https://www.cell.com/immunity/fulltext/S1074-7613(24)00275-9
| 서론 NK 세포는 순환하는 세포독성 림프구로, MHC class I에 독립적으로 종양 세포를 공격하며, 과립(lytic granules)과 IFN-γ 생산을 통해 암 면역에 핵심적입니다. 종양 환자에서 NK 세포 시그니처가 생존율 향상과 연관되지만, TME(저산소, 고지질, 저당분 환경)에서 NK 세포 기능이 억제됩니다. NK 세포는 해당작용(glycolysis)에 의존적이지만, 단식 같은 식이 제한은 대사, 암, 면역에 영향을 미칩니다. 이 연구는 주기적 단식(CFD: 48시간 물만 먹는 단식 후 재급식)이 NK 세포를 재프로그래밍하여 항종양 효과를 내는 것을 중점으로 합니다. 이는 T 세포나 B 세포와 달리 NK 세포의 고유 면역(innate immunity) 강화에 초점을 맞춥니다. 방법
단식은 조직 특이적 적응을 통해 NK 세포를 강화: 비장(FA 산화로 대사 재프로그래밍), 골수(IL-12 프라이밍으로 기능 향상). NK 세포는 GCs에 내성적이며, FA를 OXPHOS 에너지로 사용(지방 축적 없이). 이는 T 세포(해당작용 의존, 단식 시 사멸 증가)와 대조. 골수 주재가 영양 스트레스 시 생존 지원. IL-12+ 골수세포 증가는 지방생성(adipogenesis)이나 미생물총과 연관 가능. 지방산 산화나 골수 이동 중 하나만 방해해도 종양 제어 실패. |
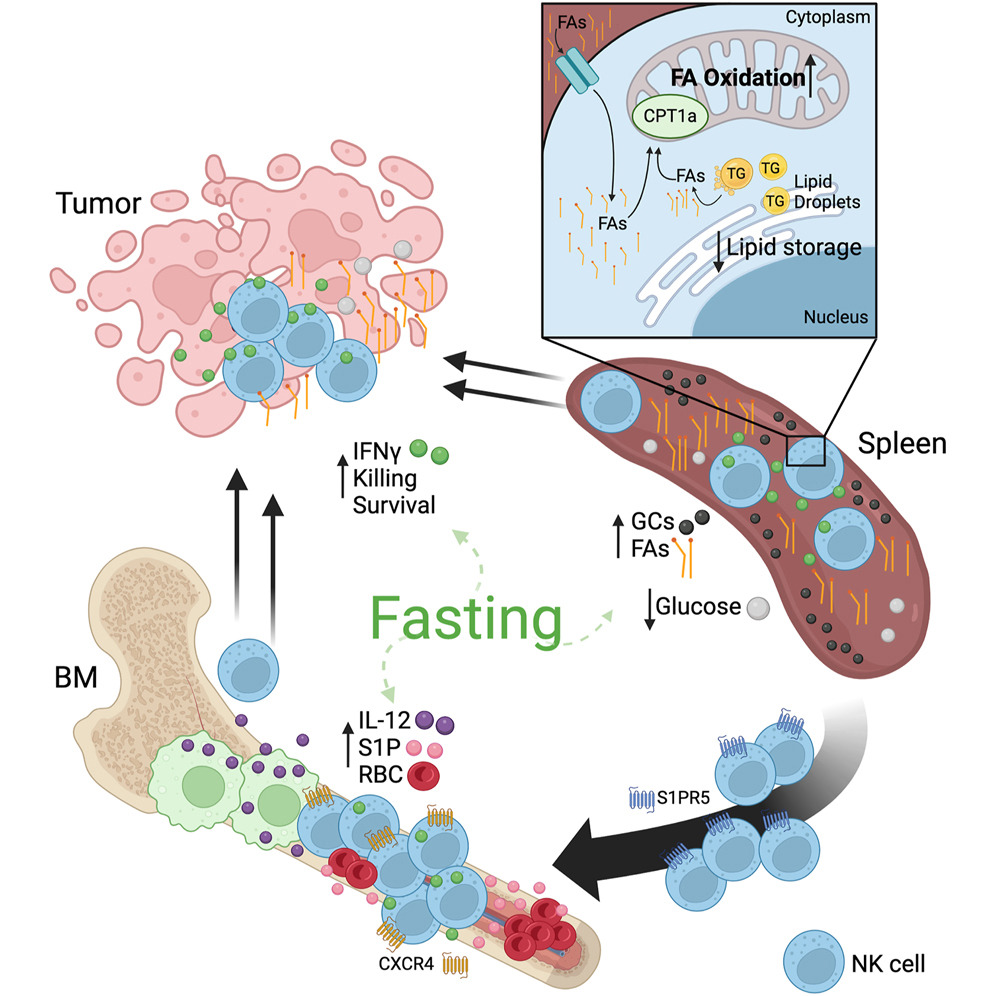
그림 설명: 단식과 NK 세포의 항종양 메커니즘 요약 (Graphical Abstract) 1. 전체 개요
|
Highlights
•
Cyclic fasting promotes NK cell-mediated anti-tumor immunity
•
Cyclic fasting promotes metabolic reprogramming of splenic NK cells
•
Fasting-induced fatty acid oxidation improves the NK cell anti-tumor response
•
Cyclic fasting redistributes NK cells to the bone marrow, where they are primed by IL-12
Summary
Fasting is associated with improved outcomes in cancer. Here, we investigated the impact of fasting on natural killer (NK) cell anti-tumor immunity. Cyclic fasting improved immunity against solid and metastatic tumors in an NK cell-dependent manner. During fasting, NK cells underwent redistribution from peripheral tissues to the bone marrow (BM). In humans, fasting also reduced circulating NK cell numbers. NK cells in the spleen of fasted mice were metabolically rewired by elevated concentrations of fatty acids and glucocorticoids, augmenting fatty acid metabolism via increased expression of the enzyme CPT1A, and Cpt1a deletion impaired NK cell survival and function in this setting. In parallel, redistribution of NK cells to the BM during fasting required the trafficking mediators S1PR5 and CXCR4. These cells were primed by an increased pool of interleukin (IL)-12-expressing BM myeloid cells, which improved IFN-γ production. Our findings identify a link between dietary restriction and optimized innate immune responses, with the potential to enhance immunotherapy strategies.
주기적 단식은 NK 세포 매개 항종양 면역을 촉진한다 •
주기적 단식은 비장 NK 세포의 대사 재프로그래밍을 촉진한다 •
단식 유발 지방산 산화는 NK 세포의 항종양 반응을 개선한다 •
주기적 단식은 NK 세포를 골수로 재분포시키며, 그곳에서 IL-12에 의해 프라이밍된다

Summary
단식은
암에서 개선된 결과와 연관되어 있다.
여기서 우리는 단식이
자연 살해(NK) 세포 항종양 면역에 미치는 영향을 조사했다.
주기적 단식은
NK 세포 의존적 방식으로 고형 종양과 전이 종양에 대한 면역을 개선했다.
단식 중 NK 세포는
주변 조직에서 골수(BM)로 재분포되었다.
인간에서도 단식은
순환 NK 세포 수를 감소시켰다.
단식한 쥐의 비장 NK 세포는
지방산과 글루코코르티코이드 농도 상승에 의해 대사적으로 재배선되었으며,
효소 CPT1A 발현 증가를 통해 지방산 대사를 증강시켰다.
Cpt1a 결핍은
이 환경에서 NK 세포 생존과 기능을 손상시켰다.
병행하여,
단식 중 NK 세포의 골수 재분포는 이동 매개체 S1PR5와 CXCR4를 요구했다.
이러한 세포들은
IL-12 발현 골수 골수세포(myeloid cells)의 증가된 풀에 의해 프라이밍되어 IFN-γ 생산을 개선했다.
우리의 발견은
식이 제한과 최적화된 고유 면역 반응 간의 연결을 확인하며,
면역요법 전략을 강화할 잠재력을 가진다.
Introduction
Natural killer (NK) cells are circulating cytotoxic lymphocytes that are crucial to host immunity against cancer via the secretion of lytic granules and the production of effector cytokines such as interferon (IFN)-γ. Unlike antigen-specific cytotoxic CD8+ T cells that require tumor antigen presentation by major histocompatibility complex (MHC) molecules, NK cells possess a broad range of germline-encoded activating receptors to target transformed or malignant cells.1 In this way, tumors that have decreased expression of MHC class I to evade T cell killing can be targeted by NK cells.2 Indeed, in clinical studies, the presence of an “NK cell signature” or increase in NK cells within the tumor is predictive of improved survival of patients across a broad range of cancers, including melanoma, breast cancer, and gastric cancer.3,4,5,6 However, the potential to harness NK cells for therapeutic use is limited, as their survival and functionality in the hostile tumor microenvironment (TME) are often suppressed. The TME is hypoxic, rich in lipids and lactic acid, but depleted of glucose, all of which suppress immune cell function and survival and promote tumor growth.7,8 Because NK cells primarily rely on glycolysis for homeostasis and effector functions,9,10,11,12 identifying strategies that reprogram NK cell metabolism to overcome the metabolically immunosuppressive TME is essential to develop novel anti-tumor therapies.
Dietary restriction in the form of fasting or fasting-mimicking diets has garnered significant interest due to its profound effects on metabolism, cancer incidence, and anti-tumor immunity.13 Cyclic fasting impacts tumor cells by inducing sensitivity to chemotherapy and promoting an anti-Warburg effect.14,15 As such, clinical trials implementing these diets alongside various standard-of-care cancer therapies have yielded exciting and promising results, including improved tumor responsiveness to therapy and enhanced immune responses.16,17 Indeed, dietary restriction influences the redistribution and function of immune populations throughout the host, including T cells, B cells, and myeloid cells, and is associated with enhanced immunity.18,19,20,21,22
Here, we examined the impact of dietary restriction on NK cell function. We found that cyclic fasting remodels distinct tissue-specific microenvironments that reprogram the metabolic and functional state of NK cells to enhance anti-tumor immunity and reveal a critical role for NK cells in improved anti-tumor responses induced by fasting. Our findings suggest that NK cell function should be examined in clinical trials employing fasting regimens and that cyclic or periodic fasting may exemplify a dietary strategy for improved innate immunity in the treatment of solid and metastatic cancers.
Introduction
자연 살해(NK) 세포는 순환하는 세포독성 림프구로,
용해 과립 분비와 인터페론(IFN)-γ 같은 효과 인자 사이토카인 생산을 통해
암에 대한 숙주 면역에 중요하다.
종양 항원 제시가 주요 조직 적합성 복합체(MHC) 분자에 의해 요구되는 항원 특이적 세포독성 CD8+ T 세포와 달리, NK 세포는 변형되거나 악성 세포를 타겟으로 하는 광범위한 생식선-인코딩된 활성화 수용체를 보유한다.1 이러한 방식으로, T 세포 살해를 피하기 위해 MHC class I 발현이 감소된 종양은 NK 세포에 의해 타겟될 수 있다.2 실제로, 임상 연구에서 “NK 세포 시그니처”의 존재나 종양 내 NK 세포 증가는 흑색종, 유방암, 위암을 포함한 광범위한 암에서 환자 생존율 향상을 예측한다.3,4,5,6 그러나 NK 세포를 치료적으로 활용할 잠재력은 적대적인 종양 미세환경(TME)에서 NK 세포의 생존과 기능이 종종 억제되기 때문에 제한된다. TME는 저산소, 지질과 젖산이 풍부하지만 포도당은 고갈되어 있으며, 이는 모두 면역 세포 기능과 생존을 억제하고 종양 성장을 촉진한다.7,8 NK 세포가 항상성과 효과 기능에 대해 주로 해당작용(glycolysis)에 의존하기 때문에,9,10,11,12 대사적으로 억제적인 TME를 극복하기 위한 NK 세포 대사 재프로그래밍 전략을 식별하는 것은 새로운 항종양 치료를 개발하는 데 필수적이다. 단식 또는 단식 모방 식이 형태의 식이 제한은 대사, 암 발생, 항종양 면역에 미치는 심오한 영향으로 인해 상당한 관심을 모았다.13 주기적 단식은 화학요법에 대한 민감성을 유발하고 항-와버그 효과를 촉진하여 종양 세포에 영향을 미친다.14,15 따라서, 이러한 식이를 다양한 표준 치료 암 요법과 함께 시행한 임상 시험은 흥미롭고 유망한 결과를 산출했으며, 치료에 대한 종양 반응성 향상과 면역 반응 강화가 포함된다.16,17 실제로, 식이 제한은 T 세포, B 세포, 골수세포를 포함한 숙주 전반의 면역 집단의 재분포와 기능에 영향을 미치며, 향상된 면역과 연관된다.18,19,20,21,22 여기서 우리는 식이 제한이 NK 세포 기능에 미치는 영향을 조사했다. 우리는 주기적 단식이 NK 세포의 대사적 및 기능적 상태를 재프로그래밍하여 항종양 면역을 강화하는 독특한 조직 특이적 미세환경을 재구성한다는 것을 발견했으며, 단식 유발 개선된 항종양 반응에서 NK 세포의 중요한 역할을 드러냈다. 우리의 발견은 단식 요법을 사용하는 임상 시험에서 NK 세포 기능을 검사해야 하며, 주기적 또는 주기적 단식이 고형 및 전이 암 치료에서 개선된 고유 면역을 위한 식이 전략의 예가 될 수 있음을 시사한다.
Results
Cyclic fasting promotes NK cell-mediated anti-tumor immunity
To explore the effects of dietary restriction on NK cell-mediated anti-tumor immune responses, we developed a cyclic fasting diet (CFD) that is implemented for 3 weeks (Figure 1A). This diet was maintained without net weight loss in healthy or in tumor-bearing mice at endpoint when compared with mice fed ad libitum (Figures S1A and S1B). Adherence to a CFD improved the anti-tumor response against MC38 colon carcinoma (Figure 1B) and B16F10 melanoma metastasis (Figure 1C). B16F10 melanoma was measured using lung metastasis counts as well as lung weight as a measurement of tumor burden because, in some cases, the number of metastases was too high to be enumerated accurately. Improved MC38 and B16F10 anti-tumor immunity was dependent on NK cell activity, as depleting NK cells reduced the effects of a CFD on MC38 and B16F10 tumor growth, indicating broad protection of CFD-induced NK cell anti-tumor immunity against both cancers (Figures 1D and 1E). Although many NK cell ligands remained unchanged in the fasted state (Figure S1C), we observed a CFD-mediated decrease in MHC class I expression (Figure 1F), in agreement with the enhanced control of tumors mediated via “missing self” recognition by NK cells. Tumor-infiltrating lymphocyte (TIL) analysis of CFD and ad libitum mice injected with either tumor did not show any major alterations in absolute NK cell numbers (Figures 1G and 1H) or NK cell maturation (Figures S1D and S1E). Further, TIL analysis showed no significant changes in other lymphocyte, myeloid, or dendritic cell (DC) populations in both tumor models (Figures S1F–S1K). We also assessed the functionality of CD8+ T cells, CD4+ T cells, and tumor-associated macrophages (TAMs) and found functional capacity to be relatively unchanged by a CFD (Figures S2A–S2C). However, the CFD did enhance peripheral and tumor-infiltrating NK cell function, as assessed by IFN-γ production and degranulation (Figures 1I, 1J, S2D, and S2E).
Results
주기적 단식은 NK 세포 매개 항종양 면역을 촉진한다 식이 제한이 NK 세포 매개 항종양 면역 반응에 미치는 영향을 탐구하기 위해, 우리는 3주 동안 시행되는 주기적 단식 식이(CFD)를 개발했다 (Figure 1A). 이 식이는 건강한 또는 종양 보유 쥐에서 ad libitum(자유 섭식) 쥐와 비교할 때 종점에서 순 체중 감소 없이 유지되었다 (Figures S1A and S1B). CFD 준수는 MC38 대장암에 대한 항종양 반응을 개선했다 (Figure 1B) 그리고 B16F10 흑색종 전이 (Figure 1C). B16F10 흑색종은 폐 전이 수와 종양 부하 측정으로 폐 무게를 사용해 측정되었으며, 일부 경우 전이 수가 정확히 열거하기 어려울 정도로 많았기 때문이다. 개선된 MC38 및 B16F10 항종양 면역은 NK 세포 활동에 의존적이었으며, NK 세포 고갈 시 CFD가 MC38 및 B16F10 종양 성장에 미치는 효과가 감소했으며, 이는 CFD 유발 NK 세포 항종양 면역의 광범위한 보호를 나타낸다 (Figures 1D and 1E). 단식 상태에서 많은 NK 세포 리간드가 변하지 않았음에도 (Figure S1C), 우리는 CFD 매개 MHC class I 발현 감소를 관찰했으며, 이는 NK 세포에 의한 “결여 자아(missing self)” 인식을 통해 매개된 종양 제어 강화와 일치한다 (Figure 1F). CFD 및 ad libitum 쥐에 주사된 종양의 종양 침윤 림프구(TIL) 분석은 절대 NK 세포 수에 큰 변화를 보이지 않았다 (Figures 1G and 1H) 또는 NK 세포 성숙 (Figures S1D and S1E). 또한, TIL 분석은 두 종양 모델에서 다른 림프구, 골수세포, 또는 수지상 세포(DC) 집단에 유의한 변화를 보이지 않았다 (Figures S1F–S1K). 우리는 또한 CD8+ T 세포, CD4+ T 세포, 종양 관련 대식세포(TAMs)의 기능을 평가했으며, 기능 용량이 CFD에 의해 상대적으로 변하지 않았음을 발견했다 (Figures S2A–S2C). 그러나 CFD는 IFN-γ 생산과 탈과립(degranulation)으로 평가된 주변 및 종양 침윤 NK 세포 기능을 강화했다 (Figures 1I, 1J, S2D, and S2E).

Figure 1 Cyclic fasting promotes NK cell-mediated anti-tumor immunity
Show full captionFigure viewer
Nutrient restriction induces metabolic changes to maintain survival of NK cells
To investigate underlying molecular mechanisms initiating improved NK cell function and tumor control during a CFD, we focused on cellular changes that occur during a singular, acute fasting period. We profiled multiple lymphoid and myeloid cell populations and found that the majority of these cell populations were significantly decreased in the spleen of mice after a 48 h fast (Figure S3A). Because MHC class I expression was altered on tumor cells (Figure 1F), we shifted our focus toward NK cells and their adaptive cytotoxic counterparts, CD8+ T cells (Figures 2A and 2B). Of note, human peripheral blood NK cells were also decreased in this setting of a single acute fast (Figure S3B). Although we observed an increase in cell death in CD8+ T cells, NK cells did not show this same level of cell death at 48 h (Figure 2C). Aligned with this finding, we observed increased levels of apoptosis in CD8+ T cells when compared with NK cells early in the fasting window (0–12 h; Figures S3C and S3D), suggesting NK cells can adapt to changes in nutrient availability by using alternative mechanisms to ensure their survival.

Figure 2 Nutrient restriction induces metabolic changes to maintain survival of NK cells
Show full captionFigure viewer
Acute periods of fasting are known to cause a myriad of systemic metabolic changes that may affect NK cell survival.23 We observed that fasted mice displayed a sharp decline in serum glucose concentrations within 24 h, coinciding with a significant increase in free fatty acid (FA) concentrations (Figure 2D). Because homeostatic maintenance of both innate and adaptive lymphocytes primarily relies on glucose to fuel basal levels of oxidative phosphorylation (OXPHOS),9,24 we performed Seahorse metabolic flux assays on NK cells and total CD8+ T cells from ad libitum or fasted mice. Although both CD8+ T cells and NK cells showed a marked reduction in glycolysis and glycolytic capacity (Figures S3E and S3F), only fasted NK cells were surprisingly able to maintain their capacity for OXPHOS (Figures 2E and 2F). Alternative metabolic substrates, including FAs, are also known to fuel OXPHOS,25 and NK cells and CD8+ T cells from fasted mice showed increased uptake of FAs (Figures 2G and 2H). At the same time, NK cells from fasted mice also displayed a more prominent reduction in neutral lipids compared with CD8+ T cells (Figures 2I and S3G), indicating that increased FA uptake did not result in a net increase in lipid storage within lipid droplets (LDs). This decrease in neutral lipids may also suggest that the liberation of stored FAs can be used to fuel OXPHOS,26 and, as such, we observed increased autophagy in NK cells but not CD8+ T cells in the fasted state (Figures 2J and S3H). To further strengthen our findings, we inhibited FA oxidation and observed that NK cells from fasted mice were unable to maintain OXPHOS compared with ad libitum-fed controls (Figures 2K and S3I). Altogether, these data indicate that NK cells utilize FAs as an energy source to fuel OXPHOS during times of glucose withdrawal, ensuring their survival.
Cyclic fasting metabolically reprograms NK cells
Because NK cells showed a remarkable capacity to switch to FA use during fasting, we further investigated whether NK cells from mice that had undergone a CFD showed similar changes. We found that splenic NK cells from mice subjected to a CFD displayed an increased capacity to uptake FAs from the local environment (Figure 3A) but did not show an accumulation of lipids (Figure 3B). This coincided with a slight decrease in glucose uptake (Figure S4A). To gain insight into how this metabolic switch affects the transcriptomic profile of fasted NK cells, we performed single-cell RNA sequencing (scRNA-seq) on NK cells from tumor-bearing mice subjected to either a CFD or ad libitum feeding. Uniform manifold approximation and projection (UMAP) embedding suggested that fasted NK cells were distinct from their ad libitum-fed counterparts (Figure 3C). We then used unbiased louvain clustering and identified 9 distinct clusters shared between ad libitum and CFD NK cells (Figure S4B). Among the 9 clusters, clusters 0 and 4 were enriched for NK cells from CFD mice (Figure S4C). Gene set enrichment analysis (GSEA) performed on differential gene expression identified by MAST between ad libitum and CFD NK cells showed an upregulation of FA metabolism genes in CFD NK cells (Figure 3D), which may be primarily driven by CFD-enriched clusters, consistent with our previous observations (Figures S4B–S4D). Select genes known to be critical for FA metabolism were upregulated in CFD NK cells, including Cpt1a, encoding the critical enzyme that drives FA metabolism (Figure 3E). We also observed a decrease in the expression of Ldha, an important gene required to maintain glycolysis (Figure S3E). Together, these data suggest that a CFD pushes splenic NK cells toward a reprogrammed metabolic state involving increased FA metabolism and decreased glycolysis.

Figure 3 Cyclic fasting metabolically reprograms NK cells
Show full captionFigure viewer
Interestingly, in addition to FA metabolism, some of the most significantly upregulated differentially expressed genes in CFD NK cells were genes known to be induced by glucocorticoids (GCs; Figure 3F). This increase in GC receptor (GR) target gene expression (Figure 3G) coincided with a spike in GC concentration at both 24 and 48 h in the spleen and serum of fasted mice compared with those that were fed ad libitum (Figure 3H). GCs can induce FA metabolism in cardiomyocytes and acute lymphoblastic leukemia27,28; however, whether this occurs in immune cells has not been established. We treated mouse and human NK cells with the synthetic GC, dexamethasone (Dex), and found that a 24 h treatment of NK cells from both species with Dex increased CPT1A expression (Figures 3I and S4F). The induction was evident as early as 2 h (Figure S4G), demonstrating a rapid and sustained induction of CPT1A by GCs. Taken together, these data suggest that increased GC levels during periods of fasting drive expression of CPT1A, inducing FA oxidation in NK cells and thus promoting optimal metabolism depending on specific fuel availability.
CFD-induced FA oxidation improves the NK cell anti-tumor response
The TME can promote tumor growth while dampening TIL functions by limiting glucose, a metabolite required for optimal T cell anti-tumor function,29 and enriching for immunosuppressive lipids.30 Because NK cells from mice subjected to a CFD showed increased expression of CPT1A, we hypothesized that a preference for using FAs instead of glucose within the TME could improve NK cell function. Indeed, tumor-infiltrating NK cells from CFD mice showed increased CPT1A levels and increased FA uptake when compared with ad libitum-fed mice (Figures 4A and 4B). To study the effects of Cpt1a deletion on NK cells in vivo, we used Cpt1aflox/flox mice31 crossed with Ncr1iCre mice to generate mice that harbor an NK cell-specific deletion of CPT1A (NK-Cpt1a−/−; Figure S5A).32 CPT1A is required for the uptake of long-chain FAs into the mitochondria for FA oxidation (Figure 4C), and, as such, we measured the effects of a CFD on FA oxidation and anti-tumor function. NK-Cpt1a−/− and wild-type (WT) NK-Cpt1a+/+ littermate controls were challenged with tumors and subjected to either ad libitum or CFD conditions. Remarkably, NK cells deficient in Cpt1a were not able to control tumor growth in the CFD setting when compared with Cpt1a-sufficient NK cells (Figures 4D and S5B). The striking difference in tumor volume was associated with a significant decrease in the number of intra-tumoral Cpt1a−/− NK cells when compared with Cpt1a+/+ NK cells in CFD mice (Figures 4E and S5C). This reduction was linked to diminished expression of the anti-apoptotic protein BCL2 when compared with NK-Cpt1a+/+ controls (Figure 4F). Expression of the pro-apoptotic protein BIM was maintained (Figure 4G), increasing the BIM:BCL2 ratio and indicating that the balance of these proteins tipped in favor of cell death within NK cells that lack Cpt1a (Figure 4H). Tumor-infiltrating Cpt1a-deficient NK cells showed similar levels of FA uptake compared with control NK cells (Figure 4I), suggesting that although Cpt1a-deficient NK cells cannot perform mitochondrial FA oxidation, FA uptake is not disrupted. However, Cpt1a-deficient NK cells did show a marked reduction in effector function during a CFD (Figures 4J and S5D), suggesting that an inability to efficiently process intracellular FAs during fasting hampers NK cell survival and anti-tumor function, disrupting NK cell-mediated control of metastatic disease.

