxidants that act to suppress or prevent the formation of free radicals or reactive species in cells. They are very fast in neutralizing any molecule with the potential of developing into a free radical or any free radical with the ability to induce the production of other radicals. Three key enzymes: superoxide dismutase, catalase and glutathione peroxidase are top on the list. These enzymes respectively dismutate superoxide radical, breakdown hydrogen peroxides and hydroperoxides to harmless molecules (H2O2/alcohol and O2). The class also includes metal ion binding proteins like transferrin and caeruloplasmin which chelate or sequester iron and copper respectively, consequently preventing them from free radical formation.
2.2. Second line defense antioxidants
This group of antioxidants is often referred to as scavenging antioxidants. They scavenge active radicals to inhibit chain initiation and break chain propagation reactions. They neutralize or scavenge free radicals by donating electron to them, and in the process become free radicals themselves but of lesser damaging effects. These ‘new radicals’ are easily neutralized and made completely harmless by other antioxidants in this group. Most antioxidants including ascorbic acid, uric acid, glutathione, which are hydrophilic and alpha tocopherol (vitamin E) and ubiquinol which are lipophilic belong to this category.
2.3. Third line defense antioxidants
This category of antioxidants only comes into play after free radical damage has occurred. They are de novo enzymes which repair the damage caused by free radicals to biomolecules and reconstitute the damaged cell membrane. They are a group of enzymes for repair of damaged DNA, protein and lipids. They also do a sort of ‘clean up duty’, they recognize, breakdown and remove oxidized or damaged proteins, DNA and lipids, to prevent their accumulation which can be toxic to body tissues. Common examples include The DNA repair enzyme systems (polymerases, glycosylases and nucleases), proteolytic enzymes (proteinases, proteases and peptidases) which are located both in cytosol and mitochondria of mammalian cells.
2.4. Fourth line defense antioxidants
The action of these ‘antioxidants’ basically involves an adaptation mechanism in which they utilize the signals required for free radicals production and reaction to prevent the formation or reaction of such free radicals. The signal generated from the free radical formed induces the formation and transport of an appropriate antioxidant to the right site.8
3. Oxygen radicals (e.g. superoxide anion) play fundamental role in oxidative stress and oxidative damage in biological systems
The primary role of oxidative stress (OS) in the etiology and progression of several disease conditions is evident by a number of clinical and experimental studies. OS appears to be the foundation for the induction of multiple cellular pathways associated with damage of important biomolecules and subcellular structures in cells. Different reactive species are involved in cellular oxidative stress and oxidative damage. A few examples include hydroxyl radical (∗OH); superoxide anion (∗O2), singlet oxygen radical (1O2−), peroxyl radical (∗ROO), nitric oxide radical (∗NO) and lipid peroxy radical (∗LOO) as well as peroxynitrate (ONOO−), trichloromethane (∗CHCl3) and hypochlorous acid (∗HOCl) which are not necessarily free radicals but have high tendency to generate free radicals. The whole process of radical-induced toxicity appears to stem from the generation of oxygen radicals (superoxide anion or singlet oxygen radical) from which other reactive oxygen species (ROS) and reactive nitrogen species (RNS) can be derived.
4. Formation of oxygen radicals in biological systems
There are many and various types of free radicals in biological systems, however, those derived from oxygen, generally termed reactive oxygen species (ROS) are of major concern and interest to researchers in the field of free radical chemistry. Oxygen is highly susceptible to free radical formation because of its electronic structure. The molecule has in its outer shell, two unpaired electrons in separate electronic orbitals. Oxygen radicals such as superoxide anion (∗O2) and singlet oxygen, (1O2−) are easily generated from the sequential reduction of molecular oxygen via step-wise addition of electrons.
They are constantly formed through a couple of normal cell reactions or processes. In most cases, they are generated for beneficial reasons, for instance they are used for destruction of microbes and pathogens by white blood cells. It is documented that oxygen radicals are involved in intercellular and intracellular signaling. Precisely, superoxide anion and hydrogen peroxide have been reported to act as mitogens, thereby promoting the rate of DNA replication and cell proliferation in a variety of cultured cells.9, 10 Nonetheless, in excess amounts, oxygen-derived radicals are very harmful to living organisms. Besides being injurious themselves, they are capable of generating other free radicals that could be more destructive.
