Re:Re:Glutamine addiction: a new therapeutic target in cancer - 2010년 Cell 리뷰논문
작성자문형철작성시간20.02.17조회수715 목록 댓글 0BEYOND REASON
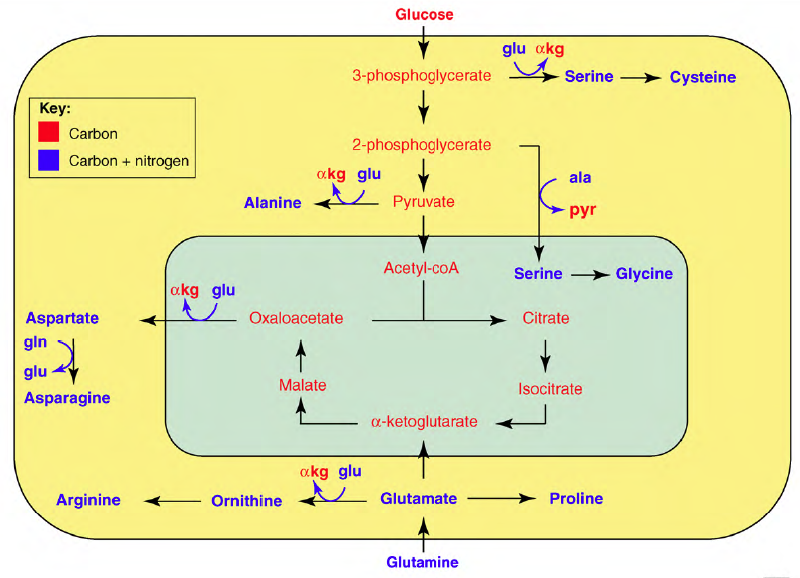
Figure 1. Glucose and glutamine can provide the carbon and nitrogen for the synthesis of the nonessential amino acids. High throughput glucose and glutamine (gln) uptake provide the growing cell with a large pool of carbon and nitrogen for the biosynthesis of the nonessential amino acids. Compounds containing carbon, but not nitrogen, are shown in red, whereas those containing carbon and nitrogen are shown in blue. Carbon precursors derived from glycolysis (3-phosphoglycerate, 2-
phosphoglycerate, pyruvate) and glutaminolysis (OAA, GA g-semialdehyde) serve as the carbon substrate for amino acid biosynthesis. Glutamine-derived GA (glu) donates its amine group to these carbon substrates to produce nonessential amino acids (serine, alanine, aspartate, ornithine) and a-ketoglutarate (akg). Alanine (ala) serves as the amine donor to produce serine and pyruvate (pyr) in the mitochondrion for the synthesis of glycine. Glutamine provides the carbon and nitrogen for the synthesis of proline, ornithine and arginine. Glutamine can also serve as a direct nitrogen donor in the synthesis of asparagine from aspartic acid.
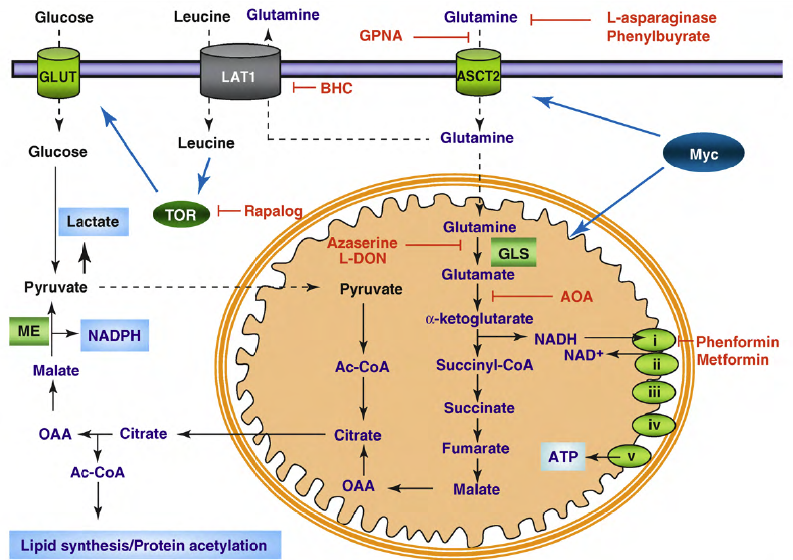
Figure 2. Pharmacologic targets in glutamine metabolism. Myc enables cancer cells to maximize glutamine uptake from the extracellular space through upregulation of the glutamine importer, ASCT2. Uptake of glutamine can be suppressed both by depletion of glutamine from the extracellular space with L-asparaginase and phenylbutyrate or through inhibition of ASCT2-dependent uptake with L-g-glutamyl-p-nitroanilide (GPNA).