Figure 4 CFD-induced FA oxidation improves NK cell anti-tumor immunity
Show full captionFigure viewer
S1PR5 and CXCR4 govern redistribution of NK cells to the BM during fasting
Because acute periods of fasting resulted in decreased NK cell numbers in the spleen and blood without any significant changes in cell death (Figures 2A–2C and S3C), we reasoned that they could be accumulating in other organs in this setting (Figure S5E). Indeed, a significant increase in NK cells was observed in the bone marrow (BM) after 48 h of fasting (Figure 5A). The accumulation of NK cells in the BM during fasting coincided with a loss in blood and peripheral tissues and was followed by a restoration of NK cell numbers within 24 h of unrestricted refeeding (Figure 5B). These rapid and fluid changes suggested that a proportion of peripheral NK cells were redistributed to the BM during periods of fasting. To confirm this, we transferred mature CD45.1-expressing splenic NK cells into CD45.2-expressing recipient mice that were either fasted for 48 h or fed ad libitum. The transferred NK cells were recovered at reduced numbers in the spleen and blood and increased numbers in the BM (Figure 5C). In addition, we used the iNkp46tdTomato mouse model to “time stamp” NK cells by inducing expression of tdTomato (Tom+), permanently marking mature peripheral NK cells.33 Similar to the adoptive transfer experiment, Tom+ NK cells were redistributed to the BM during periods of fasting (Figure 5D). Together, these data support that while a population of NK cells remains in peripheral organs such as the spleen during fasting, some mature NK cells are preferentially redistributed to the BM.

Figure 5 Fasting promotes NK cell redistribution to the BM
Show full captionFigure viewer
In contrast to the metabolically reprogrammed NK cells recovered from the spleen of mice subjected to 48 h of fasting, NK cells from the BM showed a reduction in the uptake of FAs and CPT1A expression (Figures 5E and 5F). These changes coincided with reduced levels of GCs in the BM compared with serum and splenic levels when compared with ad libitum-fed controls (Figure 5G). Similarly, NK cells from the BM of mice subjected to a CFD showed reduced FA uptake but maintained glucose uptake (Figures S5F and S5G). The BM has previously been suggested as a niche that supports lymphocyte survival during periods of nutrient deprivation,18,19,20 and, altogether, our data suggest that the tissue-specific localization of NK cells during fasting may have alternate effects on metabolic and functional capacity.
To understand the consequences of blocking entry into the BM niche during a CFD, we determined which signal(s) were driving NK cells into the BM during periods of fasting. We observed increased numbers of Ter119-expressing red blood cells (RBCs) in the BM during fasting compared with ad libitum-fed controls (Figure S6A). Increased RBCs in the BM are a phenomenon previously reported during dietary restriction and may be a consequence of increased GCs in the periphery.18,34,35 RBCs are a major source of sphingosine-1-phosphate (S1P), a molecule important for lymphocyte trafficking.36,37 NK cells express high levels of S1PR5, a receptor required for efficient NK cell trafficking through lymph nodes and the BM.38,39 We used a knockin S1pr5eGFP-T2A-iCre reporter mouse40 and found that S1pr5eGFP expression increased in the BM over 48 h of fasting (Figure 6A), suggesting that an increase of RBCs might also increase the local S1P concentration in the BM.

Figure 6 Redistribution of NK cells to the BM during a CFD promotes tumor control
Show full captionFigure viewer
We compared NK cell numbers in the BM of reconstituted S1pr5+/+ and S1pr5−/− mixed BM chimeras (Figure S6B) that had been subjected to 48 h fasting and found that fasting-mediated NK cell accumulation in the BM was reduced in the absence of S1PR5 (Figure 6B). Because S1pr5−/− mice have increased numbers of NK cells in the BM under ad libitum conditions, the differences between fasted and ad libitum-fed mice were compared within genotypes and presented as fold-change (fasted/average in ad libitum). An increase in mature CD27−CD11b+ NK cells was observed in the BM after fasting, and S1pr5eGFP expression was also significantly increased in this population (Figures S6C and S6D). Deletion of S1PR5 selectively limited the entry of CD27−CD11b+ NK cells into the BM after 48 h of fasting (Figure 6C). To ensure that the loss of cells in the BM was due to defective trafficking, we transferred equal numbers of WT and S1pr5−/− NK cells into recipient mice (Figure S6E) that were either fasted for 48 h or fed ad libitum and found reduced numbers of S1pr5−/− NK cells in the BM compared with S1PR5-expressing WT NK cells (Figure S6F), altogether suggesting the S1P-S1PR5 axis is required for CFD-mediated NK cell migration into the BM during fasting.
Because NK cells and other lymphocytes enter and exit the BM through sinusoids that are connected to blood circulation,41,42 we incorporated an intravenous CD45 labeling technique43 to discriminate between sinusoid- and parenchyma-resident cells. CD45 labeling revealed a stepwise increase in parenchymal NK cells and a concomitant reduction in sinusoidal NK cells within 48 h of fasting (Figures 6D and S6G). Given that CXCR4 expression is tightly linked to NK cell retention in the parenchyma,41 we hypothesized that CXCR4 may also be integral to BM entry/exit during fasting. We compared the fold-change increase in BM NK cells from fasted mice treated with or without the CXCR4 antagonist, AMD3100, to ad libitum-fed mice and found a reduced capacity for NK cells to enter the BM (Figure 6E). Altogether, these data suggest that BM entry during fasting is mediated by an interplay of CXCR4-mediated parenchymal retention and S1P-guided NK cell accumulation.
Redistribution of NK cells to the BM during CFD promotes tumor control
To determine the effects of blocking NK cell entry into the BM on CFD-mediated tumor control, we challenged either S1pr5−/− mice or mice treated with AMD3100 with tumor cells and subjected them to either a CFD or ad libitum feeding (Figures S6H and S6I). Blocking fasting-induced BM entry of NK cells via either method abrogated CFD-mediated anti-tumor control (Figures 6F, 6G, and S6J–S6L). We next profiled the tumors of S1pr5−/− mice or mice treated with AMD3100 subjected to a CFD and found no differences in the total numbers of NK cells found in the TME (Figures 6H, 6I, and S6M). Furthermore, NK cells from the tumor and spleen no longer possessed CFD-mediated differences in IFN-γ production (Figures 6J, 6K, and S6N). Altogether, these data suggest that BM entry may promote the CFD-facilitated anti-tumor effector function observed in NK cells.
The fasted BM niche promotes NK cell priming via IL-12 production
To understand why NK cell redistribution to the BM was important for CFD-mediated anti-tumor responses, we performed scRNA-seq on NK cells from the BM of mice that were subjected to either CFD or ad libitum feeding (Figure S7A). NK cells were partially separated based on whether they had been subjected to a CFD or ad libitum feeding using UMAP projections (Figure 7A). We found several interleukin (IL)-12 pathway genes, such as Stat4, upregulated in NK cells from the BM of CFD mice (Figure 7B). We also observed upregulation of STAT4 signaling components JAK2 and TYK2, and a number of transcription factors that are induced by STAT444 (Figures S7B and S7C). Coinciding with the induction of IL-12 and STAT4 target genes in NK cells, we found increased IL-12 concentrations in the BM after 48 h fasting and reduction in the spleen and serum (Figure 7C). This coincided with an increase in IL-12-producing cells in the BM compared with the spleen (Figures 7D and S7D). IL-12+ cells in the BM expressed CD11b (Figures S7E and S7F) and were negative for lymphoid lineage markers CD19, CD3, TCRβ, and NK1.1 (Figure S7G), suggesting that the cell population is of myeloid origin. Because IL-12 is a potent stimulator of IFN-γ secretion in NK cells,45 we determined whether the increase in BM IL-12 during fasting could functionally prime NK cells. Indeed, NK cells from the BM of CFD mice showed a significant increase in IFN-γ production (Figure 7E). Furthermore, NK cells from the spleen of CFD mice that had been refed (thus redistributing NK cells that were previously in the BM back to the spleen) also secreted more IFN-γ (Figure 7F).

Figure 7 The fasted BM niche promotes NK cell priming via IL-12 production
Show full captionFigure viewer
To assess whether a link between fatty acid oxidation and IL-12 priming exists, we tested whether NK cells lacking fatty acid oxidation capacity still showed BM-specific reprogramming. After 48 h of fasting, we found NK-Cpt1a−/− NK cells redistributed to the BM in a similar manner to WT NK-Cpt1a+/+ NK cells (Figure S7H). In agreement with intact redistribution during fasting, NK cells from NK-Cpt1a−/− mice subjected to a CFD showed similar increases in IFN-γ production in the BM when compared with ad libitum-fed controls (Figure S7I). These findings suggest that the inability of NK cells in the BM to perform fatty acid oxidation does not impair IL-12 priming and subsequent IFN-γ production. Altogether, these data suggest that, during periods of fasting, the BM niche is specifically reshaped to provide increased local concentrations of IL-12, priming NK cells that have preferentially redistributed to the BM and inducing local and peripheral IFN-γ secretion by NK cells important for anti-tumor immunity.
Discussion
Dietary restriction in the form of fasting and limiting caloric intake redistributes both adaptive and innate immune cell subsets such as B cells, T cells, and myeloid cells.18,19,20 Because NK cells are often placed at the intersection of innate and adaptive immunity, they represent a unique cell type that possesses features of both.46 Unlike adaptive T and B cells, NK cells act as a first line of defense against infections and tumor metastasis, and unlike innate myeloid cells, which can become detrimental in the TME as myeloid-derived suppressor cells, NK cell presence within tumors is almost always associated with better prognosis.6,47,48 Here, we show evidence that tissue repositioning during periods of fasting is advantageous for peripheral NK cell function, poising them for enhanced metabolic status and survival in the TME.
A significant advantage that NK cells have over T cells in the context of fasting is their response to GCs. Unlike T cells, which are sensitive to high levels of endogenous GCs that cause excessive cell death,18,49 we found that NK cells did not undergo greater death in response to GC exposure in vivo and in vitro. Instead, GCs promoted upregulation of CPT1A for increased FA oxidation capability, a phenomenon also observed in cardiomyocytes and muscle.27,50 Like M2 macrophages, NK cells increase lipid uptake, OXPHOS, and FA oxidation.51 Their ability to metabolically adapt in conditions of high GCs and excess FAs may be a contributing factor that distinguishes the innate functionality of NK cells from T or B cells in fasting conditions.
Metabolic rewiring in several cell types can be induced by nutrient restriction, shifting cellular metabolism from reliance on glycolysis to dependence on mitochondrial FA oxidation.26 However, this specific adaptation has not been mechanistically characterized in NK cells. Although FA oxidation was recently shown to be crucial for NK cell antiviral responses,32 much of our understanding of lipid uptake has been in the context of obesity and NK cell dysfunction, and thus the use of FAs as an energy source during cyclic fasting to improve functional responses against cancer has not been previously appreciated. In obese individuals, NK cells show an accumulation of lipids, which is correlated with reduced functionality.52 Similarly, in a mouse model of lymphoma, NK cell dysfunction is correlated with lipid accumulation and increased lipid metabolism.53 Unlike these previous studies, our work shows that cyclic fasting results in increased NK cell FA oxidation without lipid accumulation, suggesting that fasting might be an optimal approach to utilize excess lipids.
Both the obesity and lymphoma environments raise the interesting observation that NK cells in these conditions are continuously exposed to lipid-rich environments. By contrast, our model of a CFD, where a proportion of NK cells are acutely and intermittently exposed to high concentrations of FAs, represents a scenario where NK cells are constantly being shifted from glucose-depleted, FA-rich environments back to baseline during each period of fasting/re-feeding. Whether cyclic fasting can improve NK cell function in the continuously lipid-laden environments found in obese mice or humans requires further investigation. Of note, many prior and current studies, including our own here, do not assess NK cell function and FA oxidation capacity in adipose tissue. Our recent work suggests that NK cells have the ability to conditionally alter their metabolism based on the tissue in which they reside54; thus, future studies will seek to understand such additional tissue-specific roles of fasting, particularly because adipose-residing innate lymphocytes have metabolic and functional adaptations in obesity-induced disease settings.55,56,57,58
Acute myeloid leukemia (AML) is an aggressive hematological malignancy where improved outcomes are strongly associated with increased NK cell cytolytic activity.59,60,61 As such, adoptive NK cell therapies in the clinic have primarily been targeted to AML.8 Thus, whether AML maintains a lipid-rich BM environment similar to lymphoma is of interest, as the BM can also represent a site of tumor growth. In AML, the effects of cyclic fasting may be opposing. On the one hand, NK cells are preferentially redistributed to the BM during CFD conditions, where they are primed to produce more IFN-γ, which could promote leukemia tumor control. On the other hand (and as we have also observed in our study), cyclic fasting does not appear to increase FA uptake or LD abundance in NK cells in the BM, even after multiple cycles of fasting. Because our study does not address cancers of BM origin, future studies will investigate how fasting-induced NK cell responses to leukemias may differ from the solid tumor and metastasis models used in this study.
We observed that cyclic fasting improved NK cell functionality in a tissue-specific manner via parallel but disparate processes occurring in the BM versus the spleen. In mice, the half-life of an NK cell is ∼19.4 days, and thus adherence to 3 weeks of a CFD would assume that the NK cells profiled at tumor endpoint are heterogeneous in age33,62 and certainly harboring a proportion of cells that have cycled through more than one round of fasting and refeeding. Although we showed that NK cells are redistributed from the spleen and blood to the BM, we recognize that there are technological limitations with the current tools available to determine whether the same NK cell population has preferentially redistributed to the BM during each fasting cycle and, thus, whether an individual NK cell has been exposed to one or both tissue-specific processes. We provide evidence in our study that disrupting either process (i.e., FA oxidation via CPT1A deletion or BM trafficking via S1PR5 deletion or CXCR4 antagonism) has profound consequences on NK cell anti-tumor function, emphasizing the contribution of both BM entry and splenic reprogramming. Future cell-tracing studies may be able to determine whether NK cells found in the tumor have experienced fasting-induced signals from one or both tissue sites.
IL-12-producing CD45+CD11b+ cells were increased in the BM after adherence to cyclic fasting, suggesting that the effects of fasting can impose indirect consequences on NK cells. Although we have not established which subset of CD11b-expressing cells is producing IL-12, we can speculate that it may be derived from a subgroup of the myeloid lineage, as DCs and macrophages are potent producers of IL-12 and express CD11b. The BM is a complex tissue as it not only contains these subgroups but also their progenitors and a complex set of downstream putative populations. However, whether dietary restriction directly redistributes macrophages or DCs into the BM is still unknown, as most dietary studies have focused on the relocation and function of adipose tissue macrophages, suggesting that they accumulate in areas with increased lipids.63,64 Dietary restriction increases adipogenesis in the BM of mice18,65; thus, it is interesting to consider whether this increased adipogenesis may be responsible for an induction of IL-12-producing macrophages. Furthermore, a potential role of inflammatory macrophages is to metabolize lipids63; thus, increased IL-12-producing macrophages may be associated with increased lipid content within the BM. Additionally, fasting can modulate the gut bacteria flora and enrich for lactobacillus species,66 some strains of which can induce IL-12 production from macrophages.67,68,69 Because changes in gut microbiota can influence BM production of inflammatory cell types,70 we can speculate that these alterations may be responsible for the increased IL-12 production we observe in our model.
Altogether, our work recognizes a critical role for the innate immune system in the improved anti-tumor response induced by fasting. This work will be important in dissecting the immune landscape in patients from clinical trials using fasting and fasting-mimicking diets, given that NK cell infiltration and function have yet to be fully appreciated. Furthermore, we show that cyclic fasting can remodel different tissues and thus represents a novel method of improving tissue-specific NK cell function and enhancing innate immunity for the treatment of solid and metastatic cancers. Introduction
Natural killer (NK) cells are circulating cytotoxic lymphocytes that are crucial to host immunity against cancer via the secretion of lytic granules and the production of effector cytokines such as interferon (IFN)-γ. Unlike antigen-specific cytotoxic CD8+ T cells that require tumor antigen presentation by major histocompatibility complex (MHC) molecules, NK cells possess a broad range of germline-encoded activating receptors to target transformed or malignant cells.1 In this way, tumors that have decreased expression of MHC class I to evade T cell killing can be targeted by NK cells.2 Indeed, in clinical studies, the presence of an “NK cell signature” or increase in NK cells within the tumor is predictive of improved survival of patients across a broad range of cancers, including melanoma, breast cancer, and gastric cancer.3,4,5,6 However, the potential to harness NK cells for therapeutic use is limited, as their survival and functionality in the hostile tumor microenvironment (TME) are often suppressed. The TME is hypoxic, rich in lipids and lactic acid, but depleted of glucose, all of which suppress immune cell function and survival and promote tumor growth.7,8 Because NK cells primarily rely on glycolysis for homeostasis and effector functions,9,10,11,12 identifying strategies that reprogram NK cell metabolism to overcome the metabolically immunosuppressive TME is essential to develop novel anti-tumor therapies.
Dietary restriction in the form of fasting or fasting-mimicking diets has garnered significant interest due to its profound effects on metabolism, cancer incidence, and anti-tumor immunity.13 Cyclic fasting impacts tumor cells by inducing sensitivity to chemotherapy and promoting an anti-Warburg effect.14,15 As such, clinical trials implementing these diets alongside various standard-of-care cancer therapies have yielded exciting and promising results, including improved tumor responsiveness to therapy and enhanced immune responses.16,17 Indeed, dietary restriction influences the redistribution and function of immune populations throughout the host, including T cells, B cells, and myeloid cells, and is associated with enhanced immunity.18,19,20,21,22
Here, we examined the impact of dietary restriction on NK cell function. We found that cyclic fasting remodels distinct tissue-specific microenvironments that reprogram the metabolic and functional state of NK cells to enhance anti-tumor immunity and reveal a critical role for NK cells in improved anti-tumor responses induced by fasting. Our findings suggest that NK cell function should be examined in clinical trials employing fasting regimens and that cyclic or periodic fasting may exemplify a dietary strategy for improved innate immunity in the treatment of solid and metastatic cancers.
ResultsCyclic fasting promotes NK cell-mediated anti-tumor immunity
To explore the effects of dietary restriction on NK cell-mediated anti-tumor immune responses, we developed a cyclic fasting diet (CFD) that is implemented for 3 weeks (Figure 1A). This diet was maintained without net weight loss in healthy or in tumor-bearing mice at endpoint when compared with mice fed ad libitum (Figures S1A and S1B). Adherence to a CFD improved the anti-tumor response against MC38 colon carcinoma (Figure 1B) and B16F10 melanoma metastasis (Figure 1C). B16F10 melanoma was measured using lung metastasis counts as well as lung weight as a measurement of tumor burden because, in some cases, the number of metastases was too high to be enumerated accurately. Improved MC38 and B16F10 anti-tumor immunity was dependent on NK cell activity, as depleting NK cells reduced the effects of a CFD on MC38 and B16F10 tumor growth, indicating broad protection of CFD-induced NK cell anti-tumor immunity against both cancers (Figures 1D and 1E). Although many NK cell ligands remained unchanged in the fasted state (Figure S1C), we observed a CFD-mediated decrease in MHC class I expression (Figure 1F), in agreement with the enhanced control of tumors mediated via “missing self” recognition by NK cells. Tumor-infiltrating lymphocyte (TIL) analysis of CFD and ad libitum mice injected with either tumor did not show any major alterations in absolute NK cell numbers (Figures 1G and 1H) or NK cell maturation (Figures S1D and S1E). Further, TIL analysis showed no significant changes in other lymphocyte, myeloid, or dendritic cell (DC) populations in both tumor models (Figures S1F–S1K). We also assessed the functionality of CD8+ T cells, CD4+ T cells, and tumor-associated macrophages (TAMs) and found functional capacity to be relatively unchanged by a CFD (Figures S2A–S2C). However, the CFD did enhance peripheral and tumor-infiltrating NK cell function, as assessed by IFN-γ production and degranulation (Figures 1I, 1J, S2D, and S2E).

Figure 1 Cyclic fasting promotes NK cell-mediated anti-tumor immunity
Show full captionFigure viewer
Nutrient restriction induces metabolic changes to maintain survival of NK cells
To investigate underlying molecular mechanisms initiating improved NK cell function and tumor control during a CFD, we focused on cellular changes that occur during a singular, acute fasting period. We profiled multiple lymphoid and myeloid cell populations and found that the majority of these cell populations were significantly decreased in the spleen of mice after a 48 h fast (Figure S3A). Because MHC class I expression was altered on tumor cells (Figure 1F), we shifted our focus toward NK cells and their adaptive cytotoxic counterparts, CD8+ T cells (Figures 2A and 2B). Of note, human peripheral blood NK cells were also decreased in this setting of a single acute fast (Figure S3B). Although we observed an increase in cell death in CD8+ T cells, NK cells did not show this same level of cell death at 48 h (Figure 2C). Aligned with this finding, we observed increased levels of apoptosis in CD8+ T cells when compared with NK cells early in the fasting window (0–12 h; Figures S3C and S3D), suggesting NK cells can adapt to changes in nutrient availability by using alternative mechanisms to ensure their survival.