Before we consider how other radicals are derived from oxygen radicals, it is necessary to mention some normal physiological processes that lead to formation of oxygen radicals. Mitochondrial energy production pathway (MEPP) is a major source of oxygen radicals. These radicals are formed as oxygen is reduced along the electron transport chain which is located in the inner membrane of the organelle. Phagocytic leucocytes (white blood cells) especially neutrophils produce abundant of oxygen radicals through NADPH-Oxidase activity (O2 + e− → ∗O2−) as effort to destroy invading pathogens in defense or protection of the host organism.11 Oxygen-derived radicals are also produced as integral metabolites in cascades of enzyme catalyzed reactions. Exposure to ionizing radiation is well known to promote oxygen radical formation within biological systems through radiolysis (interaction of radiation and water).12 Hypoxia or/and hyperoxia condition in cells has been noted to generate numerous oxygen-derived radicals.13 Besides, a couple of therapeutic drugs such as acetaminophen have been reported to cause oxidizing effects on cells, consequently leading to formation of oxygen radicals through the activity of drug metabolizing enzymes known as cytochrome P-450 system.14
5. Role of superoxide anion in the generation of other reactive species
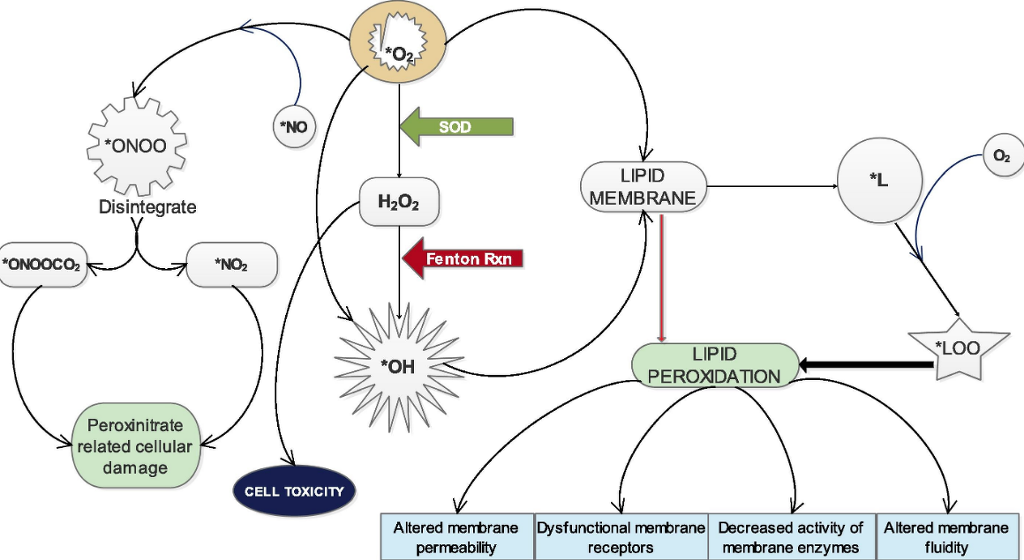
Superoxide anion (∗O2) which is constantly being generated through endogenous processes and exogenous sources can through different pathways or chain reactions cause the formation of several other reactive species.15 It can directly produce hydroxy radical (∗OH) (∗O2 + 2e− → ∗OH) or indirectly from hydrogen peroxide which results from its dismutation by SOD (H2O2 + e− → ∗OH), through Fenton reaction which occurs in the presence of Fe2+ (Fig. 2). ∗OH is the most dangerous free radical and major culprit in free radical–mediated toxicity in cells.16, 3 Both ∗O2 and ∗OH can individually act on lipid membranes to promote the formation of lipid radical (∗L) which in the presence of oxygen is capable of generating lipid peroxy radical (∗LOO)3 (Fig. 2). These radicals attack lipid membranes to initiate the most devastating effect of oxidative stress which is membrane lipid peroxidation. Peroxidation of membrane results in huge compromise of the structural and functional integrity of the cell characterized by altered membrane permeability,17 dysfunctionality of membrane receptors,18 decreased activity of membrane-bound enzymes19 and increased membrane rigidity which decreases membrane fluidity.20 The possibility of free radicals directly attacking membrane proteins, induce lipid-lipid, lipid-protein and protein-protein cross linking also exist.21 Peroxynitrate (ONOO−), a powerful oxidant is formed when ∗O2 reacts with nitric oxide radical (∗NO) in the presence of arginine and NADPH-Oxidase enzyme. (ONOO−) is a damaging oxidant which can cause lipid peroxidation, protein oxidation, protein nitration and enzyme inactivation. It can also reacts with carbon dioxide to generate nitrosoperoxycarbonate (ONOOCO2−) which readily disintegrate to form carbonate radical and nitrogen dioxide radical (∗NO2−) (Fig. 2). These two radicals are believed to be the main actors in peroxynitrite-related cellular damage.22 Besides, there are a couple of other reactive species that result from superoxide anion.