Once glutamine enters the cell, it can be metabolized through glutaminolysis to provide NADPH or exported to facilitate TOR kinase activation. Myc enables conversion of glutamine into GA via upregulation of GLS, an enzyme whose activity can be inhibited by treatment with L-DON and azaserine. Transamination of GA to a-ketoglutarate can be inhibited with AOA. Mitochondrial metabolism of a-ketoglutarate leads to the production of citrate, its cleavage into OAA, reduction of OAA into malate and oxidation of malate to pyruvate via ME to produce pyruvate and NADPH. The high rate of glutamine metabolism through successive steps into OAA establishes glutamine as the primary anaplerotic substrate. This metabolism requires the regeneration of NAD+ through the electron transport chain, a process that can be inhibited with the biguanides, phenformin and metformin. The use of glutamine as a substrate for the amino acid exchanger LAT1 can be suppressed with BCH treatment, and TOR activity can be suppressed by rapamycin treatment.
Most cancers depend on a high rate of aerobic glycolysis for their continued growth and survival. Paradoxically, some cancer cell lines also display addiction to gluta- mine despite the fact that glutamine is a nonessential amino acid that can be synthesized from glucose. The high rate of glutamine uptake exhibited by glutamine- dependent cells does not appear to result solely from its role as a nitrogen donor in nucleotide and amino acid biosynthesis. Instead, glutamine plays a required role in the uptake of essential amino acids and in maintaining activation of TOR (target of rapamycin) kinase. More- over, in many cancer cells, glutamine is the primary mitochondrial substrate and is required for maintenance of mitochondrial membrane potential and integrity and for support of the NADPH production needed for redox control and macromolecular synthesis.
Cancer cells can be addicted to glutamine
The advent of molecular cancer genetics diverted biologists away from studies of cancer cell metabolism. However, two recent sets of observations have begun to stimulate in- terest into how signal transduction is integrated with metabolism. First, several oncogenes and tumor suppres- sors have been linked to the regulation of metabolic pro- cesses [1–3]. Second, the efficacy of several cancer therapies has been associated with their effects on metab- olism [4–7]. One of the implications of these studies is the synergistic potential of combining pharmacologics that target signal transduction components with those that target metabolic pathways.
One of the oldest conundrums in cancer biology is Warburg’s observation that cancer cells tended to take up more glucose and produce more lactic acid than normal tissue [8]. These observations led Warburg to hypothesize that cancer resulted from the regression of cells to the more primitive metabolism exhibited by proliferating single cell eukaryotes [9]. Recent studies have implicated oncogenic activation of glucose uptake as the cause of the ‘Warburg effect’ [2]. Constitutively activated components of phos- phoinositide 3-kinase (PI3K) signaling can directly stimu- late levels of glucose uptake and metabolism that exceed the capacity of the cell to use glucose in support of bioe- nergetic and macromolecular synthesis. When this occurs, the cancer cell secretes excess glycolytic metabolites in the form of lactic acid. In some tumors, this seemingly wasteful metabolism of glucose is mirrored by a similarly inefficient metabolism of glutamine [10,11]. Such cancer cells, in fact,
cannot survive in the absence of exogenous glutamine and exhibit ‘glutamine addiction’ [12]. Most cancer researchers have viewed the switch of glutamine from being a nones- sential amino acid (NEAA) to an essential amino acid (EAA) as an artifact of in vitro culture. However, recent studies have suggested that glutamine is a key substrate required for anabolic growth of mammalian cells. This review addresses the role of glutamine in cell growth, the signaling pathways that regulate the cellular utiliz- ation of glutamine and potential therapeutic strategies that might exploit the dependence of certain cancer cells on glutamine.
Glutamine provides nitrogen for protein and nucleotide
synthesis
The growing cancer must synthesize nitrogenous com-
pounds in the form of nucleotides and NEAAs. Glutamine
is the obligate nitrogen donor in as many as three
independent enzymatic steps for purine synthesis [phos-
phoribosylpyrophosphate (PRPP) amidotranferase, phos-
phoribosylformylglycinamidine (FGAM) synthetase, GMP
synthetase] and in two independent enzymatic steps for
pyrimidine synthesis (carbamoyl phosphate synthetase II,
CTP synthetase) [13–15]. In these reactions, glutamine
donates its amide (g nitrogen) group and is converted to
glutamic acid (GA) in the process.
GA is the primary nitrogen donor for the synthesis of the
NEAAs [13,15] (Figure 1). The transaminases transfer the
amine group from GA (originally the a-nitrogen of gluta-
mine) to a-ketoacids. The a-ketoacids used to generate
NEAAs are the carbon catabolites of glucose or glutamine:
pyruvate, 3-phosphoglycerate, oxaloacetate, and glutamic
acid gamma-semialdehyde, which are used to synthesize
alanine, serine, aspartate, and ornithine, respectively.
Serine is a precursor for glycine and cysteine biosynthesis,
ornithine is a precursor for arginine biosynthesis, and
aspartate is a precursor for asparagine biosynthesis (glu-
tamine obligately donates its amide group to this reaction).
GA contributes its carbon skeleton and nitrogen to the
synthesis of proline. Tyrosine is the only NEAA that is not
derived from glucose or glutamine; it is directly produced
from the EAA phenylalanine.