Figure 2 Nutrient restriction induces metabolic changes to maintain survival of NK cells
Show full captionFigure viewer
Acute periods of fasting are known to cause a myriad of systemic metabolic changes that may affect NK cell survival.23 We observed that fasted mice displayed a sharp decline in serum glucose concentrations within 24 h, coinciding with a significant increase in free fatty acid (FA) concentrations (Figure 2D). Because homeostatic maintenance of both innate and adaptive lymphocytes primarily relies on glucose to fuel basal levels of oxidative phosphorylation (OXPHOS),9,24 we performed Seahorse metabolic flux assays on NK cells and total CD8+ T cells from ad libitum or fasted mice. Although both CD8+ T cells and NK cells showed a marked reduction in glycolysis and glycolytic capacity (Figures S3E and S3F), only fasted NK cells were surprisingly able to maintain their capacity for OXPHOS (Figures 2E and 2F). Alternative metabolic substrates, including FAs, are also known to fuel OXPHOS,25 and NK cells and CD8+ T cells from fasted mice showed increased uptake of FAs (Figures 2G and 2H). At the same time, NK cells from fasted mice also displayed a more prominent reduction in neutral lipids compared with CD8+ T cells (Figures 2I and S3G), indicating that increased FA uptake did not result in a net increase in lipid storage within lipid droplets (LDs). This decrease in neutral lipids may also suggest that the liberation of stored FAs can be used to fuel OXPHOS,26 and, as such, we observed increased autophagy in NK cells but not CD8+ T cells in the fasted state (Figures 2J and S3H). To further strengthen our findings, we inhibited FA oxidation and observed that NK cells from fasted mice were unable to maintain OXPHOS compared with ad libitum-fed controls (Figures 2K and S3I). Altogether, these data indicate that NK cells utilize FAs as an energy source to fuel OXPHOS during times of glucose withdrawal, ensuring their survival.
Cyclic fasting metabolically reprograms NK cells
Because NK cells showed a remarkable capacity to switch to FA use during fasting, we further investigated whether NK cells from mice that had undergone a CFD showed similar changes. We found that splenic NK cells from mice subjected to a CFD displayed an increased capacity to uptake FAs from the local environment (Figure 3A) but did not show an accumulation of lipids (Figure 3B). This coincided with a slight decrease in glucose uptake (Figure S4A). To gain insight into how this metabolic switch affects the transcriptomic profile of fasted NK cells, we performed single-cell RNA sequencing (scRNA-seq) on NK cells from tumor-bearing mice subjected to either a CFD or ad libitum feeding. Uniform manifold approximation and projection (UMAP) embedding suggested that fasted NK cells were distinct from their ad libitum-fed counterparts (Figure 3C). We then used unbiased louvain clustering and identified 9 distinct clusters shared between ad libitum and CFD NK cells (Figure S4B). Among the 9 clusters, clusters 0 and 4 were enriched for NK cells from CFD mice (Figure S4C). Gene set enrichment analysis (GSEA) performed on differential gene expression identified by MAST between ad libitum and CFD NK cells showed an upregulation of FA metabolism genes in CFD NK cells (Figure 3D), which may be primarily driven by CFD-enriched clusters, consistent with our previous observations (Figures S4B–S4D). Select genes known to be critical for FA metabolism were upregulated in CFD NK cells, including Cpt1a, encoding the critical enzyme that drives FA metabolism (Figure 3E). We also observed a decrease in the expression of Ldha, an important gene required to maintain glycolysis (Figure S3E). Together, these data suggest that a CFD pushes splenic NK cells toward a reprogrammed metabolic state involving increased FA metabolism and decreased glycolysis.

Figure 3 Cyclic fasting metabolically reprograms NK cells
Show full captionFigure viewer
Interestingly, in addition to FA metabolism, some of the most significantly upregulated differentially expressed genes in CFD NK cells were genes known to be induced by glucocorticoids (GCs; Figure 3F). This increase in GC receptor (GR) target gene expression (Figure 3G) coincided with a spike in GC concentration at both 24 and 48 h in the spleen and serum of fasted mice compared with those that were fed ad libitum (Figure 3H). GCs can induce FA metabolism in cardiomyocytes and acute lymphoblastic leukemia27,28; however, whether this occurs in immune cells has not been established. We treated mouse and human NK cells with the synthetic GC, dexamethasone (Dex), and found that a 24 h treatment of NK cells from both species with Dex increased CPT1A expression (Figures 3I and S4F). The induction was evident as early as 2 h (Figure S4G), demonstrating a rapid and sustained induction of CPT1A by GCs. Taken together, these data suggest that increased GC levels during periods of fasting drive expression of CPT1A, inducing FA oxidation in NK cells and thus promoting optimal metabolism depending on specific fuel availability.
CFD-induced FA oxidation improves the NK cell anti-tumor response
The TME can promote tumor growth while dampening TIL functions by limiting glucose, a metabolite required for optimal T cell anti-tumor function,29 and enriching for immunosuppressive lipids.30 Because NK cells from mice subjected to a CFD showed increased expression of CPT1A, we hypothesized that a preference for using FAs instead of glucose within the TME could improve NK cell function. Indeed, tumor-infiltrating NK cells from CFD mice showed increased CPT1A levels and increased FA uptake when compared with ad libitum-fed mice (Figures 4A and 4B). To study the effects of Cpt1a deletion on NK cells in vivo, we used Cpt1aflox/flox mice31 crossed with Ncr1iCre mice to generate mice that harbor an NK cell-specific deletion of CPT1A (NK-Cpt1a−/−; Figure S5A).32 CPT1A is required for the uptake of long-chain FAs into the mitochondria for FA oxidation (Figure 4C), and, as such, we measured the effects of a CFD on FA oxidation and anti-tumor function. NK-Cpt1a−/− and wild-type (WT) NK-Cpt1a+/+ littermate controls were challenged with tumors and subjected to either ad libitum or CFD conditions. Remarkably, NK cells deficient in Cpt1a were not able to control tumor growth in the CFD setting when compared with Cpt1a-sufficient NK cells (Figures 4D and S5B). The striking difference in tumor volume was associated with a significant decrease in the number of intra-tumoral Cpt1a−/− NK cells when compared with Cpt1a+/+ NK cells in CFD mice (Figures 4E and S5C). This reduction was linked to diminished expression of the anti-apoptotic protein BCL2 when compared with NK-Cpt1a+/+ controls (Figure 4F). Expression of the pro-apoptotic protein BIM was maintained (Figure 4G), increasing the BIM:BCL2 ratio and indicating that the balance of these proteins tipped in favor of cell death within NK cells that lack Cpt1a (Figure 4H). Tumor-infiltrating Cpt1a-deficient NK cells showed similar levels of FA uptake compared with control NK cells (Figure 4I), suggesting that although Cpt1a-deficient NK cells cannot perform mitochondrial FA oxidation, FA uptake is not disrupted. However, Cpt1a-deficient NK cells did show a marked reduction in effector function during a CFD (Figures 4J and S5D), suggesting that an inability to efficiently process intracellular FAs during fasting hampers NK cell survival and anti-tumor function, disrupting NK cell-mediated control of metastatic disease.

Figure 4 CFD-induced FA oxidation improves NK cell anti-tumor immunity
Show full captionFigure viewer
S1PR5 and CXCR4 govern redistribution of NK cells to the BM during fasting
Because acute periods of fasting resulted in decreased NK cell numbers in the spleen and blood without any significant changes in cell death (Figures 2A–2C and S3C), we reasoned that they could be accumulating in other organs in this setting (Figure S5E). Indeed, a significant increase in NK cells was observed in the bone marrow (BM) after 48 h of fasting (Figure 5A). The accumulation of NK cells in the BM during fasting coincided with a loss in blood and peripheral tissues and was followed by a restoration of NK cell numbers within 24 h of unrestricted refeeding (Figure 5B). These rapid and fluid changes suggested that a proportion of peripheral NK cells were redistributed to the BM during periods of fasting. To confirm this, we transferred mature CD45.1-expressing splenic NK cells into CD45.2-expressing recipient mice that were either fasted for 48 h or fed ad libitum. The transferred NK cells were recovered at reduced numbers in the spleen and blood and increased numbers in the BM (Figure 5C). In addition, we used the iNkp46tdTomato mouse model to “time stamp” NK cells by inducing expression of tdTomato (Tom+), permanently marking mature peripheral NK cells.33 Similar to the adoptive transfer experiment, Tom+ NK cells were redistributed to the BM during periods of fasting (Figure 5D). Together, these data support that while a population of NK cells remains in peripheral organs such as the spleen during fasting, some mature NK cells are preferentially redistributed to the BM.

Figure 5 Fasting promotes NK cell redistribution to the BM
Show full captionFigure viewer
In contrast to the metabolically reprogrammed NK cells recovered from the spleen of mice subjected to 48 h of fasting, NK cells from the BM showed a reduction in the uptake of FAs and CPT1A expression (Figures 5E and 5F). These changes coincided with reduced levels of GCs in the BM compared with serum and splenic levels when compared with ad libitum-fed controls (Figure 5G). Similarly, NK cells from the BM of mice subjected to a CFD showed reduced FA uptake but maintained glucose uptake (Figures S5F and S5G). The BM has previously been suggested as a niche that supports lymphocyte survival during periods of nutrient deprivation,18,19,20 and, altogether, our data suggest that the tissue-specific localization of NK cells during fasting may have alternate effects on metabolic and functional capacity.
To understand the consequences of blocking entry into the BM niche during a CFD, we determined which signal(s) were driving NK cells into the BM during periods of fasting. We observed increased numbers of Ter119-expressing red blood cells (RBCs) in the BM during fasting compared with ad libitum-fed controls (Figure S6A). Increased RBCs in the BM are a phenomenon previously reported during dietary restriction and may be a consequence of increased GCs in the periphery.18,34,35 RBCs are a major source of sphingosine-1-phosphate (S1P), a molecule important for lymphocyte trafficking.36,37 NK cells express high levels of S1PR5, a receptor required for efficient NK cell trafficking through lymph nodes and the BM.38,39 We used a knockin S1pr5eGFP-T2A-iCre reporter mouse40 and found that S1pr5eGFP expression increased in the BM over 48 h of fasting (Figure 6A), suggesting that an increase of RBCs might also increase the local S1P concentration in the BM.

Figure 6 Redistribution of NK cells to the BM during a CFD promotes tumor control
Show full captionFigure viewer
We compared NK cell numbers in the BM of reconstituted S1pr5+/+ and S1pr5−/− mixed BM chimeras (Figure S6B) that had been subjected to 48 h fasting and found that fasting-mediated NK cell accumulation in the BM was reduced in the absence of S1PR5 (Figure 6B). Because S1pr5−/− mice have increased numbers of NK cells in the BM under ad libitum conditions, the differences between fasted and ad libitum-fed mice were compared within genotypes and presented as fold-change (fasted/average in ad libitum). An increase in mature CD27−CD11b+ NK cells was observed in the BM after fasting, and S1pr5eGFP expression was also significantly increased in this population (Figures S6C and S6D). Deletion of S1PR5 selectively limited the entry of CD27−CD11b+ NK cells into the BM after 48 h of fasting (Figure 6C). To ensure that the loss of cells in the BM was due to defective trafficking, we transferred equal numbers of WT and S1pr5−/− NK cells into recipient mice (Figure S6E) that were either fasted for 48 h or fed ad libitum and found reduced numbers of S1pr5−/− NK cells in the BM compared with S1PR5-expressing WT NK cells (Figure S6F), altogether suggesting the S1P-S1PR5 axis is required for CFD-mediated NK cell migration into the BM during fasting.
Because NK cells and other lymphocytes enter and exit the BM through sinusoids that are connected to blood circulation,41,42 we incorporated an intravenous CD45 labeling technique43 to discriminate between sinusoid- and parenchyma-resident cells. CD45 labeling revealed a stepwise increase in parenchymal NK cells and a concomitant reduction in sinusoidal NK cells within 48 h of fasting (Figures 6D and S6G). Given that CXCR4 expression is tightly linked to NK cell retention in the parenchyma,41 we hypothesized that CXCR4 may also be integral to BM entry/exit during fasting. We compared the fold-change increase in BM NK cells from fasted mice treated with or without the CXCR4 antagonist, AMD3100, to ad libitum-fed mice and found a reduced capacity for NK cells to enter the BM (Figure 6E). Altogether, these data suggest that BM entry during fasting is mediated by an interplay of CXCR4-mediated parenchymal retention and S1P-guided NK cell accumulation.
Redistribution of NK cells to the BM during CFD promotes tumor control
To determine the effects of blocking NK cell entry into the BM on CFD-mediated tumor control, we challenged either S1pr5−/− mice or mice treated with AMD3100 with tumor cells and subjected them to either a CFD or ad libitum feeding (Figures S6H and S6I). Blocking fasting-induced BM entry of NK cells via either method abrogated CFD-mediated anti-tumor control (Figures 6F, 6G, and S6J–S6L). We next profiled the tumors of S1pr5−/− mice or mice treated with AMD3100 subjected to a CFD and found no differences in the total numbers of NK cells found in the TME (Figures 6H, 6I, and S6M). Furthermore, NK cells from the tumor and spleen no longer possessed CFD-mediated differences in IFN-γ production (Figures 6J, 6K, and S6N). Altogether, these data suggest that BM entry may promote the CFD-facilitated anti-tumor effector function observed in NK cells.
The fasted BM niche promotes NK cell priming via IL-12 production
To understand why NK cell redistribution to the BM was important for CFD-mediated anti-tumor responses, we performed scRNA-seq on NK cells from the BM of mice that were subjected to either CFD or ad libitum feeding (Figure S7A). NK cells were partially separated based on whether they had been subjected to a CFD or ad libitum feeding using UMAP projections (Figure 7A). We found several interleukin (IL)-12 pathway genes, such as Stat4, upregulated in NK cells from the BM of CFD mice (Figure 7B). We also observed upregulation of STAT4 signaling components JAK2 and TYK2, and a number of transcription factors that are induced by STAT444 (Figures S7B and S7C). Coinciding with the induction of IL-12 and STAT4 target genes in NK cells, we found increased IL-12 concentrations in the BM after 48 h fasting and reduction in the spleen and serum (Figure 7C). This coincided with an increase in IL-12-producing cells in the BM compared with the spleen (Figures 7D and S7D). IL-12+ cells in the BM expressed CD11b (Figures S7E and S7F) and were negative for lymphoid lineage markers CD19, CD3, TCRβ, and NK1.1 (Figure S7G), suggesting that the cell population is of myeloid origin. Because IL-12 is a potent stimulator of IFN-γ secretion in NK cells,45 we determined whether the increase in BM IL-12 during fasting could functionally prime NK cells. Indeed, NK cells from the BM of CFD mice showed a significant increase in IFN-γ production (Figure 7E). Furthermore, NK cells from the spleen of CFD mice that had been refed (thus redistributing NK cells that were previously in the BM back to the spleen) also secreted more IFN-γ (Figure 7F).

Figure 7 The fasted BM niche promotes NK cell priming via IL-12 production
Show full captionFigure viewer
To assess whether a link between fatty acid oxidation and IL-12 priming exists, we tested whether NK cells lacking fatty acid oxidation capacity still showed BM-specific reprogramming. After 48 h of fasting, we found NK-Cpt1a−/− NK cells redistributed to the BM in a similar manner to WT NK-Cpt1a+/+ NK cells (Figure S7H). In agreement with intact redistribution during fasting, NK cells from NK-Cpt1a−/− mice subjected to a CFD showed similar increases in IFN-γ production in the BM when compared with ad libitum-fed controls (Figure S7I). These findings suggest that the inability of NK cells in the BM to perform fatty acid oxidation does not impair IL-12 priming and subsequent IFN-γ production. Altogether, these data suggest that, during periods of fasting, the BM niche is specifically reshaped to provide increased local concentrations of IL-12, priming NK cells that have preferentially redistributed to the BM and inducing local and peripheral IFN-γ secretion by NK cells important for anti-tumor immunity.
Discussion
Dietary restriction in the form of fasting and limiting caloric intake redistributes both adaptive and innate immune cell subsets such as B cells, T cells, and myeloid cells.18,19,20 Because NK cells are often placed at the intersection of innate and adaptive immunity, they represent a unique cell type that possesses features of both.46 Unlike adaptive T and B cells, NK cells act as a first line of defense against infections and tumor metastasis, and unlike innate myeloid cells, which can become detrimental in the TME as myeloid-derived suppressor cells, NK cell presence within tumors is almost always associated with better prognosis.6,47,48 Here, we show evidence that tissue repositioning during periods of fasting is advantageous for peripheral NK cell function, poising them for enhanced metabolic status and survival in the TME.
A significant advantage that NK cells have over T cells in the context of fasting is their response to GCs. Unlike T cells, which are sensitive to high levels of endogenous GCs that cause excessive cell death,18,49 we found that NK cells did not undergo greater death in response to GC exposure in vivo and in vitro. Instead, GCs promoted upregulation of CPT1A for increased FA oxidation capability, a phenomenon also observed in cardiomyocytes and muscle.27,50 Like M2 macrophages, NK cells increase lipid uptake, OXPHOS, and FA oxidation.51 Their ability to metabolically adapt in conditions of high GCs and excess FAs may be a contributing factor that distinguishes the innate functionality of NK cells from T or B cells in fasting conditions.
Metabolic rewiring in several cell types can be induced by nutrient restriction, shifting cellular metabolism from reliance on glycolysis to dependence on mitochondrial FA oxidation.26 However, this specific adaptation has not been mechanistically characterized in NK cells. Although FA oxidation was recently shown to be crucial for NK cell antiviral responses,32 much of our understanding of lipid uptake has been in the context of obesity and NK cell dysfunction, and thus the use of FAs as an energy source during cyclic fasting to improve functional responses against cancer has not been previously appreciated. In obese individuals, NK cells show an accumulation of lipids, which is correlated with reduced functionality.52 Similarly, in a mouse model of lymphoma, NK cell dysfunction is correlated with lipid accumulation and increased lipid metabolism.53 Unlike these previous studies, our work shows that cyclic fasting results in increased NK cell FA oxidation without lipid accumulation, suggesting that fasting might be an optimal approach to utilize excess lipids.
Both the obesity and lymphoma environments raise the interesting observation that NK cells in these conditions are continuously exposed to lipid-rich environments. By contrast, our model of a CFD, where a proportion of NK cells are acutely and intermittently exposed to high concentrations of FAs, represents a scenario where NK cells are constantly being shifted from glucose-depleted, FA-rich environments back to baseline during each period of fasting/re-feeding. Whether cyclic fasting can improve NK cell function in the continuously lipid-laden environments found in obese mice or humans requires further investigation. Of note, many prior and current studies, including our own here, do not assess NK cell function and FA oxidation capacity in adipose tissue. Our recent work suggests that NK cells have the ability to conditionally alter their metabolism based on the tissue in which they reside54; thus, future studies will seek to understand such additional tissue-specific roles of fasting, particularly because adipose-residing innate lymphocytes have metabolic and functional adaptations in obesity-induced disease settings.55,56,57,58
Acute myeloid leukemia (AML) is an aggressive hematological malignancy where improved outcomes are strongly associated with increased NK cell cytolytic activity.59,60,61 As such, adoptive NK cell therapies in the clinic have primarily been targeted to AML.8 Thus, whether AML maintains a lipid-rich BM environment similar to lymphoma is of interest, as the BM can also represent a site of tumor growth. In AML, the effects of cyclic fasting may be opposing. On the one hand, NK cells are preferentially redistributed to the BM during CFD conditions, where they are primed to produce more IFN-γ, which could promote leukemia tumor control. On the other hand (and as we have also observed in our study), cyclic fasting does not appear to increase FA uptake or LD abundance in NK cells in the BM, even after multiple cycles of fasting. Because our study does not address cancers of BM origin, future studies will investigate how fasting-induced NK cell responses to leukemias may differ from the solid tumor and metastasis models used in this study.
We observed that cyclic fasting improved NK cell functionality in a tissue-specific manner via parallel but disparate processes occurring in the BM versus the spleen. In mice, the half-life of an NK cell is ∼19.4 days, and thus adherence to 3 weeks of a CFD would assume that the NK cells profiled at tumor endpoint are heterogeneous in age33,62 and certainly harboring a proportion of cells that have cycled through more than one round of fasting and refeeding. Although we showed that NK cells are redistributed from the spleen and blood to the BM, we recognize that there are technological limitations with the current tools available to determine whether the same NK cell population has preferentially redistributed to the BM during each fasting cycle and, thus, whether an individual NK cell has been exposed to one or both tissue-specific processes. We provide evidence in our study that disrupting either process (i.e., FA oxidation via CPT1A deletion or BM trafficking via S1PR5 deletion or CXCR4 antagonism) has profound consequences on NK cell anti-tumor function, emphasizing the contribution of both BM entry and splenic reprogramming. Future cell-tracing studies may be able to determine whether NK cells found in the tumor have experienced fasting-induced signals from one or both tissue sites.
IL-12-producing CD45+CD11b+ cells were increased in the BM after adherence to cyclic fasting, suggesting that the effects of fasting can impose indirect consequences on NK cells. Although we have not established which subset of CD11b-expressing cells is producing IL-12, we can speculate that it may be derived from a subgroup of the myeloid lineage, as DCs and macrophages are potent producers of IL-12 and express CD11b. The BM is a complex tissue as it not only contains these subgroups but also their progenitors and a complex set of downstream putative populations. However, whether dietary restriction directly redistributes macrophages or DCs into the BM is still unknown, as most dietary studies have focused on the relocation and function of adipose tissue macrophages, suggesting that they accumulate in areas with increased lipids.63,64 Dietary restriction increases adipogenesis in the BM of mice18,65; thus, it is interesting to consider whether this increased adipogenesis may be responsible for an induction of IL-12-producing macrophages. Furthermore, a potential role of inflammatory macrophages is to metabolize lipids63; thus, increased IL-12-producing macrophages may be associated with increased lipid content within the BM. Additionally, fasting can modulate the gut bacteria flora and enrich for lactobacillus species,66 some strains of which can induce IL-12 production from macrophages.67,68,69 Because changes in gut microbiota can influence BM production of inflammatory cell types,70 we can speculate that these alterations may be responsible for the increased IL-12 production we observe in our model.
Altogether, our work recognizes a critical role for the innate immune system in the improved anti-tumor response induced by fasting. This work will be important in dissecting the immune landscape in patients from clinical trials using fasting and fasting-mimicking diets, given that NK cell infiltration and function have yet to be fully appreciated. Furthermore, we show that cyclic fasting can remodel different tissues and thus represents a novel method of improving tissue-specific NK cell function and enhancing innate immunity for the treatment of solid and metastatic cancers. Introduction
Natural killer (NK) cells are circulating cytotoxic lymphocytes that are crucial to host immunity against cancer via the secretion of lytic granules and the production of effector cytokines such as interferon (IFN)-γ. Unlike antigen-specific cytotoxic CD8+ T cells that require tumor antigen presentation by major histocompatibility complex (MHC) molecules, NK cells possess a broad range of germline-encoded activating receptors to target transformed or malignant cells.1 In this way, tumors that have decreased expression of MHC class I to evade T cell killing can be targeted by NK cells.2 Indeed, in clinical studies, the presence of an “NK cell signature” or increase in NK cells within the tumor is predictive of improved survival of patients across a broad range of cancers, including melanoma, breast cancer, and gastric cancer.3,4,5,6 However, the potential to harness NK cells for therapeutic use is limited, as their survival and functionality in the hostile tumor microenvironment (TME) are often suppressed. The TME is hypoxic, rich in lipids and lactic acid, but depleted of glucose, all of which suppress immune cell function and survival and promote tumor growth.7,8 Because NK cells primarily rely on glycolysis for homeostasis and effector functions,9,10,11,12 identifying strategies that reprogram NK cell metabolism to overcome the metabolically immunosuppressive TME is essential to develop novel anti-tumor therapies.
Dietary restriction in the form of fasting or fasting-mimicking diets has garnered significant interest due to its profound effects on metabolism, cancer incidence, and anti-tumor immunity.13 Cyclic fasting impacts tumor cells by inducing sensitivity to chemotherapy and promoting an anti-Warburg effect.14,15 As such, clinical trials implementing these diets alongside various standard-of-care cancer therapies have yielded exciting and promising results, including improved tumor responsiveness to therapy and enhanced immune responses.16,17 Indeed, dietary restriction influences the redistribution and function of immune populations throughout the host, including T cells, B cells, and myeloid cells, and is associated with enhanced immunity.18,19,20,21,22
Here, we examined the impact of dietary restriction on NK cell function. We found that cyclic fasting remodels distinct tissue-specific microenvironments that reprogram the metabolic and functional state of NK cells to enhance anti-tumor immunity and reveal a critical role for NK cells in improved anti-tumor responses induced by fasting. Our findings suggest that NK cell function should be examined in clinical trials employing fasting regimens and that cyclic or periodic fasting may exemplify a dietary strategy for improved innate immunity in the treatment of solid and metastatic cancers.
ResultsCyclic fasting promotes NK cell-mediated anti-tumor immunity
To explore the effects of dietary restriction on NK cell-mediated anti-tumor immune responses, we developed a cyclic fasting diet (CFD) that is implemented for 3 weeks (Figure 1A). This diet was maintained without net weight loss in healthy or in tumor-bearing mice at endpoint when compared with mice fed ad libitum (Figures S1A and S1B). Adherence to a CFD improved the anti-tumor response against MC38 colon carcinoma (Figure 1B) and B16F10 melanoma metastasis (Figure 1C). B16F10 melanoma was measured using lung metastasis counts as well as lung weight as a measurement of tumor burden because, in some cases, the number of metastases was too high to be enumerated accurately. Improved MC38 and B16F10 anti-tumor immunity was dependent on NK cell activity, as depleting NK cells reduced the effects of a CFD on MC38 and B16F10 tumor growth, indicating broad protection of CFD-induced NK cell anti-tumor immunity against both cancers (Figures 1D and 1E). Although many NK cell ligands remained unchanged in the fasted state (Figure S1C), we observed a CFD-mediated decrease in MHC class I expression (Figure 1F), in agreement with the enhanced control of tumors mediated via “missing self” recognition by NK cells. Tumor-infiltrating lymphocyte (TIL) analysis of CFD and ad libitum mice injected with either tumor did not show any major alterations in absolute NK cell numbers (Figures 1G and 1H) or NK cell maturation (Figures S1D and S1E). Further, TIL analysis showed no significant changes in other lymphocyte, myeloid, or dendritic cell (DC) populations in both tumor models (Figures S1F–S1K). We also assessed the functionality of CD8+ T cells, CD4+ T cells, and tumor-associated macrophages (TAMs) and found functional capacity to be relatively unchanged by a CFD (Figures S2A–S2C). However, the CFD did enhance peripheral and tumor-infiltrating NK cell function, as assessed by IFN-γ production and degranulation (Figures 1I, 1J, S2D, and S2E).