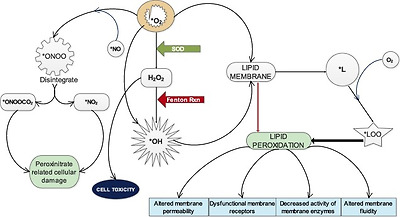
Fig. 2. Role of superoxide anion in the generation of other reactive species.
6. Antioxidant response against oxygen radicals of endogenous or exogenous sources
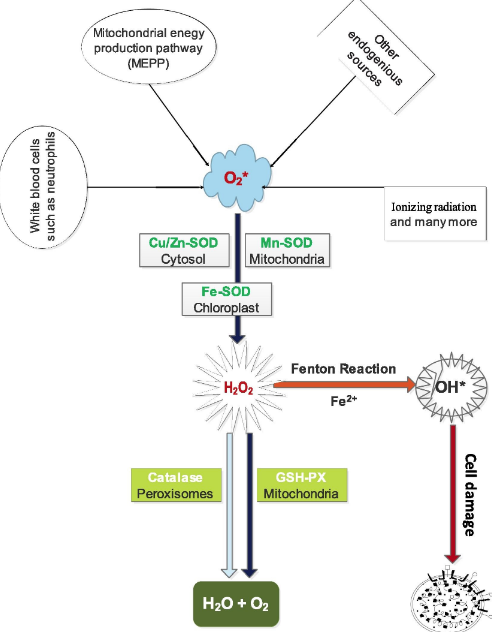
Superoxide dismutase, catalase and glutathione peroxidase are antioxidant enzymes which do not only play fundamental but indispensable role in the antioxidant protective capacity of biological systems against free radical attack. The superoxide radical (∗O2) or singlet oxygen radical (1O2−) generated in tissues through metabolism or reactions in cells is catalytically converted to hydrogen peroxide (H2O2) and molecular oxygen (O2) by superoxide dismutase (SOD). H2O2 when accumulated is toxic to body tissues or cells. Also, in the presence of Fe2+ it is converted to deleterious hydroxyl radical (∗OH) through Fenton reaction. In order to prevent this phenomenon, catalase (another antioxidant enzyme) which is abundant in the peroxisomes breaks down H2O2 into water and molecular oxygen, consequently curtailing free radical-induced damage (Fig. 3). However, catalase is absent in the mitochondria, hence the reduction of H2O2 to water and lipid peroxides to their corresponding alcohols is carried out by Glutathione Peroxidase (GPx). This collective protective effort is termed first line antioxidant defense, and the antioxidants involved are referred to as first line defense antioxidants. The role and effectiveness of the first line defense antioxidants which basically include superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX) is therefore important and indispensable in the entire defense strategy of antioxidants, especially in reference to super oxide anion radical (∗O2) which is perpetually generated in normal body metabolism through several processes.
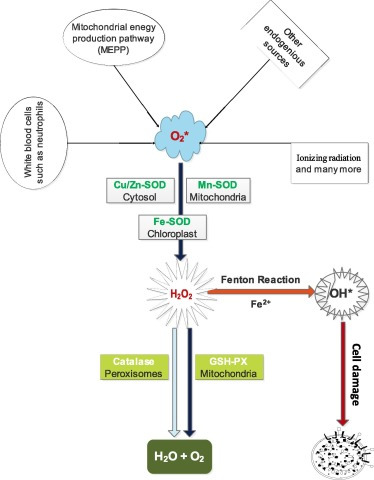
Fig. 3. First line antioxidants defense against reactive oxygen species.
7. Superoxide dismutase (SOD, EC 1.15.1.1)
Superoxide dismutase (SOD) is the first detoxification enzyme and most powerful antioxidant in the cell. It is an important endogenous antioxidant enzyme that acts as a component of first line defense system against reactive oxygen species (ROS). It catalyzes the dismutation of two molecules of superoxide anion (∗O2) to hydrogen peroxide (H2O2) and molecular oxygen (O2), consequently rendering the potentially harmful superoxide anion less hazardous. SOD is a metalloenzyme and hence, requires a metal cofactor for its activity. On the basis of the type of metal ion required as cofactor by SOD, various forms of the enzyme exist.23, 24 The metal ions which are normally bound by SOD are iron (Fe), zinc (Zn) copper (Cu) and manganese (Mn). In this regards, SODs are classified into three forms and these include (i) Fe-SOD which is commonly found in prokaryotes and chloroplasts of some plants (ii) Mn-SOD which is present in prokaryotes and mitochondria of eukaryotes and (iii) Cu/Zn-SOD is predominant in eukaryotes and more distributed, localized basically in cytosol but also found in chloroplasts and peroxisomes.25, 26
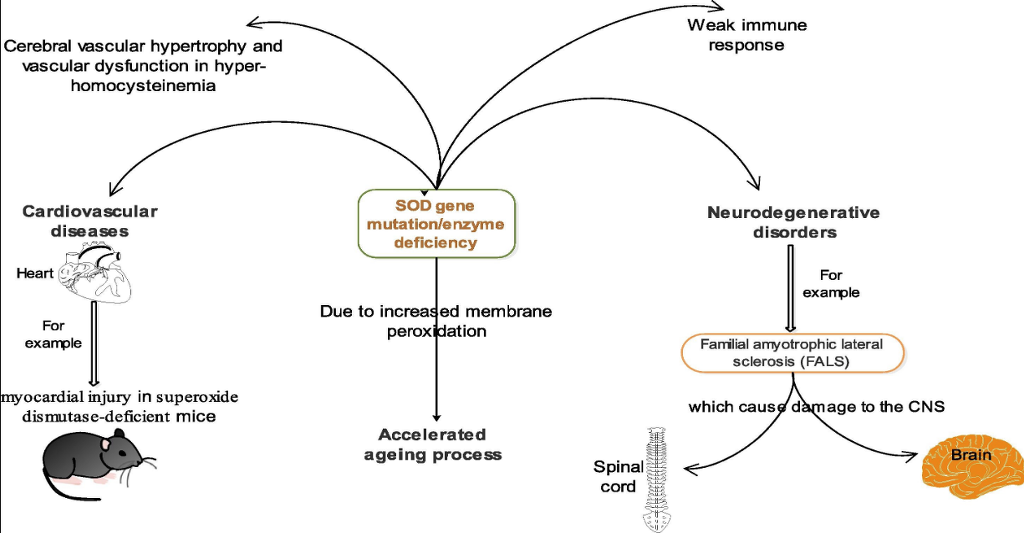
The various ‘isozymes’ of SOD are encoded by different genes. According to Rosen et al., Cu/Zn-SOD is encoded by the SOD1 gene mapping to chromosome 21. MnSOD is encoded by the SOD2 gene mapping to chromosome 6. The eukaryotic extracellular SOD Cu-Zn SOD is encoded by the SOD3 gene mapping to chromosome 4.27 Substantial amount of SOD3 is found virtually in all human tissues. A number of tissues including the heart have been observed to possess the cellular resources to transcribe SOD3 mRNA from SOD DNA. This is of great importance since SOD3 is the major enzymatic antioxidant defense against vascular and cardiovascular diseases (neurological diseases, lung disease, atherosclerosis, diabetes, hypertension, inflammatory conditions and ischemia-reperfusion injury. Association between SOD deficiency and a number of pathologies has been observed in both animals and humans (Fig. 4). Lebovitz et al.28 noted neurodegeneration, myocardial injury, and perinatal death in mitochondrial