Figure 1 Cyclic fasting promotes NK cell-mediated anti-tumor immunity
Show full captionFigure viewer
Nutrient restriction induces metabolic changes to maintain survival of NK cells
To investigate underlying molecular mechanisms initiating improved NK cell function and tumor control during a CFD, we focused on cellular changes that occur during a singular, acute fasting period. We profiled multiple lymphoid and myeloid cell populations and found that the majority of these cell populations were significantly decreased in the spleen of mice after a 48 h fast (Figure S3A). Because MHC class I expression was altered on tumor cells (Figure 1F), we shifted our focus toward NK cells and their adaptive cytotoxic counterparts, CD8+ T cells (Figures 2A and 2B). Of note, human peripheral blood NK cells were also decreased in this setting of a single acute fast (Figure S3B). Although we observed an increase in cell death in CD8+ T cells, NK cells did not show this same level of cell death at 48 h (Figure 2C). Aligned with this finding, we observed increased levels of apoptosis in CD8+ T cells when compared with NK cells early in the fasting window (0–12 h; Figures S3C and S3D), suggesting NK cells can adapt to changes in nutrient availability by using alternative mechanisms to ensure their survival.

Figure 2 Nutrient restriction induces metabolic changes to maintain survival of NK cells
Show full captionFigure viewer
Acute periods of fasting are known to cause a myriad of systemic metabolic changes that may affect NK cell survival.23 We observed that fasted mice displayed a sharp decline in serum glucose concentrations within 24 h, coinciding with a significant increase in free fatty acid (FA) concentrations (Figure 2D). Because homeostatic maintenance of both innate and adaptive lymphocytes primarily relies on glucose to fuel basal levels of oxidative phosphorylation (OXPHOS),9,24 we performed Seahorse metabolic flux assays on NK cells and total CD8+ T cells from ad libitum or fasted mice. Although both CD8+ T cells and NK cells showed a marked reduction in glycolysis and glycolytic capacity (Figures S3E and S3F), only fasted NK cells were surprisingly able to maintain their capacity for OXPHOS (Figures 2E and 2F). Alternative metabolic substrates, including FAs, are also known to fuel OXPHOS,25 and NK cells and CD8+ T cells from fasted mice showed increased uptake of FAs (Figures 2G and 2H). At the same time, NK cells from fasted mice also displayed a more prominent reduction in neutral lipids compared with CD8+ T cells (Figures 2I and S3G), indicating that increased FA uptake did not result in a net increase in lipid storage within lipid droplets (LDs). This decrease in neutral lipids may also suggest that the liberation of stored FAs can be used to fuel OXPHOS,26 and, as such, we observed increased autophagy in NK cells but not CD8+ T cells in the fasted state (Figures 2J and S3H). To further strengthen our findings, we inhibited FA oxidation and observed that NK cells from fasted mice were unable to maintain OXPHOS compared with ad libitum-fed controls (Figures 2K and S3I). Altogether, these data indicate that NK cells utilize FAs as an energy source to fuel OXPHOS during times of glucose withdrawal, ensuring their survival.
Cyclic fasting metabolically reprograms NK cells
Because NK cells showed a remarkable capacity to switch to FA use during fasting, we further investigated whether NK cells from mice that had undergone a CFD showed similar changes. We found that splenic NK cells from mice subjected to a CFD displayed an increased capacity to uptake FAs from the local environment (Figure 3A) but did not show an accumulation of lipids (Figure 3B). This coincided with a slight decrease in glucose uptake (Figure S4A). To gain insight into how this metabolic switch affects the transcriptomic profile of fasted NK cells, we performed single-cell RNA sequencing (scRNA-seq) on NK cells from tumor-bearing mice subjected to either a CFD or ad libitum feeding. Uniform manifold approximation and projection (UMAP) embedding suggested that fasted NK cells were distinct from their ad libitum-fed counterparts (Figure 3C). We then used unbiased louvain clustering and identified 9 distinct clusters shared between ad libitum and CFD NK cells (Figure S4B). Among the 9 clusters, clusters 0 and 4 were enriched for NK cells from CFD mice (Figure S4C). Gene set enrichment analysis (GSEA) performed on differential gene expression identified by MAST between ad libitum and CFD NK cells showed an upregulation of FA metabolism genes in CFD NK cells (Figure 3D), which may be primarily driven by CFD-enriched clusters, consistent with our previous observations (Figures S4B–S4D). Select genes known to be critical for FA metabolism were upregulated in CFD NK cells, including Cpt1a, encoding the critical enzyme that drives FA metabolism (Figure 3E). We also observed a decrease in the expression of Ldha, an important gene required to maintain glycolysis (Figure S3E). Together, these data suggest that a CFD pushes splenic NK cells toward a reprogrammed metabolic state involving increased FA metabolism and decreased glycolysis.

Figure 3 Cyclic fasting metabolically reprograms NK cells
Show full captionFigure viewer
Interestingly, in addition to FA metabolism, some of the most significantly upregulated differentially expressed genes in CFD NK cells were genes known to be induced by glucocorticoids (GCs; Figure 3F). This increase in GC receptor (GR) target gene expression (Figure 3G) coincided with a spike in GC concentration at both 24 and 48 h in the spleen and serum of fasted mice compared with those that were fed ad libitum (Figure 3H). GCs can induce FA metabolism in cardiomyocytes and acute lymphoblastic leukemia27,28; however, whether this occurs in immune cells has not been established. We treated mouse and human NK cells with the synthetic GC, dexamethasone (Dex), and found that a 24 h treatment of NK cells from both species with Dex increased CPT1A expression (Figures 3I and S4F). The induction was evident as early as 2 h (Figure S4G), demonstrating a rapid and sustained induction of CPT1A by GCs. Taken together, these data suggest that increased GC levels during periods of fasting drive expression of CPT1A, inducing FA oxidation in NK cells and thus promoting optimal metabolism depending on specific fuel availability.
CFD-induced FA oxidation improves the NK cell anti-tumor response
The TME can promote tumor growth while dampening TIL functions by limiting glucose, a metabolite required for optimal T cell anti-tumor function,29 and enriching for immunosuppressive lipids.30 Because NK cells from mice subjected to a CFD showed increased expression of CPT1A, we hypothesized that a preference for using FAs instead of glucose within the TME could improve NK cell function. Indeed, tumor-infiltrating NK cells from CFD mice showed increased CPT1A levels and increased FA uptake when compared with ad libitum-fed mice (Figures 4A and 4B). To study the effects of Cpt1a deletion on NK cells in vivo, we used Cpt1aflox/flox mice31 crossed with Ncr1iCre mice to generate mice that harbor an NK cell-specific deletion of CPT1A (NK-Cpt1a−/−; Figure S5A).32 CPT1A is required for the uptake of long-chain FAs into the mitochondria for FA oxidation (Figure 4C), and, as such, we measured the effects of a CFD on FA oxidation and anti-tumor function. NK-Cpt1a−/− and wild-type (WT) NK-Cpt1a+/+ littermate controls were challenged with tumors and subjected to either ad libitum or CFD conditions. Remarkably, NK cells deficient in Cpt1a were not able to control tumor growth in the CFD setting when compared with Cpt1a-sufficient NK cells (Figures 4D and S5B). The striking difference in tumor volume was associated with a significant decrease in the number of intra-tumoral Cpt1a−/− NK cells when compared with Cpt1a+/+ NK cells in CFD mice (Figures 4E and S5C). This reduction was linked to diminished expression of the anti-apoptotic protein BCL2 when compared with NK-Cpt1a+/+ controls (Figure 4F). Expression of the pro-apoptotic protein BIM was maintained (Figure 4G), increasing the BIM:BCL2 ratio and indicating that the balance of these proteins tipped in favor of cell death within NK cells that lack Cpt1a (Figure 4H). Tumor-infiltrating Cpt1a-deficient NK cells showed similar levels of FA uptake compared with control NK cells (Figure 4I), suggesting that although Cpt1a-deficient NK cells cannot perform mitochondrial FA oxidation, FA uptake is not disrupted. However, Cpt1a-deficient NK cells did show a marked reduction in effector function during a CFD (Figures 4J and S5D), suggesting that an inability to efficiently process intracellular FAs during fasting hampers NK cell survival and anti-tumor function, disrupting NK cell-mediated control of metastatic disease.

Figure 4 CFD-induced FA oxidation improves NK cell anti-tumor immunity
Show full captionFigure viewer
S1PR5 and CXCR4 govern redistribution of NK cells to the BM during fasting
Because acute periods of fasting resulted in decreased NK cell numbers in the spleen and blood without any significant changes in cell death (Figures 2A–2C and S3C), we reasoned that they could be accumulating in other organs in this setting (Figure S5E). Indeed, a significant increase in NK cells was observed in the bone marrow (BM) after 48 h of fasting (Figure 5A). The accumulation of NK cells in the BM during fasting coincided with a loss in blood and peripheral tissues and was followed by a restoration of NK cell numbers within 24 h of unrestricted refeeding (Figure 5B). These rapid and fluid changes suggested that a proportion of peripheral NK cells were redistributed to the BM during periods of fasting. To confirm this, we transferred mature CD45.1-expressing splenic NK cells into CD45.2-expressing recipient mice that were either fasted for 48 h or fed ad libitum. The transferred NK cells were recovered at reduced numbers in the spleen and blood and increased numbers in the BM (Figure 5C). In addition, we used the iNkp46tdTomato mouse model to “time stamp” NK cells by inducing expression of tdTomato (Tom+), permanently marking mature peripheral NK cells.33 Similar to the adoptive transfer experiment, Tom+ NK cells were redistributed to the BM during periods of fasting (Figure 5D). Together, these data support that while a population of NK cells remains in peripheral organs such as the spleen during fasting, some mature NK cells are preferentially redistributed to the BM.

Figure 5 Fasting promotes NK cell redistribution to the BM
Show full captionFigure viewer
In contrast to the metabolically reprogrammed NK cells recovered from the spleen of mice subjected to 48 h of fasting, NK cells from the BM showed a reduction in the uptake of FAs and CPT1A expression (Figures 5E and 5F). These changes coincided with reduced levels of GCs in the BM compared with serum and splenic levels when compared with ad libitum-fed controls (Figure 5G). Similarly, NK cells from the BM of mice subjected to a CFD showed reduced FA uptake but maintained glucose uptake (Figures S5F and S5G). The BM has previously been suggested as a niche that supports lymphocyte survival during periods of nutrient deprivation,18,19,20 and, altogether, our data suggest that the tissue-specific localization of NK cells during fasting may have alternate effects on metabolic and functional capacity.
To understand the consequences of blocking entry into the BM niche during a CFD, we determined which signal(s) were driving NK cells into the BM during periods of fasting. We observed increased numbers of Ter119-expressing red blood cells (RBCs) in the BM during fasting compared with ad libitum-fed controls (Figure S6A). Increased RBCs in the BM are a phenomenon previously reported during dietary restriction and may be a consequence of increased GCs in the periphery.18,34,35 RBCs are a major source of sphingosine-1-phosphate (S1P), a molecule important for lymphocyte trafficking.36,37 NK cells express high levels of S1PR5, a receptor required for efficient NK cell trafficking through lymph nodes and the BM.38,39 We used a knockin S1pr5eGFP-T2A-iCre reporter mouse40 and found that S1pr5eGFP expression increased in the BM over 48 h of fasting (Figure 6A), suggesting that an increase of RBCs might also increase the local S1P concentration in the BM.

Figure 6 Redistribution of NK cells to the BM during a CFD promotes tumor control
Show full captionFigure viewer
We compared NK cell numbers in the BM of reconstituted S1pr5+/+ and S1pr5−/− mixed BM chimeras (Figure S6B) that had been subjected to 48 h fasting and found that fasting-mediated NK cell accumulation in the BM was reduced in the absence of S1PR5 (Figure 6B). Because S1pr5−/− mice have increased numbers of NK cells in the BM under ad libitum conditions, the differences between fasted and ad libitum-fed mice were compared within genotypes and presented as fold-change (fasted/average in ad libitum). An increase in mature CD27−CD11b+ NK cells was observed in the BM after fasting, and S1pr5eGFP expression was also significantly increased in this population (Figures S6C and S6D). Deletion of S1PR5 selectively limited the entry of CD27−CD11b+ NK cells into the BM after 48 h of fasting (Figure 6C). To ensure that the loss of cells in the BM was due to defective trafficking, we transferred equal numbers of WT and S1pr5−/− NK cells into recipient mice (Figure S6E) that were either fasted for 48 h or fed ad libitum and found reduced numbers of S1pr5−/− NK cells in the BM compared with S1PR5-expressing WT NK cells (Figure S6F), altogether suggesting the S1P-S1PR5 axis is required for CFD-mediated NK cell migration into the BM during fasting.
Because NK cells and other lymphocytes enter and exit the BM through sinusoids that are connected to blood circulation,41,42 we incorporated an intravenous CD45 labeling technique43 to discriminate between sinusoid- and parenchyma-resident cells. CD45 labeling revealed a stepwise increase in parenchymal NK cells and a concomitant reduction in sinusoidal NK cells within 48 h of fasting (Figures 6D and S6G). Given that CXCR4 expression is tightly linked to NK cell retention in the parenchyma,41 we hypothesized that CXCR4 may also be integral to BM entry/exit during fasting. We compared the fold-change increase in BM NK cells from fasted mice treated with or without the CXCR4 antagonist, AMD3100, to ad libitum-fed mice and found a reduced capacity for NK cells to enter the BM (Figure 6E). Altogether, these data suggest that BM entry during fasting is mediated by an interplay of CXCR4-mediated parenchymal retention and S1P-guided NK cell accumulation.
Redistribution of NK cells to the BM during CFD promotes tumor control
To determine the effects of blocking NK cell entry into the BM on CFD-mediated tumor control, we challenged either S1pr5−/− mice or mice treated with AMD3100 with tumor cells and subjected them to either a CFD or ad libitum feeding (Figures S6H and S6I). Blocking fasting-induced BM entry of NK cells via either method abrogated CFD-mediated anti-tumor control (Figures 6F, 6G, and S6J–S6L). We next profiled the tumors of S1pr5−/− mice or mice treated with AMD3100 subjected to a CFD and found no differences in the total numbers of NK cells found in the TME (Figures 6H, 6I, and S6M). Furthermore, NK cells from the tumor and spleen no longer possessed CFD-mediated differences in IFN-γ production (Figures 6J, 6K, and S6N). Altogether, these data suggest that BM entry may promote the CFD-facilitated anti-tumor effector function observed in NK cells.
The fasted BM niche promotes NK cell priming via IL-12 production
To understand why NK cell redistribution to the BM was important for CFD-mediated anti-tumor responses, we performed scRNA-seq on NK cells from the BM of mice that were subjected to either CFD or ad libitum feeding (Figure S7A). NK cells were partially separated based on whether they had been subjected to a CFD or ad libitum feeding using UMAP projections (Figure 7A). We found several interleukin (IL)-12 pathway genes, such as Stat4, upregulated in NK cells from the BM of CFD mice (Figure 7B). We also observed upregulation of STAT4 signaling components JAK2 and TYK2, and a number of transcription factors that are induced by STAT444 (Figures S7B and S7C). Coinciding with the induction of IL-12 and STAT4 target genes in NK cells, we found increased IL-12 concentrations in the BM after 48 h fasting and reduction in the spleen and serum (Figure 7C). This coincided with an increase in IL-12-producing cells in the BM compared with the spleen (Figures 7D and S7D). IL-12+ cells in the BM expressed CD11b (Figures S7E and S7F) and were negative for lymphoid lineage markers CD19, CD3, TCRβ, and NK1.1 (Figure S7G), suggesting that the cell population is of myeloid origin. Because IL-12 is a potent stimulator of IFN-γ secretion in NK cells,45 we determined whether the increase in BM IL-12 during fasting could functionally prime NK cells. Indeed, NK cells from the BM of CFD mice showed a significant increase in IFN-γ production (Figure 7E). Furthermore, NK cells from the spleen of CFD mice that had been refed (thus redistributing NK cells that were previously in the BM back to the spleen) also secreted more IFN-γ (Figure 7F).

Figure 7 The fasted BM niche promotes NK cell priming via IL-12 production
Show full captionFigure viewer
To assess whether a link between fatty acid oxidation and IL-12 priming exists, we tested whether NK cells lacking fatty acid oxidation capacity still showed BM-specific reprogramming. After 48 h of fasting, we found NK-Cpt1a−/− NK cells redistributed to the BM in a similar manner to WT NK-Cpt1a+/+ NK cells (Figure S7H). In agreement with intact redistribution during fasting, NK cells from NK-Cpt1a−/− mice subjected to a CFD showed similar increases in IFN-γ production in the BM when compared with ad libitum-fed controls (Figure S7I). These findings suggest that the inability of NK cells in the BM to perform fatty acid oxidation does not impair IL-12 priming and subsequent IFN-γ production. Altogether, these data suggest that, during periods of fasting, the BM niche is specifically reshaped to provide increased local concentrations of IL-12, priming NK cells that have preferentially redistributed to the BM and inducing local and peripheral IFN-γ secretion by NK cells important for anti-tumor immunity.
Discussion
Dietary restriction in the form of fasting and limiting caloric intake redistributes both adaptive and innate immune cell subsets such as B cells, T cells, and myeloid cells.18,19,20 Because NK cells are often placed at the intersection of innate and adaptive immunity, they represent a unique cell type that possesses features of both.46 Unlike adaptive T and B cells, NK cells act as a first line of defense against infections and tumor metastasis, and unlike innate myeloid cells, which can become detrimental in the TME as myeloid-derived suppressor cells, NK cell presence within tumors is almost always associated with better prognosis.6,47,48 Here, we show evidence that tissue repositioning during periods of fasting is advantageous for peripheral NK cell function, poising them for enhanced metabolic status and survival in the TME.
A significant advantage that NK cells have over T cells in the context of fasting is their response to GCs. Unlike T cells, which are sensitive to high levels of endogenous GCs that cause excessive cell death,18,49 we found that NK cells did not undergo greater death in response to GC exposure in vivo and in vitro. Instead, GCs promoted upregulation of CPT1A for increased FA oxidation capability, a phenomenon also observed in cardiomyocytes and muscle.27,50 Like M2 macrophages, NK cells increase lipid uptake, OXPHOS, and FA oxidation.51 Their ability to metabolically adapt in conditions of high GCs and excess FAs may be a contributing factor that distinguishes the innate functionality of NK cells from T or B cells in fasting conditions.
Metabolic rewiring in several cell types can be induced by nutrient restriction, shifting cellular metabolism from reliance on glycolysis to dependence on mitochondrial FA oxidation.26 However, this specific adaptation has not been mechanistically characterized in NK cells. Although FA oxidation was recently shown to be crucial for NK cell antiviral responses,32 much of our understanding of lipid uptake has been in the context of obesity and NK cell dysfunction, and thus the use of FAs as an energy source during cyclic fasting to improve functional responses against cancer has not been previously appreciated. In obese individuals, NK cells show an accumulation of lipids, which is correlated with reduced functionality.52 Similarly, in a mouse model of lymphoma, NK cell dysfunction is correlated with lipid accumulation and increased lipid metabolism.53 Unlike these previous studies, our work shows that cyclic fasting results in increased NK cell FA oxidation without lipid accumulation, suggesting that fasting might be an optimal approach to utilize excess lipids.
Both the obesity and lymphoma environments raise the interesting observation that NK cells in these conditions are continuously exposed to lipid-rich environments. By contrast, our model of a CFD, where a proportion of NK cells are acutely and intermittently exposed to high concentrations of FAs, represents a scenario where NK cells are constantly being shifted from glucose-depleted, FA-rich environments back to baseline during each period of fasting/re-feeding. Whether cyclic fasting can improve NK cell function in the continuously lipid-laden environments found in obese mice or humans requires further investigation. Of note, many prior and current studies, including our own here, do not assess NK cell function and FA oxidation capacity in adipose tissue. Our recent work suggests that NK cells have the ability to conditionally alter their metabolism based on the tissue in which they reside54; thus, future studies will seek to understand such additional tissue-specific roles of fasting, particularly because adipose-residing innate lymphocytes have metabolic and functional adaptations in obesity-induced disease settings.55,56,57,58
Acute myeloid leukemia (AML) is an aggressive hematological malignancy where improved outcomes are strongly associated with increased NK cell cytolytic activity.59,60,61 As such, adoptive NK cell therapies in the clinic have primarily been targeted to AML.8 Thus, whether AML maintains a lipid-rich BM environment similar to lymphoma is of interest, as the BM can also represent a site of tumor growth. In AML, the effects of cyclic fasting may be opposing. On the one hand, NK cells are preferentially redistributed to the BM during CFD conditions, where they are primed to produce more IFN-γ, which could promote leukemia tumor control. On the other hand (and as we have also observed in our study), cyclic fasting does not appear to increase FA uptake or LD abundance in NK cells in the BM, even after multiple cycles of fasting. Because our study does not address cancers of BM origin, future studies will investigate how fasting-induced NK cell responses to leukemias may differ from the solid tumor and metastasis models used in this study.
We observed that cyclic fasting improved NK cell functionality in a tissue-specific manner via parallel but disparate processes occurring in the BM versus the spleen. In mice, the half-life of an NK cell is ∼19.4 days, and thus adherence to 3 weeks of a CFD would assume that the NK cells profiled at tumor endpoint are heterogeneous in age33,62 and certainly harboring a proportion of cells that have cycled through more than one round of fasting and refeeding. Although we showed that NK cells are redistributed from the spleen and blood to the BM, we recognize that there are technological limitations with the current tools available to determine whether the same NK cell population has preferentially redistributed to the BM during each fasting cycle and, thus, whether an individual NK cell has been exposed to one or both tissue-specific processes. We provide evidence in our study that disrupting either process (i.e., FA oxidation via CPT1A deletion or BM trafficking via S1PR5 deletion or CXCR4 antagonism) has profound consequences on NK cell anti-tumor function, emphasizing the contribution of both BM entry and splenic reprogramming. Future cell-tracing studies may be able to determine whether NK cells found in the tumor have experienced fasting-induced signals from one or both tissue sites.
IL-12-producing CD45+CD11b+ cells were increased in the BM after adherence to cyclic fasting, suggesting that the effects of fasting can impose indirect consequences on NK cells. Although we have not established which subset of CD11b-expressing cells is producing IL-12, we can speculate that it may be derived from a subgroup of the myeloid lineage, as DCs and macrophages are potent producers of IL-12 and express CD11b. The BM is a complex tissue as it not only contains these subgroups but also their progenitors and a complex set of downstream putative populations. However, whether dietary restriction directly redistributes macrophages or DCs into the BM is still unknown, as most dietary studies have focused on the relocation and function of adipose tissue macrophages, suggesting that they accumulate in areas with increased lipids.63,64 Dietary restriction increases adipogenesis in the BM of mice18,65; thus, it is interesting to consider whether this increased adipogenesis may be responsible for an induction of IL-12-producing macrophages. Furthermore, a potential role of inflammatory macrophages is to metabolize lipids63; thus, increased IL-12-producing macrophages may be associated with increased lipid content within the BM. Additionally, fasting can modulate the gut bacteria flora and enrich for lactobacillus species,66 some strains of which can induce IL-12 production from macrophages.67,68,69 Because changes in gut microbiota can influence BM production of inflammatory cell types,70 we can speculate that these alterations may be responsible for the increased IL-12 production we observe in our model.
Altogether, our work recognizes a critical role for the innate immune system in the improved anti-tumor response induced by fasting. This work will be important in dissecting the immune landscape in patients from clinical trials using fasting and fasting-mimicking diets, given that NK cell infiltration and function have yet to be fully appreciated. Furthermore, we show that cyclic fasting can remodel different tissues and thus represents a novel method of improving tissue-specific NK cell function and enhancing innate immunity for the treatment of solid and metastatic cancers. Introduction
Natural killer (NK) cells are circulating cytotoxic lymphocytes that are crucial to host immunity against cancer via the secretion of lytic granules and the production of effector cytokines such as interferon (IFN)-γ. Unlike antigen-specific cytotoxic CD8+ T cells that require tumor antigen presentation by major histocompatibility complex (MHC) molecules, NK cells possess a broad range of germline-encoded activating receptors to target transformed or malignant cells.1 In this way, tumors that have decreased expression of MHC class I to evade T cell killing can be targeted by NK cells.2 Indeed, in clinical studies, the presence of an “NK cell signature” or increase in NK cells within the tumor is predictive of improved survival of patients across a broad range of cancers, including melanoma, breast cancer, and gastric cancer.3,4,5,6 However, the potential to harness NK cells for therapeutic use is limited, as their survival and functionality in the hostile tumor microenvironment (TME) are often suppressed. The TME is hypoxic, rich in lipids and lactic acid, but depleted of glucose, all of which suppress immune cell function and survival and promote tumor growth.7,8 Because NK cells primarily rely on glycolysis for homeostasis and effector functions,9,10,11,12 identifying strategies that reprogram NK cell metabolism to overcome the metabolically immunosuppressive TME is essential to develop novel anti-tumor therapies.
Dietary restriction in the form of fasting or fasting-mimicking diets has garnered significant interest due to its profound effects on metabolism, cancer incidence, and anti-tumor immunity.13 Cyclic fasting impacts tumor cells by inducing sensitivity to chemotherapy and promoting an anti-Warburg effect.14,15 As such, clinical trials implementing these diets alongside various standard-of-care cancer therapies have yielded exciting and promising results, including improved tumor responsiveness to therapy and enhanced immune responses.16,17 Indeed, dietary restriction influences the redistribution and function of immune populations throughout the host, including T cells, B cells, and myeloid cells, and is associated with enhanced immunity.18,19,20,21,22
Here, we examined the impact of dietary restriction on NK cell function. We found that cyclic fasting remodels distinct tissue-specific microenvironments that reprogram the metabolic and functional state of NK cells to enhance anti-tumor immunity and reveal a critical role for NK cells in improved anti-tumor responses induced by fasting. Our findings suggest that NK cell function should be examined in clinical trials employing fasting regimens and that cyclic or periodic fasting may exemplify a dietary strategy for improved innate immunity in the treatment of solid and metastatic cancers.
ResultsCyclic fasting promotes NK cell-mediated anti-tumor immunity
To explore the effects of dietary restriction on NK cell-mediated anti-tumor immune responses, we developed a cyclic fasting diet (CFD) that is implemented for 3 weeks (Figure 1A). This diet was maintained without net weight loss in healthy or in tumor-bearing mice at endpoint when compared with mice fed ad libitum (Figures S1A and S1B). Adherence to a CFD improved the anti-tumor response against MC38 colon carcinoma (Figure 1B) and B16F10 melanoma metastasis (Figure 1C). B16F10 melanoma was measured using lung metastasis counts as well as lung weight as a measurement of tumor burden because, in some cases, the number of metastases was too high to be enumerated accurately. Improved MC38 and B16F10 anti-tumor immunity was dependent on NK cell activity, as depleting NK cells reduced the effects of a CFD on MC38 and B16F10 tumor growth, indicating broad protection of CFD-induced NK cell anti-tumor immunity against both cancers (Figures 1D and 1E). Although many NK cell ligands remained unchanged in the fasted state (Figure S1C), we observed a CFD-mediated decrease in MHC class I expression (Figure 1F), in agreement with the enhanced control of tumors mediated via “missing self” recognition by NK cells. Tumor-infiltrating lymphocyte (TIL) analysis of CFD and ad libitum mice injected with either tumor did not show any major alterations in absolute NK cell numbers (Figures 1G and 1H) or NK cell maturation (Figures S1D and S1E). Further, TIL analysis showed no significant changes in other lymphocyte, myeloid, or dendritic cell (DC) populations in both tumor models (Figures S1F–S1K). We also assessed the functionality of CD8+ T cells, CD4+ T cells, and tumor-associated macrophages (TAMs) and found functional capacity to be relatively unchanged by a CFD (Figures S2A–S2C). However, the CFD did enhance peripheral and tumor-infiltrating NK cell function, as assessed by IFN-γ production and degranulation (Figures 1I, 1J, S2D, and S2E).