superoxide dismutase-deficient mice. Zn-deficient wild-type and mutant human SOD1 have been implicated in the disease familial amyotrophic lateral sclerosis (FALS), also known as Lou Gehrig’s disease which affects the nerve cells in the spinal cord and the brain.29, 30 Recently, Dayal and colleagues informed that deficiency of superoxide dismutase promoted cerebral vascular hypertrophy and vascular dysfunction in hyperhomocysteinemia.31
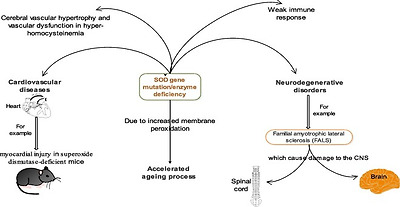
Fig. 4. Some pathological effects associated with SOD gene mutation or SOD deficiency.
SOD enzyme deficiency is common. Hence, the enzyme is indispensable to cellular health, protecting body cells from excessive oxygen radicals, free radicals and other harmful agents that promote aging or cell death. The levels of SODs decline with age, whereas free radical formation increases. It has been suggested that proper daily SOD supplementation will protect the immune system and significantly reduce one’s chances of diseases and ultimately slow down aging process. Krishnamurthy and Wadhwani encouraged the consumption of cabbage, Brussels sprouts, wheat grass, barley grass and broccoli as natural sources of SOD.32
7.1. Catalase (CAT, EC 1.11.1.6)
Catalase (CAT) is a 240 kilodalton (kDa) tetrameric protein with four similar subunits and is encoded by ctt1 gene mapping to chromosome 11.33 Each polypeptide subunit is 60 kDa in weight and contains a single ferriprotoporphyrin.34 CAT is a common antioxidant enzyme present almost in all living tissues that utilize oxygen. The enzyme uses either iron or manganese as a cofactor and catalyzes the degradation or reduction of hydrogen peroxide (H2O2) to water and molecular oxygen, consequently completing the detoxification process imitated by SOD.35, 36 It is abundant in cells, where it continuously scouts for hydrogen peroxide molecules. CAT is highly efficient; it can breaks down millions of hydrogen peroxide molecules in one second. The enzyme is located primarily in the peroxisomes but absent in mitochondria of mammalian cells. The only exception is the mitochondria present in the heart of rat.33 This implies that the breakdown of hydrogen peroxide to water and oxygen is carried out by another enzyme known as glutathione peroxidase in mammalian cell mitochondria.
CAT also reacts efficiently with hydrogen donors such as methanol, ethanol, formic acid, or phenols with peroxidase activity. The activity of CAT takes place in two steps. A molecule of hydrogen peroxide oxidizes the heme to an oxyferryl species. A porphyrin cation radical is generated when one oxidation equivalent is removed from iron and one from the porphyrin ring. A second hydrogen peroxide molecule acts as a reducing agent to regenerate the resting state enzyme, producing a molecule of oxygen and water.36 Hydrogen peroxide though at low amounts tends to regulate some physiological processes such as signaling in cell proliferation, cell death, carbohydrate metabolism, mitochondrial function, and platelet activation and maintenance of normal thiol redox-balance,37 however, at high concentrations it has been reported to be very deleterious to cells.38 Hence, the ability of CAT to effectively limit H2O2 concentration in cells underlines its importance in the aforementioned physiological processes as well as being a first line antioxidant defense enzyme. The deficiency or mutation of the enzyme has been linked with various disease conditions and abnormalities.39
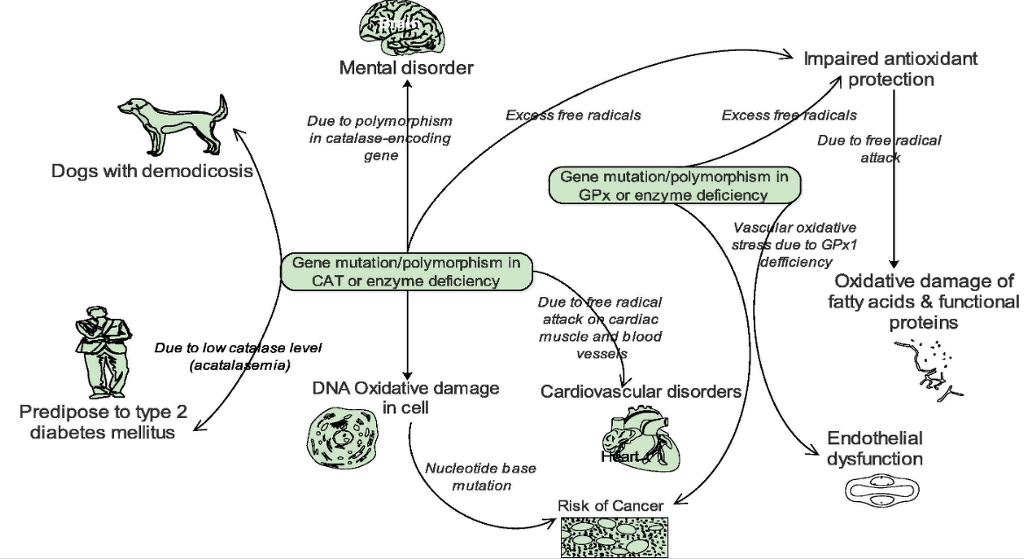
Zámocký and Koller associated oxidative DNA damage and subsequent risk of cancer susceptibility in individuals with genetic polymorphisms and altered gene expression/activity in catalase.40 Development of mental disorder has also resulted from polymorphism in the catalase-encoding gene.41 In addition, humans with low catalase levels (acatalasemia) are said to be more predisposed to type 2 diabetes mellitus (Fig. 5).39 and are sometimes affected by atherosclerosis and neoplasm. A significant reduction in catalase activity has been observed in dogs with localized and generalized demodicosis.42, 43
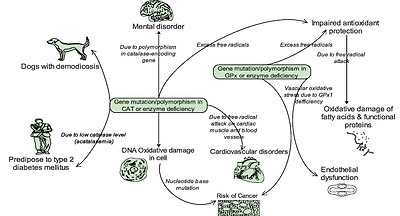
Fig. 5. Some pathological effects associated with gene mutation, polymorphism and deficiency of CAT and GPx.