Figure 1 Cyclic fasting promotes NK cell-mediated anti-tumor immunity
Show full captionFigure viewer
Nutrient restriction induces metabolic changes to maintain survival of NK cells
To investigate underlying molecular mechanisms initiating improved NK cell function and tumor control during a CFD, we focused on cellular changes that occur during a singular, acute fasting period. We profiled multiple lymphoid and myeloid cell populations and found that the majority of these cell populations were significantly decreased in the spleen of mice after a 48 h fast (Figure S3A). Because MHC class I expression was altered on tumor cells (Figure 1F), we shifted our focus toward NK cells and their adaptive cytotoxic counterparts, CD8+ T cells (Figures 2A and 2B). Of note, human peripheral blood NK cells were also decreased in this setting of a single acute fast (Figure S3B). Although we observed an increase in cell death in CD8+ T cells, NK cells did not show this same level of cell death at 48 h (Figure 2C). Aligned with this finding, we observed increased levels of apoptosis in CD8+ T cells when compared with NK cells early in the fasting window (0–12 h; Figures S3C and S3D), suggesting NK cells can adapt to changes in nutrient availability by using alternative mechanisms to ensure their survival.

Figure 2 Nutrient restriction induces metabolic changes to maintain survival of NK cells
Show full captionFigure viewer
Acute periods of fasting are known to cause a myriad of systemic metabolic changes that may affect NK cell survival.23 We observed that fasted mice displayed a sharp decline in serum glucose concentrations within 24 h, coinciding with a significant increase in free fatty acid (FA) concentrations (Figure 2D). Because homeostatic maintenance of both innate and adaptive lymphocytes primarily relies on glucose to fuel basal levels of oxidative phosphorylation (OXPHOS),9,24 we performed Seahorse metabolic flux assays on NK cells and total CD8+ T cells from ad libitum or fasted mice. Although both CD8+ T cells and NK cells showed a marked reduction in glycolysis and glycolytic capacity (Figures S3E and S3F), only fasted NK cells were surprisingly able to maintain their capacity for OXPHOS (Figures 2E and 2F). Alternative metabolic substrates, including FAs, are also known to fuel OXPHOS,25 and NK cells and CD8+ T cells from fasted mice showed increased uptake of FAs (Figures 2G and 2H). At the same time, NK cells from fasted mice also displayed a more prominent reduction in neutral lipids compared with CD8+ T cells (Figures 2I and S3G), indicating that increased FA uptake did not result in a net increase in lipid storage within lipid droplets (LDs). This decrease in neutral lipids may also suggest that the liberation of stored FAs can be used to fuel OXPHOS,26 and, as such, we observed increased autophagy in NK cells but not CD8+ T cells in the fasted state (Figures 2J and S3H). To further strengthen our findings, we inhibited FA oxidation and observed that NK cells from fasted mice were unable to maintain OXPHOS compared with ad libitum-fed controls (Figures 2K and S3I). Altogether, these data indicate that NK cells utilize FAs as an energy source to fuel OXPHOS during times of glucose withdrawal, ensuring their survival.
Cyclic fasting metabolically reprograms NK cells
Because NK cells showed a remarkable capacity to switch to FA use during fasting, we further investigated whether NK cells from mice that had undergone a CFD showed similar changes. We found that splenic NK cells from mice subjected to a CFD displayed an increased capacity to uptake FAs from the local environment (Figure 3A) but did not show an accumulation of lipids (Figure 3B). This coincided with a slight decrease in glucose uptake (Figure S4A). To gain insight into how this metabolic switch affects the transcriptomic profile of fasted NK cells, we performed single-cell RNA sequencing (scRNA-seq) on NK cells from tumor-bearing mice subjected to either a CFD or ad libitum feeding. Uniform manifold approximation and projection (UMAP) embedding suggested that fasted NK cells were distinct from their ad libitum-fed counterparts (Figure 3C). We then used unbiased louvain clustering and identified 9 distinct clusters shared between ad libitum and CFD NK cells (Figure S4B). Among the 9 clusters, clusters 0 and 4 were enriched for NK cells from CFD mice (Figure S4C). Gene set enrichment analysis (GSEA) performed on differential gene expression identified by MAST between ad libitum and CFD NK cells showed an upregulation of FA metabolism genes in CFD NK cells (Figure 3D), which may be primarily driven by CFD-enriched clusters, consistent with our previous observations (Figures S4B–S4D). Select genes known to be critical for FA metabolism were upregulated in CFD NK cells, including Cpt1a, encoding the critical enzyme that drives FA metabolism (Figure 3E). We also observed a decrease in the expression of Ldha, an important gene required to maintain glycolysis (Figure S3E). Together, these data suggest that a CFD pushes splenic NK cells toward a reprogrammed metabolic state involving increased FA metabolism and decreased glycolysis.

Figure 3 Cyclic fasting metabolically reprograms NK cells
Show full captionFigure viewer
Interestingly, in addition to FA metabolism, some of the most significantly upregulated differentially expressed genes in CFD NK cells were genes known to be induced by glucocorticoids (GCs; Figure 3F). This increase in GC receptor (GR) target gene expression (Figure 3G) coincided with a spike in GC concentration at both 24 and 48 h in the spleen and serum of fasted mice compared with those that were fed ad libitum (Figure 3H). GCs can induce FA metabolism in cardiomyocytes and acute lymphoblastic leukemia27,28; however, whether this occurs in immune cells has not been established. We treated mouse and human NK cells with the synthetic GC, dexamethasone (Dex), and found that a 24 h treatment of NK cells from both species with Dex increased CPT1A expression (Figures 3I and S4F). The induction was evident as early as 2 h (Figure S4G), demonstrating a rapid and sustained induction of CPT1A by GCs. Taken together, these data suggest that increased GC levels during periods of fasting drive expression of CPT1A, inducing FA oxidation in NK cells and thus promoting optimal metabolism depending on specific fuel availability.
CFD-induced FA oxidation improves the NK cell anti-tumor response
The TME can promote tumor growth while dampening TIL functions by limiting glucose, a metabolite required for optimal T cell anti-tumor function,29 and enriching for immunosuppressive lipids.30 Because NK cells from mice subjected to a CFD showed increased expression of CPT1A, we hypothesized that a preference for using FAs instead of glucose within the TME could improve NK cell function. Indeed, tumor-infiltrating NK cells from CFD mice showed increased CPT1A levels and increased FA uptake when compared with ad libitum-fed mice (Figures 4A and 4B). To study the effects of Cpt1a deletion on NK cells in vivo, we used Cpt1aflox/flox mice31 crossed with Ncr1iCre mice to generate mice that harbor an NK cell-specific deletion of CPT1A (NK-Cpt1a−/−; Figure S5A).32 CPT1A is required for the uptake of long-chain FAs into the mitochondria for FA oxidation (Figure 4C), and, as such, we measured the effects of a CFD on FA oxidation and anti-tumor function. NK-Cpt1a−/− and wild-type (WT) NK-Cpt1a+/+ littermate controls were challenged with tumors and subjected to either ad libitum or CFD conditions. Remarkably, NK cells deficient in Cpt1a were not able to control tumor growth in the CFD setting when compared with Cpt1a-sufficient NK cells (Figures 4D and S5B). The striking difference in tumor volume was associated with a significant decrease in the number of intra-tumoral Cpt1a−/− NK cells when compared with Cpt1a+/+ NK cells in CFD mice (Figures 4E and S5C). This reduction was linked to diminished expression of the anti-apoptotic protein BCL2 when compared with NK-Cpt1a+/+ controls (Figure 4F). Expression of the pro-apoptotic protein BIM was maintained (Figure 4G), increasing the BIM:BCL2 ratio and indicating that the balance of these proteins tipped in favor of cell death within NK cells that lack Cpt1a (Figure 4H). Tumor-infiltrating Cpt1a-deficient NK cells showed similar levels of FA uptake compared with control NK cells (Figure 4I), suggesting that although Cpt1a-deficient NK cells cannot perform mitochondrial FA oxidation, FA uptake is not disrupted. However, Cpt1a-deficient NK cells did show a marked reduction in effector function during a CFD (Figures 4J and S5D), suggesting that an inability to efficiently process intracellular FAs during fasting hampers NK cell survival and anti-tumor function, disrupting NK cell-mediated control of metastatic disease.

Figure 4 CFD-induced FA oxidation improves NK cell anti-tumor immunity
Show full captionFigure viewer
S1PR5 and CXCR4 govern redistribution of NK cells to the BM during fasting
Because acute periods of fasting resulted in decreased NK cell numbers in the spleen and blood without any significant changes in cell death (Figures 2A–2C and S3C), we reasoned that they could be accumulating in other organs in this setting (Figure S5E). Indeed, a significant increase in NK cells was observed in the bone marrow (BM) after 48 h of fasting (Figure 5A). The accumulation of NK cells in the BM during fasting coincided with a loss in blood and peripheral tissues and was followed by a restoration of NK cell numbers within 24 h of unrestricted refeeding (Figure 5B). These rapid and fluid changes suggested that a proportion of peripheral NK cells were redistributed to the BM during periods of fasting. To confirm this, we transferred mature CD45.1-expressing splenic NK cells into CD45.2-expressing recipient mice that were either fasted for 48 h or fed ad libitum. The transferred NK cells were recovered at reduced numbers in the spleen and blood and increased numbers in the BM (Figure 5C). In addition, we used the iNkp46tdTomato mouse model to “time stamp” NK cells by inducing expression of tdTomato (Tom+), permanently marking mature peripheral NK cells.33 Similar to the adoptive transfer experiment, Tom+ NK cells were redistributed to the BM during periods of fasting (Figure 5D). Together, these data support that while a population of NK cells remains in peripheral organs such as the spleen during fasting, some mature NK cells are preferentially redistributed to the BM.

Figure 5 Fasting promotes NK cell redistribution to the BM
Show full captionFigure viewer
In contrast to the metabolically reprogrammed NK cells recovered from the spleen of mice subjected to 48 h of fasting, NK cells from the BM showed a reduction in the uptake of FAs and CPT1A expression (Figures 5E and 5F). These changes coincided with reduced levels of GCs in the BM compared with serum and splenic levels when compared with ad libitum-fed controls (Figure 5G). Similarly, NK cells from the BM of mice subjected to a CFD showed reduced FA uptake but maintained glucose uptake (Figures S5F and S5G). The BM has previously been suggested as a niche that supports lymphocyte survival during periods of nutrient deprivation,18,19,20 and, altogether, our data suggest that the tissue-specific localization of NK cells during fasting may have alternate effects on metabolic and functional capacity.
To understand the consequences of blocking entry into the BM niche during a CFD, we determined which signal(s) were driving NK cells into the BM during periods of fasting. We observed increased numbers of Ter119-expressing red blood cells (RBCs) in the BM during fasting compared with ad libitum-fed controls (Figure S6A). Increased RBCs in the BM are a phenomenon previously reported during dietary restriction and may be a consequence of increased GCs in the periphery.18,34,35 RBCs are a major source of sphingosine-1-phosphate (S1P), a molecule important for lymphocyte trafficking.36,37 NK cells express high levels of S1PR5, a receptor required for efficient NK cell trafficking through lymph nodes and the BM.38,39 We used a knockin S1pr5eGFP-T2A-iCre reporter mouse40 and found that S1pr5eGFP expression increased in the BM over 48 h of fasting (Figure 6A), suggesting that an increase of RBCs might also increase the local S1P concentration in the BM.

Figure 6 Redistribution of NK cells to the BM during a CFD promotes tumor control
Show full captionFigure viewer
We compared NK cell numbers in the BM of reconstituted S1pr5+/+ and S1pr5−/− mixed BM chimeras (Figure S6B) that had been subjected to 48 h fasting and found that fasting-mediated NK cell accumulation in the BM was reduced in the absence of S1PR5 (Figure 6B). Because S1pr5−/− mice have increased numbers of NK cells in the BM under ad libitum conditions, the differences between fasted and ad libitum-fed mice were compared within genotypes and presented as fold-change (fasted/average in ad libitum). An increase in mature CD27−CD11b+ NK cells was observed in the BM after fasting, and S1pr5eGFP expression was also significantly increased in this population (Figures S6C and S6D). Deletion of S1PR5 selectively limited the entry of CD27−CD11b+ NK cells into the BM after 48 h of fasting (Figure 6C). To ensure that the loss of cells in the BM was due to defective trafficking, we transferred equal numbers of WT and S1pr5−/− NK cells into recipient mice (Figure S6E) that were either fasted for 48 h or fed ad libitum and found reduced numbers of S1pr5−/− NK cells in the BM compared with S1PR5-expressing WT NK cells (Figure S6F), altogether suggesting the S1P-S1PR5 axis is required for CFD-mediated NK cell migration into the BM during fasting.
Because NK cells and other lymphocytes enter and exit the BM through sinusoids that are connected to blood circulation,41,42 we incorporated an intravenous CD45 labeling technique43 to discriminate between sinusoid- and parenchyma-resident cells. CD45 labeling revealed a stepwise increase in parenchymal NK cells and a concomitant reduction in sinusoidal NK cells within 48 h of fasting (Figures 6D and S6G). Given that CXCR4 expression is tightly linked to NK cell retention in the parenchyma,41 we hypothesized that CXCR4 may also be integral to BM entry/exit during fasting. We compared the fold-change increase in BM NK cells from fasted mice treated with or without the CXCR4 antagonist, AMD3100, to ad libitum-fed mice and found a reduced capacity for NK cells to enter the BM (Figure 6E). Altogether, these data suggest that BM entry during fasting is mediated by an interplay of CXCR4-mediated parenchymal retention and S1P-guided NK cell accumulation.
Redistribution of NK cells to the BM during CFD promotes tumor control
To determine the effects of blocking NK cell entry into the BM on CFD-mediated tumor control, we challenged either S1pr5−/− mice or mice treated with AMD3100 with tumor cells and subjected them to either a CFD or ad libitum feeding (Figures S6H and S6I). Blocking fasting-induced BM entry of NK cells via either method abrogated CFD-mediated anti-tumor control (Figures 6F, 6G, and S6J–S6L). We next profiled the tumors of S1pr5−/− mice or mice treated with AMD3100 subjected to a CFD and found no differences in the total numbers of NK cells found in the TME (Figures 6H, 6I, and S6M). Furthermore, NK cells from the tumor and spleen no longer possessed CFD-mediated differences in IFN-γ production (Figures 6J, 6K, and S6N). Altogether, these data suggest that BM entry may promote the CFD-facilitated anti-tumor effector function observed in NK cells.
The fasted BM niche promotes NK cell priming via IL-12 production
To understand why NK cell redistribution to the BM was important for CFD-mediated anti-tumor responses, we performed scRNA-seq on NK cells from the BM of mice that were subjected to either CFD or ad libitum feeding (Figure S7A). NK cells were partially separated based on whether they had been subjected to a CFD or ad libitum feeding using UMAP projections (Figure 7A). We found several interleukin (IL)-12 pathway genes, such as Stat4, upregulated in NK cells from the BM of CFD mice (Figure 7B). We also observed upregulation of STAT4 signaling components JAK2 and TYK2, and a number of transcription factors that are induced by STAT444 (Figures S7B and S7C). Coinciding with the induction of IL-12 and STAT4 target genes in NK cells, we found increased IL-12 concentrations in the BM after 48 h fasting and reduction in the spleen and serum (Figure 7C). This coincided with an increase in IL-12-producing cells in the BM compared with the spleen (Figures 7D and S7D). IL-12+ cells in the BM expressed CD11b (Figures S7E and S7F) and were negative for lymphoid lineage markers CD19, CD3, TCRβ, and NK1.1 (Figure S7G), suggesting that the cell population is of myeloid origin. Because IL-12 is a potent stimulator of IFN-γ secretion in NK cells,45 we determined whether the increase in BM IL-12 during fasting could functionally prime NK cells. Indeed, NK cells from the BM of CFD mice showed a significant increase in IFN-γ production (Figure 7E). Furthermore, NK cells from the spleen of CFD mice that had been refed (thus redistributing NK cells that were previously in the BM back to the spleen) also secreted more IFN-γ (Figure 7F).

Figure 7 The fasted BM niche promotes NK cell priming via IL-12 production
Show full captionFigure viewer
To assess whether a link between fatty acid oxidation and IL-12 priming exists, we tested whether NK cells lacking fatty acid oxidation capacity still showed BM-specific reprogramming. After 48 h of fasting, we found NK-Cpt1a−/− NK cells redistributed to the BM in a similar manner to WT NK-Cpt1a+/+ NK cells (Figure S7H). In agreement with intact redistribution during fasting, NK cells from NK-Cpt1a−/− mice subjected to a CFD showed similar increases in IFN-γ production in the BM when compared with ad libitum-fed controls (Figure S7I). These findings suggest that the inability of NK cells in the BM to perform fatty acid oxidation does not impair IL-12 priming and subsequent IFN-γ production. Altogether, these data suggest that, during periods of fasting, the BM niche is specifically reshaped to provide increased local concentrations of IL-12, priming NK cells that have preferentially redistributed to the BM and inducing local and peripheral IFN-γ secretion by NK cells important for anti-tumor immunity.
Discussion
Dietary restriction in the form of fasting and limiting caloric intake redistributes both adaptive and innate immune cell subsets such as B cells, T cells, and myeloid cells.18,19,20 Because NK cells are often placed at the intersection of innate and adaptive immunity, they represent a unique cell type that possesses features of both.46 Unlike adaptive T and B cells, NK cells act as a first line of defense against infections and tumor metastasis, and unlike innate myeloid cells, which can become detrimental in the TME as myeloid-derived suppressor cells, NK cell presence within tumors is almost always associated with better prognosis.6,47,48 Here, we show evidence that tissue repositioning during periods of fasting is advantageous for peripheral NK cell function, poising them for enhanced metabolic status and survival in the TME.
A significant advantage that NK cells have over T cells in the context of fasting is their response to GCs. Unlike T cells, which are sensitive to high levels of endogenous GCs that cause excessive cell death,18,49 we found that NK cells did not undergo greater death in response to GC exposure in vivo and in vitro. Instead, GCs promoted upregulation of CPT1A for increased FA oxidation capability, a phenomenon also observed in cardiomyocytes and muscle.27,50 Like M2 macrophages, NK cells increase lipid uptake, OXPHOS, and FA oxidation.51 Their ability to metabolically adapt in conditions of high GCs and excess FAs may be a contributing factor that distinguishes the innate functionality of NK cells from T or B cells in fasting conditions.
Metabolic rewiring in several cell types can be induced by nutrient restriction, shifting cellular metabolism from reliance on glycolysis to dependence on mitochondrial FA oxidation.26 However, this specific adaptation has not been mechanistically characterized in NK cells. Although FA oxidation was recently shown to be crucial for NK cell antiviral responses,32 much of our understanding of lipid uptake has been in the context of obesity and NK cell dysfunction, and thus the use of FAs as an energy source during cyclic fasting to improve functional responses against cancer has not been previously appreciated. In obese individuals, NK cells show an accumulation of lipids, which is correlated with reduced functionality.52 Similarly, in a mouse model of lymphoma, NK cell dysfunction is correlated with lipid accumulation and increased lipid metabolism.53 Unlike these previous studies, our work shows that cyclic fasting results in increased NK cell FA oxidation without lipid accumulation, suggesting that fasting might be an optimal approach to utilize excess lipids.
Both the obesity and lymphoma environments raise the interesting observation that NK cells in these conditions are continuously exposed to lipid-rich environments. By contrast, our model of a CFD, where a proportion of NK cells are acutely and intermittently exposed to high concentrations of FAs, represents a scenario where NK cells are constantly being shifted from glucose-depleted, FA-rich environments back to baseline during each period of fasting/re-feeding. Whether cyclic fasting can improve NK cell function in the continuously lipid-laden environments found in obese mice or humans requires further investigation. Of note, many prior and current studies, including our own here, do not assess NK cell function and FA oxidation capacity in adipose tissue. Our recent work suggests that NK cells have the ability to conditionally alter their metabolism based on the tissue in which they reside54; thus, future studies will seek to understand such additional tissue-specific roles of fasting, particularly because adipose-residing innate lymphocytes have metabolic and functional adaptations in obesity-induced disease settings.55,56,57,58
Acute myeloid leukemia (AML) is an aggressive hematological malignancy where improved outcomes are strongly associated with increased NK cell cytolytic activity.59,60,61 As such, adoptive NK cell therapies in the clinic have primarily been targeted to AML.8 Thus, whether AML maintains a lipid-rich BM environment similar to lymphoma is of interest, as the BM can also represent a site of tumor growth. In AML, the effects of cyclic fasting may be opposing. On the one hand, NK cells are preferentially redistributed to the BM during CFD conditions, where they are primed to produce more IFN-γ, which could promote leukemia tumor control. On the other hand (and as we have also observed in our study), cyclic fasting does not appear to increase FA uptake or LD abundance in NK cells in the BM, even after multiple cycles of fasting. Because our study does not address cancers of BM origin, future studies will investigate how fasting-induced NK cell responses to leukemias may differ from the solid tumor and metastasis models used in this study.
We observed that cyclic fasting improved NK cell functionality in a tissue-specific manner via parallel but disparate processes occurring in the BM versus the spleen. In mice, the half-life of an NK cell is ∼19.4 days, and thus adherence to 3 weeks of a CFD would assume that the NK cells profiled at tumor endpoint are heterogeneous in age33,62 and certainly harboring a proportion of cells that have cycled through more than one round of fasting and refeeding. Although we showed that NK cells are redistributed from the spleen and blood to the BM, we recognize that there are technological limitations with the current tools available to determine whether the same NK cell population has preferentially redistributed to the BM during each fasting cycle and, thus, whether an individual NK cell has been exposed to one or both tissue-specific processes. We provide evidence in our study that disrupting either process (i.e., FA oxidation via CPT1A deletion or BM trafficking via S1PR5 deletion or CXCR4 antagonism) has profound consequences on NK cell anti-tumor function, emphasizing the contribution of both BM entry and splenic reprogramming. Future cell-tracing studies may be able to determine whether NK cells found in the tumor have experienced fasting-induced signals from one or both tissue sites.
IL-12-producing CD45+CD11b+ cells were increased in the BM after adherence to cyclic fasting, suggesting that the effects of fasting can impose indirect consequences on NK cells. Although we have not established which subset of CD11b-expressing cells is producing IL-12, we can speculate that it may be derived from a subgroup of the myeloid lineage, as DCs and macrophages are potent producers of IL-12 and express CD11b. The BM is a complex tissue as it not only contains these subgroups but also their progenitors and a complex set of downstream putative populations. However, whether dietary restriction directly redistributes macrophages or DCs into the BM is still unknown, as most dietary studies have focused on the relocation and function of adipose tissue macrophages, suggesting that they accumulate in areas with increased lipids.63,64 Dietary restriction increases adipogenesis in the BM of mice18,65; thus, it is interesting to consider whether this increased adipogenesis may be responsible for an induction of IL-12-producing macrophages. Furthermore, a potential role of inflammatory macrophages is to metabolize lipids63; thus, increased IL-12-producing macrophages may be associated with increased lipid content within the BM. Additionally, fasting can modulate the gut bacteria flora and enrich for lactobacillus species,66 some strains of which can induce IL-12 production from macrophages.67,68,69 Because changes in gut microbiota can influence BM production of inflammatory cell types,70 we can speculate that these alterations may be responsible for the increased IL-12 production we observe in our model.
Altogether, our work recognizes a critical role for the innate immune system in the improved anti-tumor response induced by fasting. This work will be important in dissecting the immune landscape in patients from clinical trials using fasting and fasting-mimicking diets, given that NK cell infiltration and function have yet to be fully appreciated. Furthermore, we show that cyclic fasting can remodel different tissues and thus represents a novel method of improving tissue-specific NK cell function and enhancing innate immunity for the treatment of solid and metastatic cancers. Introduction
Natural killer (NK) cells are circulating cytotoxic lymphocytes that are crucial to host immunity against cancer via the secretion of lytic granules and the production of effector cytokines such as interferon (IFN)-γ. Unlike antigen-specific cytotoxic CD8+ T cells that require tumor antigen presentation by major histocompatibility complex (MHC) molecules, NK cells possess a broad range of germline-encoded activating receptors to target transformed or malignant cells.1 In this way, tumors that have decreased expression of MHC class I to evade T cell killing can be targeted by NK cells.2 Indeed, in clinical studies, the presence of an “NK cell signature” or increase in NK cells within the tumor is predictive of improved survival of patients across a broad range of cancers, including melanoma, breast cancer, and gastric cancer.3,4,5,6 However, the potential to harness NK cells for therapeutic use is limited, as their survival and functionality in the hostile tumor microenvironment (TME) are often suppressed. The TME is hypoxic, rich in lipids and lactic acid, but depleted of glucose, all of which suppress immune cell function and survival and promote tumor growth.7,8 Because NK cells primarily rely on glycolysis for homeostasis and effector functions,9,10,11,12 identifying strategies that reprogram NK cell metabolism to overcome the metabolically immunosuppressive TME is essential to develop novel anti-tumor therapies.
Dietary restriction in the form of fasting or fasting-mimicking diets has garnered significant interest due to its profound effects on metabolism, cancer incidence, and anti-tumor immunity.13 Cyclic fasting impacts tumor cells by inducing sensitivity to chemotherapy and promoting an anti-Warburg effect.14,15 As such, clinical trials implementing these diets alongside various standard-of-care cancer therapies have yielded exciting and promising results, including improved tumor responsiveness to therapy and enhanced immune responses.16,17 Indeed, dietary restriction influences the redistribution and function of immune populations throughout the host, including T cells, B cells, and myeloid cells, and is associated with enhanced immunity.18,19,20,21,22
Here, we examined the impact of dietary restriction on NK cell function. We found that cyclic fasting remodels distinct tissue-specific microenvironments that reprogram the metabolic and functional state of NK cells to enhance anti-tumor immunity and reveal a critical role for NK cells in improved anti-tumor responses induced by fasting. Our findings suggest that NK cell function should be examined in clinical trials employing fasting regimens and that cyclic or periodic fasting may exemplify a dietary strategy for improved innate immunity in the treatment of solid and metastatic cancers.
ResultsCyclic fasting promotes NK cell-mediated anti-tumor immunity
To explore the effects of dietary restriction on NK cell-mediated anti-tumor immune responses, we developed a cyclic fasting diet (CFD) that is implemented for 3 weeks (Figure 1A). This diet was maintained without net weight loss in healthy or in tumor-bearing mice at endpoint when compared with mice fed ad libitum (Figures S1A and S1B). Adherence to a CFD improved the anti-tumor response against MC38 colon carcinoma (Figure 1B) and B16F10 melanoma metastasis (Figure 1C). B16F10 melanoma was measured using lung metastasis counts as well as lung weight as a measurement of tumor burden because, in some cases, the number of metastases was too high to be enumerated accurately. Improved MC38 and B16F10 anti-tumor immunity was dependent on NK cell activity, as depleting NK cells reduced the effects of a CFD on MC38 and B16F10 tumor growth, indicating broad protection of CFD-induced NK cell anti-tumor immunity against both cancers (Figures 1D and 1E). Although many NK cell ligands remained unchanged in the fasted state (Figure S1C), we observed a CFD-mediated decrease in MHC class I expression (Figure 1F), in agreement with the enhanced control of tumors mediated via “missing self” recognition by NK cells. Tumor-infiltrating lymphocyte (TIL) analysis of CFD and ad libitum mice injected with either tumor did not show any major alterations in absolute NK cell numbers (Figures 1G and 1H) or NK cell maturation (Figures S1D and S1E). Further, TIL analysis showed no significant changes in other lymphocyte, myeloid, or dendritic cell (DC) populations in both tumor models (Figures S1F–S1K). We also assessed the functionality of CD8+ T cells, CD4+ T cells, and tumor-associated macrophages (TAMs) and found functional capacity to be relatively unchanged by a CFD (Figures S2A–S2C). However, the CFD did enhance peripheral and tumor-infiltrating NK cell function, as assessed by IFN-γ production and degranulation (Figures 1I, 1J, S2D, and S2E).

Figure 1 Cyclic fasting promotes NK cell-mediated anti-tumor immunity
Show full captionFigure viewer
Nutrient restriction induces metabolic changes to maintain survival of NK cells
To investigate underlying molecular mechanisms initiating improved NK cell function and tumor control during a CFD, we focused on cellular changes that occur during a singular, acute fasting period. We profiled multiple lymphoid and myeloid cell populations and found that the majority of these cell populations were significantly decreased in the spleen of mice after a 48 h fast (Figure S3A). Because MHC class I expression was altered on tumor cells (Figure 1F), we shifted our focus toward NK cells and their adaptive cytotoxic counterparts, CD8+ T cells (Figures 2A and 2B). Of note, human peripheral blood NK cells were also decreased in this setting of a single acute fast (Figure S3B). Although we observed an increase in cell death in CD8+ T cells, NK cells did not show this same level of cell death at 48 h (Figure 2C). Aligned with this finding, we observed increased levels of apoptosis in CD8+ T cells when compared with NK cells early in the fasting window (0–12 h; Figures S3C and S3D), suggesting NK cells can adapt to changes in nutrient availability by using alternative mechanisms to ensure their survival.

Figure 2 Nutrient restriction induces metabolic changes to maintain survival of NK cells
Show full captionFigure viewer
Acute periods of fasting are known to cause a myriad of systemic metabolic changes that may affect NK cell survival.23 We observed that fasted mice displayed a sharp decline in serum glucose concentrations within 24 h, coinciding with a significant increase in free fatty acid (FA) concentrations (Figure 2D). Because homeostatic maintenance of both innate and adaptive lymphocytes primarily relies on glucose to fuel basal levels of oxidative phosphorylation (OXPHOS),9,24 we performed Seahorse metabolic flux assays on NK cells and total CD8+ T cells from ad libitum or fasted mice. Although both CD8+ T cells and NK cells showed a marked reduction in glycolysis and glycolytic capacity (Figures S3E and S3F), only fasted NK cells were surprisingly able to maintain their capacity for OXPHOS (Figures 2E and 2F). Alternative metabolic substrates, including FAs, are also known to fuel OXPHOS,25 and NK cells and CD8+ T cells from fasted mice showed increased uptake of FAs (Figures 2G and 2H). At the same time, NK cells from fasted mice also displayed a more prominent reduction in neutral lipids compared with CD8+ T cells (Figures 2I and S3G), indicating that increased FA uptake did not result in a net increase in lipid storage within lipid droplets (LDs). This decrease in neutral lipids may also suggest that the liberation of stored FAs can be used to fuel OXPHOS,26 and, as such, we observed increased autophagy in NK cells but not CD8+ T cells in the fasted state (Figures 2J and S3H). To further strengthen our findings, we inhibited FA oxidation and observed that NK cells from fasted mice were unable to maintain OXPHOS compared with ad libitum-fed controls (Figures 2K and S3I). Altogether, these data indicate that NK cells utilize FAs as an energy source to fuel OXPHOS during times of glucose withdrawal, ensuring their survival.
Cyclic fasting metabolically reprograms NK cells
Because NK cells showed a remarkable capacity to switch to FA use during fasting, we further investigated whether NK cells from mice that had undergone a CFD showed similar changes. We found that splenic NK cells from mice subjected to a CFD displayed an increased capacity to uptake FAs from the local environment (Figure 3A) but did not show an accumulation of lipids (Figure 3B). This coincided with a slight decrease in glucose uptake (Figure S4A). To gain insight into how this metabolic switch affects the transcriptomic profile of fasted NK cells, we performed single-cell RNA sequencing (scRNA-seq) on NK cells from tumor-bearing mice subjected to either a CFD or ad libitum feeding. Uniform manifold approximation and projection (UMAP) embedding suggested that fasted NK cells were distinct from their ad libitum-fed counterparts (Figure 3C). We then used unbiased louvain clustering and identified 9 distinct clusters shared between ad libitum and CFD NK cells (Figure S4B). Among the 9 clusters, clusters 0 and 4 were enriched for NK cells from CFD mice (Figure S4C). Gene set enrichment analysis (GSEA) performed on differential gene expression identified by MAST between ad libitum and CFD NK cells showed an upregulation of FA metabolism genes in CFD NK cells (Figure 3D), which may be primarily driven by CFD-enriched clusters, consistent with our previous observations (Figures S4B–S4D). Select genes known to be critical for FA metabolism were upregulated in CFD NK cells, including Cpt1a, encoding the critical enzyme that drives FA metabolism (Figure 3E). We also observed a decrease in the expression of Ldha, an important gene required to maintain glycolysis (Figure S3E). Together, these data suggest that a CFD pushes splenic NK cells toward a reprogrammed metabolic state involving increased FA metabolism and decreased glycolysis.

Figure 3 Cyclic fasting metabolically reprograms NK cells
Show full captionFigure viewer
Interestingly, in addition to FA metabolism, some of the most significantly upregulated differentially expressed genes in CFD NK cells were genes known to be induced by glucocorticoids (GCs; Figure 3F). This increase in GC receptor (GR) target gene expression (Figure 3G) coincided with a spike in GC concentration at both 24 and 48 h in the spleen and serum of fasted mice compared with those that were fed ad libitum (Figure 3H). GCs can induce FA metabolism in cardiomyocytes and acute lymphoblastic leukemia27,28; however, whether this occurs in immune cells has not been established. We treated mouse and human NK cells with the synthetic GC, dexamethasone (Dex), and found that a 24 h treatment of NK cells from both species with Dex increased CPT1A expression (Figures 3I and S4F). The induction was evident as early as 2 h (Figure S4G), demonstrating a rapid and sustained induction of CPT1A by GCs. Taken together, these data suggest that increased GC levels during periods of fasting drive expression of CPT1A, inducing FA oxidation in NK cells and thus promoting optimal metabolism depending on specific fuel availability.
CFD-induced FA oxidation improves the NK cell anti-tumor response
The TME can promote tumor growth while dampening TIL functions by limiting glucose, a metabolite required for optimal T cell anti-tumor function,29 and enriching for immunosuppressive lipids.30 Because NK cells from mice subjected to a CFD showed increased expression of CPT1A, we hypothesized that a preference for using FAs instead of glucose within the TME could improve NK cell function. Indeed, tumor-infiltrating NK cells from CFD mice showed increased CPT1A levels and increased FA uptake when compared with ad libitum-fed mice (Figures 4A and 4B). To study the effects of Cpt1a deletion on NK cells in vivo, we used Cpt1aflox/flox mice31 crossed with Ncr1iCre mice to generate mice that harbor an NK cell-specific deletion of CPT1A (NK-Cpt1a−/−; Figure S5A).32 CPT1A is required for the uptake of long-chain FAs into the mitochondria for FA oxidation (Figure 4C), and, as such, we measured the effects of a CFD on FA oxidation and anti-tumor function. NK-Cpt1a−/− and wild-type (WT) NK-Cpt1a+/+ littermate controls were challenged with tumors and subjected to either ad libitum or CFD conditions. Remarkably, NK cells deficient in Cpt1a were not able to control tumor growth in the CFD setting when compared with Cpt1a-sufficient NK cells (Figures 4D and S5B). The striking difference in tumor volume was associated with a significant decrease in the number of intra-tumoral Cpt1a−/− NK cells when compared with Cpt1a+/+ NK cells in CFD mice (Figures 4E and S5C). This reduction was linked to diminished expression of the anti-apoptotic protein BCL2 when compared with NK-Cpt1a+/+ controls (Figure 4F). Expression of the pro-apoptotic protein BIM was maintained (Figure 4G), increasing the BIM:BCL2 ratio and indicating that the balance of these proteins tipped in favor of cell death within NK cells that lack Cpt1a (Figure 4H). Tumor-infiltrating Cpt1a-deficient NK cells showed similar levels of FA uptake compared with control NK cells (Figure 4I), suggesting that although Cpt1a-deficient NK cells cannot perform mitochondrial FA oxidation, FA uptake is not disrupted. However, Cpt1a-deficient NK cells did show a marked reduction in effector function during a CFD (Figures 4J and S5D), suggesting that an inability to efficiently process intracellular FAs during fasting hampers NK cell survival and anti-tumor function, disrupting NK cell-mediated control of metastatic disease.

Figure 4 CFD-induced FA oxidation improves NK cell anti-tumor immunity
Show full captionFigure viewer
S1PR5 and CXCR4 govern redistribution of NK cells to the BM during fasting
Because acute periods of fasting resulted in decreased NK cell numbers in the spleen and blood without any significant changes in cell death (Figures 2A–2C and S3C), we reasoned that they could be accumulating in other organs in this setting (Figure S5E). Indeed, a significant increase in NK cells was observed in the bone marrow (BM) after 48 h of fasting (Figure 5A). The accumulation of NK cells in the BM during fasting coincided with a loss in blood and peripheral tissues and was followed by a restoration of NK cell numbers within 24 h of unrestricted refeeding (Figure 5B). These rapid and fluid changes suggested that a proportion of peripheral NK cells were redistributed to the BM during periods of fasting. To confirm this, we transferred mature CD45.1-expressing splenic NK cells into CD45.2-expressing recipient mice that were either fasted for 48 h or fed ad libitum. The transferred NK cells were recovered at reduced numbers in the spleen and blood and increased numbers in the BM (Figure 5C). In addition, we used the iNkp46tdTomato mouse model to “time stamp” NK cells by inducing expression of tdTomato (Tom+), permanently marking mature peripheral NK cells.33 Similar to the adoptive transfer experiment, Tom+ NK cells were redistributed to the BM during periods of fasting (Figure 5D). Together, these data support that while a population of NK cells remains in peripheral organs such as the spleen during fasting, some mature NK cells are preferentially redistributed to the BM.

Figure 5 Fasting promotes NK cell redistribution to the BM
Show full captionFigure viewer
In contrast to the metabolically reprogrammed NK cells recovered from the spleen of mice subjected to 48 h of fasting, NK cells from the BM showed a reduction in the uptake of FAs and CPT1A expression (Figures 5E and 5F). These changes coincided with reduced levels of GCs in the BM compared with serum and splenic levels when compared with ad libitum-fed controls (Figure 5G). Similarly, NK cells from the BM of mice subjected to a CFD showed reduced FA uptake but maintained glucose uptake (Figures S5F and S5G). The BM has previously been suggested as a niche that supports lymphocyte survival during periods of nutrient deprivation,18,19,20 and, altogether, our data suggest that the tissue-specific localization of NK cells during fasting may have alternate effects on metabolic and functional capacity.
To understand the consequences of blocking entry into the BM niche during a CFD, we determined which signal(s) were driving NK cells into the BM during periods of fasting. We observed increased numbers of Ter119-expressing red blood cells (RBCs) in the BM during fasting compared with ad libitum-fed controls (Figure S6A). Increased RBCs in the BM are a phenomenon previously reported during dietary restriction and may be a consequence of increased GCs in the periphery.18,34,35 RBCs are a major source of sphingosine-1-phosphate (S1P), a molecule important for lymphocyte trafficking.36,37 NK cells express high levels of S1PR5, a receptor required for efficient NK cell trafficking through lymph nodes and the BM.38,39 We used a knockin S1pr5eGFP-T2A-iCre reporter mouse40 and found that S1pr5eGFP expression increased in the BM over 48 h of fasting (Figure 6A), suggesting that an increase of RBCs might also increase the local S1P concentration in the BM.

Figure 6 Redistribution of NK cells to the BM during a CFD promotes tumor control
Show full captionFigure viewer
We compared NK cell numbers in the BM of reconstituted S1pr5+/+ and S1pr5−/− mixed BM chimeras (Figure S6B) that had been subjected to 48 h fasting and found that fasting-mediated NK cell accumulation in the BM was reduced in the absence of S1PR5 (Figure 6B). Because S1pr5−/− mice have increased numbers of NK cells in the BM under ad libitum conditions, the differences between fasted and ad libitum-fed mice were compared within genotypes and presented as fold-change (fasted/average in ad libitum). An increase in mature CD27−CD11b+ NK cells was observed in the BM after fasting, and S1pr5eGFP expression was also significantly increased in this population (Figures S6C and S6D). Deletion of S1PR5 selectively limited the entry of CD27−CD11b+ NK cells into the BM after 48 h of fasting (Figure 6C). To ensure that the loss of cells in the BM was due to defective trafficking, we transferred equal numbers of WT and S1pr5−/− NK cells into recipient mice (Figure S6E) that were either fasted for 48 h or fed ad libitum and found reduced numbers of S1pr5−/− NK cells in the BM compared with S1PR5-expressing WT NK cells (Figure S6F), altogether suggesting the S1P-S1PR5 axis is required for CFD-mediated NK cell migration into the BM during fasting.
Because NK cells and other lymphocytes enter and exit the BM through sinusoids that are connected to blood circulation,41,42 we incorporated an intravenous CD45 labeling technique43 to discriminate between sinusoid- and parenchyma-resident cells. CD45 labeling revealed a stepwise increase in parenchymal NK cells and a concomitant reduction in sinusoidal NK cells within 48 h of fasting (Figures 6D and S6G). Given that CXCR4 expression is tightly linked to NK cell retention in the parenchyma,41 we hypothesized that CXCR4 may also be integral to BM entry/exit during fasting. We compared the fold-change increase in BM NK cells from fasted mice treated with or without the CXCR4 antagonist, AMD3100, to ad libitum-fed mice and found a reduced capacity for NK cells to enter the BM (Figure 6E). Altogether, these data suggest that BM entry during fasting is mediated by an interplay of CXCR4-mediated parenchymal retention and S1P-guided NK cell accumulation.
Redistribution of NK cells to the BM during CFD promotes tumor control
To determine the effects of blocking NK cell entry into the BM on CFD-mediated tumor control, we challenged either S1pr5−/− mice or mice treated with AMD3100 with tumor cells and subjected them to either a CFD or ad libitum feeding (Figures S6H and S6I). Blocking fasting-induced BM entry of NK cells via either method abrogated CFD-mediated anti-tumor control (Figures 6F, 6G, and S6J–S6L). We next profiled the tumors of S1pr5−/− mice or mice treated with AMD3100 subjected to a CFD and found no differences in the total numbers of NK cells found in the TME (Figures 6H, 6I, and S6M). Furthermore, NK cells from the tumor and spleen no longer possessed CFD-mediated differences in IFN-γ production (Figures 6J, 6K, and S6N). Altogether, these data suggest that BM entry may promote the CFD-facilitated anti-tumor effector function observed in NK cells.
The fasted BM niche promotes NK cell priming via IL-12 production
To understand why NK cell redistribution to the BM was important for CFD-mediated anti-tumor responses, we performed scRNA-seq on NK cells from the BM of mice that were subjected to either CFD or ad libitum feeding (Figure S7A). NK cells were partially separated based on whether they had been subjected to a CFD or ad libitum feeding using UMAP projections (Figure 7A). We found several interleukin (IL)-12 pathway genes, such as Stat4, upregulated in NK cells from the BM of CFD mice (Figure 7B). We also observed upregulation of STAT4 signaling components JAK2 and TYK2, and a number of transcription factors that are induced by STAT444 (Figures S7B and S7C). Coinciding with the induction of IL-12 and STAT4 target genes in NK cells, we found increased IL-12 concentrations in the BM after 48 h fasting and reduction in the spleen and serum (Figure 7C). This coincided with an increase in IL-12-producing cells in the BM compared with the spleen (Figures 7D and S7D). IL-12+ cells in the BM expressed CD11b (Figures S7E and S7F) and were negative for lymphoid lineage markers CD19, CD3, TCRβ, and NK1.1 (Figure S7G), suggesting that the cell population is of myeloid origin. Because IL-12 is a potent stimulator of IFN-γ secretion in NK cells,45 we determined whether the increase in BM IL-12 during fasting could functionally prime NK cells. Indeed, NK cells from the BM of CFD mice showed a significant increase in IFN-γ production (Figure 7E). Furthermore, NK cells from the spleen of CFD mice that had been refed (thus redistributing NK cells that were previously in the BM back to the spleen) also secreted more IFN-γ (Figure 7F).