7.2. Glutathione peroxidases (GPx, EC 1.11.1.9)
Glutathione Peroxidase (GPx) is an important intracellular enzyme that breakdown hydrogen peroxides (H2O2) to water; and lipid peroxides to their corresponding alcohols mainly in the mitochondria and sometimes in the cytosol.39 Most times, its activity depends on a micronutrient cofactor known as selenium. For this reason, GPX is often referred to as a selenocysteine peroxidase. The enzyme plays a more crucial role of inhibiting lipid peroxidation process, and therefore protects cells from oxidative stress.25
According to Morón and Cortázar, there are at least eight GPx enzymes in human, GPx1–GPx8. The GPx 1–8 genes are mapped to chromosomes 3, 14, 5, 19, 6, 6, 1 and 5 respectively.44 Among the glutathione peroxidases, GPx1 is the most abundant selenoperoxidase and is present virtually in all cells.45 GPx2 is found much more in the gastrointestinal tract primarily in the intestine.46, 47 The kidney relative to other tissues is the primary location for GPx3, though; the enzyme is also present in extracellular fluids as a glycoprotein.48 Most forms of GPx are tetrameric in structure but GPx4 which is often regarded as phospholipid hydroperoxide is a monomer and differ in substrate specificity. This is because Gpx4 is the only GPx enzyme that breaks down phospholipid hydroperoxides. The enzyme also has a mitochondrial isoform that mediates the apoptotic response to oxidative stress49 and has a peroxidase independent structural role in sperm maturation.45, 50 GPx5 from both humans and rodents and Rodent GPx6 differ from other glutathione peroxidases in that their activities are independent of Selenium.51 This invariably implies that these forms of GPx may not be able to effectively scavenge H2O2, a feature which is characteristic of the selenium-dependent glutathione peroxidases.52
The clinical importance of GPx has been underlined by a number of studies. Chabory et al. postulated that individuals with lower GPx activity are predisposed to impaired antioxidant protection, which leads to oxidative damage to membrane fatty acid s and functional proteins (Fig. 5), and by inference, neurotoxic damage.53 Forgione and colleagues had previously hypothesized that GPX1 deficiency directly induces an increase in vascular oxidative stress, with attendant endothelial dysfunction.54 Glutathione peroxidases, particularly GPx1 have also been implicated in the development and prevention of many common and complex diseases, including cancer and cardiovascular disease (Fig. 5).55
8. Conclusion
The activities of superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX) constitute a first line antioxidant defence system which plays a key and fundamental role in the total defense mechanisms and strategies in biological systems.
Conflict of interest statement
The author unanimously declare that there is no conflict of interest concerning this article.
References
- 1
- D. Giustarini, I. Dalle-Donne, D. Tsikas, R. RossiOxidative stress and human diseases: origin, link, measurement, mechanisms, and biomarkersCrit Rev Clin Lab Sci, 46 (2009), pp. 241-281
- 2
- K. RahmanStudies on free radicals, antioxidants, and co-factorsClin Intervent Aging, 2 (2007), p. 219
- 3
- V. Lobo, A. Patil, A. Phatak, N. ChandraFree radicals, antioxidants and functional foods: impact on human healthPharmacogn Rev, 4 (2010), p. 118
- 4
- S. Hercberg, P. Galan, P. Preziosi, et al.The SU. VI. MAX Study: a randomized, placebo-controlled trial of the health effects of antioxidant vitamins and mineralsArch Int Med, 164 (2004), pp. 2335-2342
- 5
- B. Halliwell, J. Rafter, A. JennerHealth promotion by flavonoids, tocopherols, tocotrienols, and other phenols: direct or indirect effects? Antioxidant or not?Am J Clin Nutr, 81 (2005), pp. 268S-276S
- 6
- G. Yuan, B. Sun, J. Yuan, Q. WangEffect of 1-methylcyclopropene on shelf life, visual quality, antioxidant enzymes and health-promoting compounds in broccoli floretsFood Chem, 118 (2010), pp. 774-781
- 7
- M.W. Dysken, M. Sano, S. Asthana, et al.Effect of vitamin E and memantine on functional decline in Alzheimer disease: the TEAM-AD VA cooperative randomized trialJAMA, 311 (2014), pp. 33-44
- 8
- E. NikiAntioxidant defenses in eukaryotic cellsG. Poli, E. Albano, M.U. Dianzani (Eds.), Free radicals: from basic science to medicine, Birkhauser Verlag, Basel, Switzerland (1993), pp. 365-373
- 9
- M. Valko, D. Leibfritz, J. Moncol, M.T. Cronin, M. Mazur, J. TelserFree radicals and antioxidants in normal physiological functions and human diseaseInt J Biochem Cell Biol, 39 (2007), pp. 44-84
- 10
- L.A. Pham-Huy, H. He, C. Pham-HuyFree radicals, antioxidants in disease and healthInt J Biomed Sci: IJBS, 4 (2008), p. 89
- 11
- J.C. Fantone, P.A. WardRole of oxygen-derived free radicals and metabolites in leukocyte-dependent inflammatory reactionsAm J Pathol, 107 (1982), p. 395