Figure 7 The fasted BM niche promotes NK cell priming via IL-12 production
Show full captionFigure viewer
To assess whether a link between fatty acid oxidation and IL-12 priming exists, we tested whether NK cells lacking fatty acid oxidation capacity still showed BM-specific reprogramming. After 48 h of fasting, we found NK-Cpt1a−/− NK cells redistributed to the BM in a similar manner to WT NK-Cpt1a+/+ NK cells (Figure S7H). In agreement with intact redistribution during fasting, NK cells from NK-Cpt1a−/− mice subjected to a CFD showed similar increases in IFN-γ production in the BM when compared with ad libitum-fed controls (Figure S7I). These findings suggest that the inability of NK cells in the BM to perform fatty acid oxidation does not impair IL-12 priming and subsequent IFN-γ production. Altogether, these data suggest that, during periods of fasting, the BM niche is specifically reshaped to provide increased local concentrations of IL-12, priming NK cells that have preferentially redistributed to the BM and inducing local and peripheral IFN-γ secretion by NK cells important for anti-tumor immunity.
Discussion
Dietary restriction in the form of fasting and limiting caloric intake redistributes both adaptive and innate immune cell subsets such as B cells, T cells, and myeloid cells.18,19,20 Because NK cells are often placed at the intersection of innate and adaptive immunity, they represent a unique cell type that possesses features of both.46 Unlike adaptive T and B cells, NK cells act as a first line of defense against infections and tumor metastasis, and unlike innate myeloid cells, which can become detrimental in the TME as myeloid-derived suppressor cells, NK cell presence within tumors is almost always associated with better prognosis.6,47,48 Here, we show evidence that tissue repositioning during periods of fasting is advantageous for peripheral NK cell function, poising them for enhanced metabolic status and survival in the TME.
A significant advantage that NK cells have over T cells in the context of fasting is their response to GCs. Unlike T cells, which are sensitive to high levels of endogenous GCs that cause excessive cell death,18,49 we found that NK cells did not undergo greater death in response to GC exposure in vivo and in vitro. Instead, GCs promoted upregulation of CPT1A for increased FA oxidation capability, a phenomenon also observed in cardiomyocytes and muscle.27,50 Like M2 macrophages, NK cells increase lipid uptake, OXPHOS, and FA oxidation.51 Their ability to metabolically adapt in conditions of high GCs and excess FAs may be a contributing factor that distinguishes the innate functionality of NK cells from T or B cells in fasting conditions.
Metabolic rewiring in several cell types can be induced by nutrient restriction, shifting cellular metabolism from reliance on glycolysis to dependence on mitochondrial FA oxidation.26 However, this specific adaptation has not been mechanistically characterized in NK cells. Although FA oxidation was recently shown to be crucial for NK cell antiviral responses,32 much of our understanding of lipid uptake has been in the context of obesity and NK cell dysfunction, and thus the use of FAs as an energy source during cyclic fasting to improve functional responses against cancer has not been previously appreciated. In obese individuals, NK cells show an accumulation of lipids, which is correlated with reduced functionality.52 Similarly, in a mouse model of lymphoma, NK cell dysfunction is correlated with lipid accumulation and increased lipid metabolism.53 Unlike these previous studies, our work shows that cyclic fasting results in increased NK cell FA oxidation without lipid accumulation, suggesting that fasting might be an optimal approach to utilize excess lipids.
Both the obesity and lymphoma environments raise the interesting observation that NK cells in these conditions are continuously exposed to lipid-rich environments. By contrast, our model of a CFD, where a proportion of NK cells are acutely and intermittently exposed to high concentrations of FAs, represents a scenario where NK cells are constantly being shifted from glucose-depleted, FA-rich environments back to baseline during each period of fasting/re-feeding. Whether cyclic fasting can improve NK cell function in the continuously lipid-laden environments found in obese mice or humans requires further investigation. Of note, many prior and current studies, including our own here, do not assess NK cell function and FA oxidation capacity in adipose tissue. Our recent work suggests that NK cells have the ability to conditionally alter their metabolism based on the tissue in which they reside54; thus, future studies will seek to understand such additional tissue-specific roles of fasting, particularly because adipose-residing innate lymphocytes have metabolic and functional adaptations in obesity-induced disease settings.55,56,57,58
Acute myeloid leukemia (AML) is an aggressive hematological malignancy where improved outcomes are strongly associated with increased NK cell cytolytic activity.59,60,61 As such, adoptive NK cell therapies in the clinic have primarily been targeted to AML.8 Thus, whether AML maintains a lipid-rich BM environment similar to lymphoma is of interest, as the BM can also represent a site of tumor growth. In AML, the effects of cyclic fasting may be opposing. On the one hand, NK cells are preferentially redistributed to the BM during CFD conditions, where they are primed to produce more IFN-γ, which could promote leukemia tumor control. On the other hand (and as we have also observed in our study), cyclic fasting does not appear to increase FA uptake or LD abundance in NK cells in the BM, even after multiple cycles of fasting. Because our study does not address cancers of BM origin, future studies will investigate how fasting-induced NK cell responses to leukemias may differ from the solid tumor and metastasis models used in this study.
We observed that cyclic fasting improved NK cell functionality in a tissue-specific manner via parallel but disparate processes occurring in the BM versus the spleen. In mice, the half-life of an NK cell is ∼19.4 days, and thus adherence to 3 weeks of a CFD would assume that the NK cells profiled at tumor endpoint are heterogeneous in age33,62 and certainly harboring a proportion of cells that have cycled through more than one round of fasting and refeeding. Although we showed that NK cells are redistributed from the spleen and blood to the BM, we recognize that there are technological limitations with the current tools available to determine whether the same NK cell population has preferentially redistributed to the BM during each fasting cycle and, thus, whether an individual NK cell has been exposed to one or both tissue-specific processes. We provide evidence in our study that disrupting either process (i.e., FA oxidation via CPT1A deletion or BM trafficking via S1PR5 deletion or CXCR4 antagonism) has profound consequences on NK cell anti-tumor function, emphasizing the contribution of both BM entry and splenic reprogramming. Future cell-tracing studies may be able to determine whether NK cells found in the tumor have experienced fasting-induced signals from one or both tissue sites.
IL-12-producing CD45+CD11b+ cells were increased in the BM after adherence to cyclic fasting, suggesting that the effects of fasting can impose indirect consequences on NK cells. Although we have not established which subset of CD11b-expressing cells is producing IL-12, we can speculate that it may be derived from a subgroup of the myeloid lineage, as DCs and macrophages are potent producers of IL-12 and express CD11b. The BM is a complex tissue as it not only contains these subgroups but also their progenitors and a complex set of downstream putative populations. However, whether dietary restriction directly redistributes macrophages or DCs into the BM is still unknown, as most dietary studies have focused on the relocation and function of adipose tissue macrophages, suggesting that they accumulate in areas with increased lipids.63,64 Dietary restriction increases adipogenesis in the BM of mice18,65; thus, it is interesting to consider whether this increased adipogenesis may be responsible for an induction of IL-12-producing macrophages. Furthermore, a potential role of inflammatory macrophages is to metabolize lipids63; thus, increased IL-12-producing macrophages may be associated with increased lipid content within the BM. Additionally, fasting can modulate the gut bacteria flora and enrich for lactobacillus species,66 some strains of which can induce IL-12 production from macrophages.67,68,69 Because changes in gut microbiota can influence BM production of inflammatory cell types,70 we can speculate that these alterations may be responsible for the increased IL-12 production we observe in our model.
Altogether, our work recognizes a critical role for the innate immune system in the improved anti-tumor response induced by fasting. This work will be important in dissecting the immune landscape in patients from clinical trials using fasting and fasting-mimicking diets, given that NK cell infiltration and function have yet to be fully appreciated. Furthermore, we show that cyclic fasting can remodel different tissues and thus represents a novel method of improving tissue-specific NK cell function and enhancing innate immunity for the treatment of solid and metastatic cancers. Introduction
Natural killer (NK) cells are circulating cytotoxic lymphocytes that are crucial to host immunity against cancer via the secretion of lytic granules and the production of effector cytokines such as interferon (IFN)-γ. Unlike antigen-specific cytotoxic CD8+ T cells that require tumor antigen presentation by major histocompatibility complex (MHC) molecules, NK cells possess a broad range of germline-encoded activating receptors to target transformed or malignant cells.1 In this way, tumors that have decreased expression of MHC class I to evade T cell killing can be targeted by NK cells.2 Indeed, in clinical studies, the presence of an “NK cell signature” or increase in NK cells within the tumor is predictive of improved survival of patients across a broad range of cancers, including melanoma, breast cancer, and gastric cancer.3,4,5,6 However, the potential to harness NK cells for therapeutic use is limited, as their survival and functionality in the hostile tumor microenvironment (TME) are often suppressed. The TME is hypoxic, rich in lipids and lactic acid, but depleted of glucose, all of which suppress immune cell function and survival and promote tumor growth.7,8 Because NK cells primarily rely on glycolysis for homeostasis and effector functions,9,10,11,12 identifying strategies that reprogram NK cell metabolism to overcome the metabolically immunosuppressive TME is essential to develop novel anti-tumor therapies.
Dietary restriction in the form of fasting or fasting-mimicking diets has garnered significant interest due to its profound effects on metabolism, cancer incidence, and anti-tumor immunity.13 Cyclic fasting impacts tumor cells by inducing sensitivity to chemotherapy and promoting an anti-Warburg effect.14,15 As such, clinical trials implementing these diets alongside various standard-of-care cancer therapies have yielded exciting and promising results, including improved tumor responsiveness to therapy and enhanced immune responses.16,17 Indeed, dietary restriction influences the redistribution and function of immune populations throughout the host, including T cells, B cells, and myeloid cells, and is associated with enhanced immunity.18,19,20,21,22
Here, we examined the impact of dietary restriction on NK cell function. We found that cyclic fasting remodels distinct tissue-specific microenvironments that reprogram the metabolic and functional state of NK cells to enhance anti-tumor immunity and reveal a critical role for NK cells in improved anti-tumor responses induced by fasting. Our findings suggest that NK cell function should be examined in clinical trials employing fasting regimens and that cyclic or periodic fasting may exemplify a dietary strategy for improved innate immunity in the treatment of solid and metastatic cancers.
ResultsCyclic fasting promotes NK cell-mediated anti-tumor immunity
To explore the effects of dietary restriction on NK cell-mediated anti-tumor immune responses, we developed a cyclic fasting diet (CFD) that is implemented for 3 weeks (Figure 1A). This diet was maintained without net weight loss in healthy or in tumor-bearing mice at endpoint when compared with mice fed ad libitum (Figures S1A and S1B). Adherence to a CFD improved the anti-tumor response against MC38 colon carcinoma (Figure 1B) and B16F10 melanoma metastasis (Figure 1C). B16F10 melanoma was measured using lung metastasis counts as well as lung weight as a measurement of tumor burden because, in some cases, the number of metastases was too high to be enumerated accurately. Improved MC38 and B16F10 anti-tumor immunity was dependent on NK cell activity, as depleting NK cells reduced the effects of a CFD on MC38 and B16F10 tumor growth, indicating broad protection of CFD-induced NK cell anti-tumor immunity against both cancers (Figures 1D and 1E). Although many NK cell ligands remained unchanged in the fasted state (Figure S1C), we observed a CFD-mediated decrease in MHC class I expression (Figure 1F), in agreement with the enhanced control of tumors mediated via “missing self” recognition by NK cells. Tumor-infiltrating lymphocyte (TIL) analysis of CFD and ad libitum mice injected with either tumor did not show any major alterations in absolute NK cell numbers (Figures 1G and 1H) or NK cell maturation (Figures S1D and S1E). Further, TIL analysis showed no significant changes in other lymphocyte, myeloid, or dendritic cell (DC) populations in both tumor models (Figures S1F–S1K). We also assessed the functionality of CD8+ T cells, CD4+ T cells, and tumor-associated macrophages (TAMs) and found functional capacity to be relatively unchanged by a CFD (Figures S2A–S2C). However, the CFD did enhance peripheral and tumor-infiltrating NK cell function, as assessed by IFN-γ production and degranulation (Figures 1I, 1J, S2D, and S2E).

Figure 1 Cyclic fasting promotes NK cell-mediated anti-tumor immunity
Show full captionFigure viewer
Nutrient restriction induces metabolic changes to maintain survival of NK cells
To investigate underlying molecular mechanisms initiating improved NK cell function and tumor control during a CFD, we focused on cellular changes that occur during a singular, acute fasting period. We profiled multiple lymphoid and myeloid cell populations and found that the majority of these cell populations were significantly decreased in the spleen of mice after a 48 h fast (Figure S3A). Because MHC class I expression was altered on tumor cells (Figure 1F), we shifted our focus toward NK cells and their adaptive cytotoxic counterparts, CD8+ T cells (Figures 2A and 2B). Of note, human peripheral blood NK cells were also decreased in this setting of a single acute fast (Figure S3B). Although we observed an increase in cell death in CD8+ T cells, NK cells did not show this same level of cell death at 48 h (Figure 2C). Aligned with this finding, we observed increased levels of apoptosis in CD8+ T cells when compared with NK cells early in the fasting window (0–12 h; Figures S3C and S3D), suggesting NK cells can adapt to changes in nutrient availability by using alternative mechanisms to ensure their survival.

Figure 2 Nutrient restriction induces metabolic changes to maintain survival of NK cells
Show full captionFigure viewer
Acute periods of fasting are known to cause a myriad of systemic metabolic changes that may affect NK cell survival.23 We observed that fasted mice displayed a sharp decline in serum glucose concentrations within 24 h, coinciding with a significant increase in free fatty acid (FA) concentrations (Figure 2D). Because homeostatic maintenance of both innate and adaptive lymphocytes primarily relies on glucose to fuel basal levels of oxidative phosphorylation (OXPHOS),9,24 we performed Seahorse metabolic flux assays on NK cells and total CD8+ T cells from ad libitum or fasted mice. Although both CD8+ T cells and NK cells showed a marked reduction in glycolysis and glycolytic capacity (Figures S3E and S3F), only fasted NK cells were surprisingly able to maintain their capacity for OXPHOS (Figures 2E and 2F). Alternative metabolic substrates, including FAs, are also known to fuel OXPHOS,25 and NK cells and CD8+ T cells from fasted mice showed increased uptake of FAs (Figures 2G and 2H). At the same time, NK cells from fasted mice also displayed a more prominent reduction in neutral lipids compared with CD8+ T cells (Figures 2I and S3G), indicating that increased FA uptake did not result in a net increase in lipid storage within lipid droplets (LDs). This decrease in neutral lipids may also suggest that the liberation of stored FAs can be used to fuel OXPHOS,26 and, as such, we observed increased autophagy in NK cells but not CD8+ T cells in the fasted state (Figures 2J and S3H). To further strengthen our findings, we inhibited FA oxidation and observed that NK cells from fasted mice were unable to maintain OXPHOS compared with ad libitum-fed controls (Figures 2K and S3I). Altogether, these data indicate that NK cells utilize FAs as an energy source to fuel OXPHOS during times of glucose withdrawal, ensuring their survival.
Cyclic fasting metabolically reprograms NK cells
Because NK cells showed a remarkable capacity to switch to FA use during fasting, we further investigated whether NK cells from mice that had undergone a CFD showed similar changes. We found that splenic NK cells from mice subjected to a CFD displayed an increased capacity to uptake FAs from the local environment (Figure 3A) but did not show an accumulation of lipids (Figure 3B). This coincided with a slight decrease in glucose uptake (Figure S4A). To gain insight into how this metabolic switch affects the transcriptomic profile of fasted NK cells, we performed single-cell RNA sequencing (scRNA-seq) on NK cells from tumor-bearing mice subjected to either a CFD or ad libitum feeding. Uniform manifold approximation and projection (UMAP) embedding suggested that fasted NK cells were distinct from their ad libitum-fed counterparts (Figure 3C). We then used unbiased louvain clustering and identified 9 distinct clusters shared between ad libitum and CFD NK cells (Figure S4B). Among the 9 clusters, clusters 0 and 4 were enriched for NK cells from CFD mice (Figure S4C). Gene set enrichment analysis (GSEA) performed on differential gene expression identified by MAST between ad libitum and CFD NK cells showed an upregulation of FA metabolism genes in CFD NK cells (Figure 3D), which may be primarily driven by CFD-enriched clusters, consistent with our previous observations (Figures S4B–S4D). Select genes known to be critical for FA metabolism were upregulated in CFD NK cells, including Cpt1a, encoding the critical enzyme that drives FA metabolism (Figure 3E). We also observed a decrease in the expression of Ldha, an important gene required to maintain glycolysis (Figure S3E). Together, these data suggest that a CFD pushes splenic NK cells toward a reprogrammed metabolic state involving increased FA metabolism and decreased glycolysis.

Figure 3 Cyclic fasting metabolically reprograms NK cells
Show full captionFigure viewer
Interestingly, in addition to FA metabolism, some of the most significantly upregulated differentially expressed genes in CFD NK cells were genes known to be induced by glucocorticoids (GCs; Figure 3F). This increase in GC receptor (GR) target gene expression (Figure 3G) coincided with a spike in GC concentration at both 24 and 48 h in the spleen and serum of fasted mice compared with those that were fed ad libitum (Figure 3H). GCs can induce FA metabolism in cardiomyocytes and acute lymphoblastic leukemia27,28; however, whether this occurs in immune cells has not been established. We treated mouse and human NK cells with the synthetic GC, dexamethasone (Dex), and found that a 24 h treatment of NK cells from both species with Dex increased CPT1A expression (Figures 3I and S4F). The induction was evident as early as 2 h (Figure S4G), demonstrating a rapid and sustained induction of CPT1A by GCs. Taken together, these data suggest that increased GC levels during periods of fasting drive expression of CPT1A, inducing FA oxidation in NK cells and thus promoting optimal metabolism depending on specific fuel availability.
CFD-induced FA oxidation improves the NK cell anti-tumor response
The TME can promote tumor growth while dampening TIL functions by limiting glucose, a metabolite required for optimal T cell anti-tumor function,29 and enriching for immunosuppressive lipids.30 Because NK cells from mice subjected to a CFD showed increased expression of CPT1A, we hypothesized that a preference for using FAs instead of glucose within the TME could improve NK cell function. Indeed, tumor-infiltrating NK cells from CFD mice showed increased CPT1A levels and increased FA uptake when compared with ad libitum-fed mice (Figures 4A and 4B). To study the effects of Cpt1a deletion on NK cells in vivo, we used Cpt1aflox/flox mice31 crossed with Ncr1iCre mice to generate mice that harbor an NK cell-specific deletion of CPT1A (NK-Cpt1a−/−; Figure S5A).32 CPT1A is required for the uptake of long-chain FAs into the mitochondria for FA oxidation (Figure 4C), and, as such, we measured the effects of a CFD on FA oxidation and anti-tumor function. NK-Cpt1a−/− and wild-type (WT) NK-Cpt1a+/+ littermate controls were challenged with tumors and subjected to either ad libitum or CFD conditions. Remarkably, NK cells deficient in Cpt1a were not able to control tumor growth in the CFD setting when compared with Cpt1a-sufficient NK cells (Figures 4D and S5B). The striking difference in tumor volume was associated with a significant decrease in the number of intra-tumoral Cpt1a−/− NK cells when compared with Cpt1a+/+ NK cells in CFD mice (Figures 4E and S5C). This reduction was linked to diminished expression of the anti-apoptotic protein BCL2 when compared with NK-Cpt1a+/+ controls (Figure 4F). Expression of the pro-apoptotic protein BIM was maintained (Figure 4G), increasing the BIM:BCL2 ratio and indicating that the balance of these proteins tipped in favor of cell death within NK cells that lack Cpt1a (Figure 4H). Tumor-infiltrating Cpt1a-deficient NK cells showed similar levels of FA uptake compared with control NK cells (Figure 4I), suggesting that although Cpt1a-deficient NK cells cannot perform mitochondrial FA oxidation, FA uptake is not disrupted. However, Cpt1a-deficient NK cells did show a marked reduction in effector function during a CFD (Figures 4J and S5D), suggesting that an inability to efficiently process intracellular FAs during fasting hampers NK cell survival and anti-tumor function, disrupting NK cell-mediated control of metastatic disease.

Figure 4 CFD-induced FA oxidation improves NK cell anti-tumor immunity
Show full captionFigure viewer
S1PR5 and CXCR4 govern redistribution of NK cells to the BM during fasting
Because acute periods of fasting resulted in decreased NK cell numbers in the spleen and blood without any significant changes in cell death (Figures 2A–2C and S3C), we reasoned that they could be accumulating in other organs in this setting (Figure S5E). Indeed, a significant increase in NK cells was observed in the bone marrow (BM) after 48 h of fasting (Figure 5A). The accumulation of NK cells in the BM during fasting coincided with a loss in blood and peripheral tissues and was followed by a restoration of NK cell numbers within 24 h of unrestricted refeeding (Figure 5B). These rapid and fluid changes suggested that a proportion of peripheral NK cells were redistributed to the BM during periods of fasting. To confirm this, we transferred mature CD45.1-expressing splenic NK cells into CD45.2-expressing recipient mice that were either fasted for 48 h or fed ad libitum. The transferred NK cells were recovered at reduced numbers in the spleen and blood and increased numbers in the BM (Figure 5C). In addition, we used the iNkp46tdTomato mouse model to “time stamp” NK cells by inducing expression of tdTomato (Tom+), permanently marking mature peripheral NK cells.33 Similar to the adoptive transfer experiment, Tom+ NK cells were redistributed to the BM during periods of fasting (Figure 5D). Together, these data support that while a population of NK cells remains in peripheral organs such as the spleen during fasting, some mature NK cells are preferentially redistributed to the BM.

Figure 5 Fasting promotes NK cell redistribution to the BM
Show full captionFigure viewer
In contrast to the metabolically reprogrammed NK cells recovered from the spleen of mice subjected to 48 h of fasting, NK cells from the BM showed a reduction in the uptake of FAs and CPT1A expression (Figures 5E and 5F). These changes coincided with reduced levels of GCs in the BM compared with serum and splenic levels when compared with ad libitum-fed controls (Figure 5G). Similarly, NK cells from the BM of mice subjected to a CFD showed reduced FA uptake but maintained glucose uptake (Figures S5F and S5G). The BM has previously been suggested as a niche that supports lymphocyte survival during periods of nutrient deprivation,18,19,20 and, altogether, our data suggest that the tissue-specific localization of NK cells during fasting may have alternate effects on metabolic and functional capacity.
To understand the consequences of blocking entry into the BM niche during a CFD, we determined which signal(s) were driving NK cells into the BM during periods of fasting. We observed increased numbers of Ter119-expressing red blood cells (RBCs) in the BM during fasting compared with ad libitum-fed controls (Figure S6A). Increased RBCs in the BM are a phenomenon previously reported during dietary restriction and may be a consequence of increased GCs in the periphery.18,34,35 RBCs are a major source of sphingosine-1-phosphate (S1P), a molecule important for lymphocyte trafficking.36,37 NK cells express high levels of S1PR5, a receptor required for efficient NK cell trafficking through lymph nodes and the BM.38,39 We used a knockin S1pr5eGFP-T2A-iCre reporter mouse40 and found that S1pr5eGFP expression increased in the BM over 48 h of fasting (Figure 6A), suggesting that an increase of RBCs might also increase the local S1P concentration in the BM.

Figure 6 Redistribution of NK cells to the BM during a CFD promotes tumor control
Show full captionFigure viewer
We compared NK cell numbers in the BM of reconstituted S1pr5+/+ and S1pr5−/− mixed BM chimeras (Figure S6B) that had been subjected to 48 h fasting and found that fasting-mediated NK cell accumulation in the BM was reduced in the absence of S1PR5 (Figure 6B). Because S1pr5−/− mice have increased numbers of NK cells in the BM under ad libitum conditions, the differences between fasted and ad libitum-fed mice were compared within genotypes and presented as fold-change (fasted/average in ad libitum). An increase in mature CD27−CD11b+ NK cells was observed in the BM after fasting, and S1pr5eGFP expression was also significantly increased in this population (Figures S6C and S6D). Deletion of S1PR5 selectively limited the entry of CD27−CD11b+ NK cells into the BM after 48 h of fasting (Figure 6C). To ensure that the loss of cells in the BM was due to defective trafficking, we transferred equal numbers of WT and S1pr5−/− NK cells into recipient mice (Figure S6E) that were either fasted for 48 h or fed ad libitum and found reduced numbers of S1pr5−/− NK cells in the BM compared with S1PR5-expressing WT NK cells (Figure S6F), altogether suggesting the S1P-S1PR5 axis is required for CFD-mediated NK cell migration into the BM during fasting.
Because NK cells and other lymphocytes enter and exit the BM through sinusoids that are connected to blood circulation,41,42 we incorporated an intravenous CD45 labeling technique43 to discriminate between sinusoid- and parenchyma-resident cells. CD45 labeling revealed a stepwise increase in parenchymal NK cells and a concomitant reduction in sinusoidal NK cells within 48 h of fasting (Figures 6D and S6G). Given that CXCR4 expression is tightly linked to NK cell retention in the parenchyma,41 we hypothesized that CXCR4 may also be integral to BM entry/exit during fasting. We compared the fold-change increase in BM NK cells from fasted mice treated with or without the CXCR4 antagonist, AMD3100, to ad libitum-fed mice and found a reduced capacity for NK cells to enter the BM (Figure 6E). Altogether, these data suggest that BM entry during fasting is mediated by an interplay of CXCR4-mediated parenchymal retention and S1P-guided NK cell accumulation.
Redistribution of NK cells to the BM during CFD promotes tumor control
To determine the effects of blocking NK cell entry into the BM on CFD-mediated tumor control, we challenged either S1pr5−/− mice or mice treated with AMD3100 with tumor cells and subjected them to either a CFD or ad libitum feeding (Figures S6H and S6I). Blocking fasting-induced BM entry of NK cells via either method abrogated CFD-mediated anti-tumor control (Figures 6F, 6G, and S6J–S6L). We next profiled the tumors of S1pr5−/− mice or mice treated with AMD3100 subjected to a CFD and found no differences in the total numbers of NK cells found in the TME (Figures 6H, 6I, and S6M). Furthermore, NK cells from the tumor and spleen no longer possessed CFD-mediated differences in IFN-γ production (Figures 6J, 6K, and S6N). Altogether, these data suggest that BM entry may promote the CFD-facilitated anti-tumor effector function observed in NK cells.
The fasted BM niche promotes NK cell priming via IL-12 production
To understand why NK cell redistribution to the BM was important for CFD-mediated anti-tumor responses, we performed scRNA-seq on NK cells from the BM of mice that were subjected to either CFD or ad libitum feeding (Figure S7A). NK cells were partially separated based on whether they had been subjected to a CFD or ad libitum feeding using UMAP projections (Figure 7A). We found several interleukin (IL)-12 pathway genes, such as Stat4, upregulated in NK cells from the BM of CFD mice (Figure 7B). We also observed upregulation of STAT4 signaling components JAK2 and TYK2, and a number of transcription factors that are induced by STAT444 (Figures S7B and S7C). Coinciding with the induction of IL-12 and STAT4 target genes in NK cells, we found increased IL-12 concentrations in the BM after 48 h fasting and reduction in the spleen and serum (Figure 7C). This coincided with an increase in IL-12-producing cells in the BM compared with the spleen (Figures 7D and S7D). IL-12+ cells in the BM expressed CD11b (Figures S7E and S7F) and were negative for lymphoid lineage markers CD19, CD3, TCRβ, and NK1.1 (Figure S7G), suggesting that the cell population is of myeloid origin. Because IL-12 is a potent stimulator of IFN-γ secretion in NK cells,45 we determined whether the increase in BM IL-12 during fasting could functionally prime NK cells. Indeed, NK cells from the BM of CFD mice showed a significant increase in IFN-γ production (Figure 7E). Furthermore, NK cells from the spleen of CFD mice that had been refed (thus redistributing NK cells that were previously in the BM back to the spleen) also secreted more IFN-γ (Figure 7F).

Figure 7 The fasted BM niche promotes NK cell priming via IL-12 production
Show full captionFigure viewer
To assess whether a link between fatty acid oxidation and IL-12 priming exists, we tested whether NK cells lacking fatty acid oxidation capacity still showed BM-specific reprogramming. After 48 h of fasting, we found NK-Cpt1a−/− NK cells redistributed to the BM in a similar manner to WT NK-Cpt1a+/+ NK cells (Figure S7H). In agreement with intact redistribution during fasting, NK cells from NK-Cpt1a−/− mice subjected to a CFD showed similar increases in IFN-γ production in the BM when compared with ad libitum-fed controls (Figure S7I). These findings suggest that the inability of NK cells in the BM to perform fatty acid oxidation does not impair IL-12 priming and subsequent IFN-γ production. Altogether, these data suggest that, during periods of fasting, the BM niche is specifically reshaped to provide increased local concentrations of IL-12, priming NK cells that have preferentially redistributed to the BM and inducing local and peripheral IFN-γ secretion by NK cells important for anti-tumor immunity.
Discussion
Dietary restriction in the form of fasting and limiting caloric intake redistributes both adaptive and innate immune cell subsets such as B cells, T cells, and myeloid cells.18,19,20 Because NK cells are often placed at the intersection of innate and adaptive immunity, they represent a unique cell type that possesses features of both.46 Unlike adaptive T and B cells, NK cells act as a first line of defense against infections and tumor metastasis, and unlike innate myeloid cells, which can become detrimental in the TME as myeloid-derived suppressor cells, NK cell presence within tumors is almost always associated with better prognosis.6,47,48 Here, we show evidence that tissue repositioning during periods of fasting is advantageous for peripheral NK cell function, poising them for enhanced metabolic status and survival in the TME.
A significant advantage that NK cells have over T cells in the context of fasting is their response to GCs. Unlike T cells, which are sensitive to high levels of endogenous GCs that cause excessive cell death,18,49 we found that NK cells did not undergo greater death in response to GC exposure in vivo and in vitro. Instead, GCs promoted upregulation of CPT1A for increased FA oxidation capability, a phenomenon also observed in cardiomyocytes and muscle.27,50 Like M2 macrophages, NK cells increase lipid uptake, OXPHOS, and FA oxidation.51 Their ability to metabolically adapt in conditions of high GCs and excess FAs may be a contributing factor that distinguishes the innate functionality of NK cells from T or B cells in fasting conditions.
Metabolic rewiring in several cell types can be induced by nutrient restriction, shifting cellular metabolism from reliance on glycolysis to dependence on mitochondrial FA oxidation.26 However, this specific adaptation has not been mechanistically characterized in NK cells. Although FA oxidation was recently shown to be crucial for NK cell antiviral responses,32 much of our understanding of lipid uptake has been in the context of obesity and NK cell dysfunction, and thus the use of FAs as an energy source during cyclic fasting to improve functional responses against cancer has not been previously appreciated. In obese individuals, NK cells show an accumulation of lipids, which is correlated with reduced functionality.52 Similarly, in a mouse model of lymphoma, NK cell dysfunction is correlated with lipid accumulation and increased lipid metabolism.53 Unlike these previous studies, our work shows that cyclic fasting results in increased NK cell FA oxidation without lipid accumulation, suggesting that fasting might be an optimal approach to utilize excess lipids.
Both the obesity and lymphoma environments raise the interesting observation that NK cells in these conditions are continuously exposed to lipid-rich environments. By contrast, our model of a CFD, where a proportion of NK cells are acutely and intermittently exposed to high concentrations of FAs, represents a scenario where NK cells are constantly being shifted from glucose-depleted, FA-rich environments back to baseline during each period of fasting/re-feeding. Whether cyclic fasting can improve NK cell function in the continuously lipid-laden environments found in obese mice or humans requires further investigation. Of note, many prior and current studies, including our own here, do not assess NK cell function and FA oxidation capacity in adipose tissue. Our recent work suggests that NK cells have the ability to conditionally alter their metabolism based on the tissue in which they reside54; thus, future studies will seek to understand such additional tissue-specific roles of fasting, particularly because adipose-residing innate lymphocytes have metabolic and functional adaptations in obesity-induced disease settings.55,56,57,58
Acute myeloid leukemia (AML) is an aggressive hematological malignancy where improved outcomes are strongly associated with increased NK cell cytolytic activity.59,60,61 As such, adoptive NK cell therapies in the clinic have primarily been targeted to AML.8 Thus, whether AML maintains a lipid-rich BM environment similar to lymphoma is of interest, as the BM can also represent a site of tumor growth. In AML, the effects of cyclic fasting may be opposing. On the one hand, NK cells are preferentially redistributed to the BM during CFD conditions, where they are primed to produce more IFN-γ, which could promote leukemia tumor control. On the other hand (and as we have also observed in our study), cyclic fasting does not appear to increase FA uptake or LD abundance in NK cells in the BM, even after multiple cycles of fasting. Because our study does not address cancers of BM origin, future studies will investigate how fasting-induced NK cell responses to leukemias may differ from the solid tumor and metastasis models used in this study.
We observed that cyclic fasting improved NK cell functionality in a tissue-specific manner via parallel but disparate processes occurring in the BM versus the spleen. In mice, the half-life of an NK cell is ∼19.4 days, and thus adherence to 3 weeks of a CFD would assume that the NK cells profiled at tumor endpoint are heterogeneous in age33,62 and certainly harboring a proportion of cells that have cycled through more than one round of fasting and refeeding. Although we showed that NK cells are redistributed from the spleen and blood to the BM, we recognize that there are technological limitations with the current tools available to determine whether the same NK cell population has preferentially redistributed to the BM during each fasting cycle and, thus, whether an individual NK cell has been exposed to one or both tissue-specific processes. We provide evidence in our study that disrupting either process (i.e., FA oxidation via CPT1A deletion or BM trafficking via S1PR5 deletion or CXCR4 antagonism) has profound consequences on NK cell anti-tumor function, emphasizing the contribution of both BM entry and splenic reprogramming. Future cell-tracing studies may be able to determine whether NK cells found in the tumor have experienced fasting-induced signals from one or both tissue sites.
IL-12-producing CD45+CD11b+ cells were increased in the BM after adherence to cyclic fasting, suggesting that the effects of fasting can impose indirect consequences on NK cells. Although we have not established which subset of CD11b-expressing cells is producing IL-12, we can speculate that it may be derived from a subgroup of the myeloid lineage, as DCs and macrophages are potent producers of IL-12 and express CD11b. The BM is a complex tissue as it not only contains these subgroups but also their progenitors and a complex set of downstream putative populations. However, whether dietary restriction directly redistributes macrophages or DCs into the BM is still unknown, as most dietary studies have focused on the relocation and function of adipose tissue macrophages, suggesting that they accumulate in areas with increased lipids.63,64 Dietary restriction increases adipogenesis in the BM of mice18,65; thus, it is interesting to consider whether this increased adipogenesis may be responsible for an induction of IL-12-producing macrophages. Furthermore, a potential role of inflammatory macrophages is to metabolize lipids63; thus, increased IL-12-producing macrophages may be associated with increased lipid content within the BM. Additionally, fasting can modulate the gut bacteria flora and enrich for lactobacillus species,66 some strains of which can induce IL-12 production from macrophages.67,68,69 Because changes in gut microbiota can influence BM production of inflammatory cell types,70 we can speculate that these alterations may be responsible for the increased IL-12 production we observe in our model.
Altogether, our work recognizes a critical role for the innate immune system in the improved anti-tumor response induced by fasting. This work will be important in dissecting the immune landscape in patients from clinical trials using fasting and fasting-mimicking diets, given that NK cell infiltration and function have yet to be fully appreciated. Furthermore, we show that cyclic fasting can remodel different tissues and thus represents a novel method of improving tissue-specific NK cell function and enhancing innate immunity for the treatment of solid and metastatic cancers. Introduction
Natural killer (NK) cells are circulating cytotoxic lymphocytes that are crucial to host immunity against cancer via the secretion of lytic granules and the production of effector cytokines such as interferon (IFN)-γ. Unlike antigen-specific cytotoxic CD8+ T cells that require tumor antigen presentation by major histocompatibility complex (MHC) molecules, NK cells possess a broad range of germline-encoded activating receptors to target transformed or malignant cells.1 In this way, tumors that have decreased expression of MHC class I to evade T cell killing can be targeted by NK cells.2 Indeed, in clinical studies, the presence of an “NK cell signature” or increase in NK cells within the tumor is predictive of improved survival of patients across a broad range of cancers, including melanoma, breast cancer, and gastric cancer.3,4,5,6 However, the potential to harness NK cells for therapeutic use is limited, as their survival and functionality in the hostile tumor microenvironment (TME) are often suppressed. The TME is hypoxic, rich in lipids and lactic acid, but depleted of glucose, all of which suppress immune cell function and survival and promote tumor growth.7,8 Because NK cells primarily rely on glycolysis for homeostasis and effector functions,9,10,11,12 identifying strategies that reprogram NK cell metabolism to overcome the metabolically immunosuppressive TME is essential to develop novel anti-tumor therapies.
Dietary restriction in the form of fasting or fasting-mimicking diets has garnered significant interest due to its profound effects on metabolism, cancer incidence, and anti-tumor immunity.13 Cyclic fasting impacts tumor cells by inducing sensitivity to chemotherapy and promoting an anti-Warburg effect.14,15 As such, clinical trials implementing these diets alongside various standard-of-care cancer therapies have yielded exciting and promising results, including improved tumor responsiveness to therapy and enhanced immune responses.16,17 Indeed, dietary restriction influences the redistribution and function of immune populations throughout the host, including T cells, B cells, and myeloid cells, and is associated with enhanced immunity.18,19,20,21,22
Here, we examined the impact of dietary restriction on NK cell function. We found that cyclic fasting remodels distinct tissue-specific microenvironments that reprogram the metabolic and functional state of NK cells to enhance anti-tumor immunity and reveal a critical role for NK cells in improved anti-tumor responses induced by fasting. Our findings suggest that NK cell function should be examined in clinical trials employing fasting regimens and that cyclic or periodic fasting may exemplify a dietary strategy for improved innate immunity in the treatment of solid and metastatic cancers.
ResultsCyclic fasting promotes NK cell-mediated anti-tumor immunity
To explore the effects of dietary restriction on NK cell-mediated anti-tumor immune responses, we developed a cyclic fasting diet (CFD) that is implemented for 3 weeks (Figure 1A). This diet was maintained without net weight loss in healthy or in tumor-bearing mice at endpoint when compared with mice fed ad libitum (Figures S1A and S1B). Adherence to a CFD improved the anti-tumor response against MC38 colon carcinoma (Figure 1B) and B16F10 melanoma metastasis (Figure 1C). B16F10 melanoma was measured using lung metastasis counts as well as lung weight as a measurement of tumor burden because, in some cases, the number of metastases was too high to be enumerated accurately. Improved MC38 and B16F10 anti-tumor immunity was dependent on NK cell activity, as depleting NK cells reduced the effects of a CFD on MC38 and B16F10 tumor growth, indicating broad protection of CFD-induced NK cell anti-tumor immunity against both cancers (Figures 1D and 1E). Although many NK cell ligands remained unchanged in the fasted state (Figure S1C), we observed a CFD-mediated decrease in MHC class I expression (Figure 1F), in agreement with the enhanced control of tumors mediated via “missing self” recognition by NK cells. Tumor-infiltrating lymphocyte (TIL) analysis of CFD and ad libitum mice injected with either tumor did not show any major alterations in absolute NK cell numbers (Figures 1G and 1H) or NK cell maturation (Figures S1D and S1E). Further, TIL analysis showed no significant changes in other lymphocyte, myeloid, or dendritic cell (DC) populations in both tumor models (Figures S1F–S1K). We also assessed the functionality of CD8+ T cells, CD4+ T cells, and tumor-associated macrophages (TAMs) and found functional capacity to be relatively unchanged by a CFD (Figures S2A–S2C). However, the CFD did enhance peripheral and tumor-infiltrating NK cell function, as assessed by IFN-γ production and degranulation (Figures 1I, 1J, S2D, and S2E).

Figure 1 Cyclic fasting promotes NK cell-mediated anti-tumor immunity
Show full captionFigure viewer
Nutrient restriction induces metabolic changes to maintain survival of NK cells
To investigate underlying molecular mechanisms initiating improved NK cell function and tumor control during a CFD, we focused on cellular changes that occur during a singular, acute fasting period. We profiled multiple lymphoid and myeloid cell populations and found that the majority of these cell populations were significantly decreased in the spleen of mice after a 48 h fast (Figure S3A). Because MHC class I expression was altered on tumor cells (Figure 1F), we shifted our focus toward NK cells and their adaptive cytotoxic counterparts, CD8+ T cells (Figures 2A and 2B). Of note, human peripheral blood NK cells were also decreased in this setting of a single acute fast (Figure S3B). Although we observed an increase in cell death in CD8+ T cells, NK cells did not show this same level of cell death at 48 h (Figure 2C). Aligned with this finding, we observed increased levels of apoptosis in CD8+ T cells when compared with NK cells early in the fasting window (0–12 h; Figures S3C and S3D), suggesting NK cells can adapt to changes in nutrient availability by using alternative mechanisms to ensure their survival.

Figure 2 Nutrient restriction induces metabolic changes to maintain survival of NK cells
Show full captionFigure viewer
Acute periods of fasting are known to cause a myriad of systemic metabolic changes that may affect NK cell survival.23 We observed that fasted mice displayed a sharp decline in serum glucose concentrations within 24 h, coinciding with a significant increase in free fatty acid (FA) concentrations (Figure 2D). Because homeostatic maintenance of both innate and adaptive lymphocytes primarily relies on glucose to fuel basal levels of oxidative phosphorylation (OXPHOS),9,24 we performed Seahorse metabolic flux assays on NK cells and total CD8+ T cells from ad libitum or fasted mice. Although both CD8+ T cells and NK cells showed a marked reduction in glycolysis and glycolytic capacity (Figures S3E and S3F), only fasted NK cells were surprisingly able to maintain their capacity for OXPHOS (Figures 2E and 2F). Alternative metabolic substrates, including FAs, are also known to fuel OXPHOS,25 and NK cells and CD8+ T cells from fasted mice showed increased uptake of FAs (Figures 2G and 2H). At the same time, NK cells from fasted mice also displayed a more prominent reduction in neutral lipids compared with CD8+ T cells (Figures 2I and S3G), indicating that increased FA uptake did not result in a net increase in lipid storage within lipid droplets (LDs). This decrease in neutral lipids may also suggest that the liberation of stored FAs can be used to fuel OXPHOS,26 and, as such, we observed increased autophagy in NK cells but not CD8+ T cells in the fasted state (Figures 2J and S3H). To further strengthen our findings, we inhibited FA oxidation and observed that NK cells from fasted mice were unable to maintain OXPHOS compared with ad libitum-fed controls (Figures 2K and S3I). Altogether, these data indicate that NK cells utilize FAs as an energy source to fuel OXPHOS during times of glucose withdrawal, ensuring their survival.
Cyclic fasting metabolically reprograms NK cells
Because NK cells showed a remarkable capacity to switch to FA use during fasting, we further investigated whether NK cells from mice that had undergone a CFD showed similar changes. We found that splenic NK cells from mice subjected to a CFD displayed an increased capacity to uptake FAs from the local environment (Figure 3A) but did not show an accumulation of lipids (Figure 3B). This coincided with a slight decrease in glucose uptake (Figure S4A). To gain insight into how this metabolic switch affects the transcriptomic profile of fasted NK cells, we performed single-cell RNA sequencing (scRNA-seq) on NK cells from tumor-bearing mice subjected to either a CFD or ad libitum feeding. Uniform manifold approximation and projection (UMAP) embedding suggested that fasted NK cells were distinct from their ad libitum-fed counterparts (Figure 3C). We then used unbiased louvain clustering and identified 9 distinct clusters shared between ad libitum and CFD NK cells (Figure S4B). Among the 9 clusters, clusters 0 and 4 were enriched for NK cells from CFD mice (Figure S4C). Gene set enrichment analysis (GSEA) performed on differential gene expression identified by MAST between ad libitum and CFD NK cells showed an upregulation of FA metabolism genes in CFD NK cells (Figure 3D), which may be primarily driven by CFD-enriched clusters, consistent with our previous observations (Figures S4B–S4D). Select genes known to be critical for FA metabolism were upregulated in CFD NK cells, including Cpt1a, encoding the critical enzyme that drives FA metabolism (Figure 3E). We also observed a decrease in the expression of Ldha, an important gene required to maintain glycolysis (Figure S3E). Together, these data suggest that a CFD pushes splenic NK cells toward a reprogrammed metabolic state involving increased FA metabolism and decreased glycolysis.

Figure 3 Cyclic fasting metabolically reprograms NK cells
Show full captionFigure viewer
Interestingly, in addition to FA metabolism, some of the most significantly upregulated differentially expressed genes in CFD NK cells were genes known to be induced by glucocorticoids (GCs; Figure 3F). This increase in GC receptor (GR) target gene expression (Figure 3G) coincided with a spike in GC concentration at both 24 and 48 h in the spleen and serum of fasted mice compared with those that were fed ad libitum (Figure 3H). GCs can induce FA metabolism in cardiomyocytes and acute lymphoblastic leukemia27,28; however, whether this occurs in immune cells has not been established. We treated mouse and human NK cells with the synthetic GC, dexamethasone (Dex), and found that a 24 h treatment of NK cells from both species with Dex increased CPT1A expression (Figures 3I and S4F). The induction was evident as early as 2 h (Figure S4G), demonstrating a rapid and sustained induction of CPT1A by GCs. Taken together, these data suggest that increased GC levels during periods of fasting drive expression of CPT1A, inducing FA oxidation in NK cells and thus promoting optimal metabolism depending on specific fuel availability.
CFD-induced FA oxidation improves the NK cell anti-tumor response
The TME can promote tumor growth while dampening TIL functions by limiting glucose, a metabolite required for optimal T cell anti-tumor function,29 and enriching for immunosuppressive lipids.30 Because NK cells from mice subjected to a CFD showed increased expression of CPT1A, we hypothesized that a preference for using FAs instead of glucose within the TME could improve NK cell function. Indeed, tumor-infiltrating NK cells from CFD mice showed increased CPT1A levels and increased FA uptake when compared with ad libitum-fed mice (Figures 4A and 4B). To study the effects of Cpt1a deletion on NK cells in vivo, we used Cpt1aflox/flox mice31 crossed with Ncr1iCre mice to generate mice that harbor an NK cell-specific deletion of CPT1A (NK-Cpt1a−/−; Figure S5A).32 CPT1A is required for the uptake of long-chain FAs into the mitochondria for FA oxidation (Figure 4C), and, as such, we measured the effects of a CFD on FA oxidation and anti-tumor function. NK-Cpt1a−/− and wild-type (WT) NK-Cpt1a+/+ littermate controls were challenged with tumors and subjected to either ad libitum or CFD conditions. Remarkably, NK cells deficient in Cpt1a were not able to control tumor growth in the CFD setting when compared with Cpt1a-sufficient NK cells (Figures 4D and S5B). The striking difference in tumor volume was associated with a significant decrease in the number of intra-tumoral Cpt1a−/− NK cells when compared with Cpt1a+/+ NK cells in CFD mice (Figures 4E and S5C). This reduction was linked to diminished expression of the anti-apoptotic protein BCL2 when compared with NK-Cpt1a+/+ controls (Figure 4F). Expression of the pro-apoptotic protein BIM was maintained (Figure 4G), increasing the BIM:BCL2 ratio and indicating that the balance of these proteins tipped in favor of cell death within NK cells that lack Cpt1a (Figure 4H). Tumor-infiltrating Cpt1a-deficient NK cells showed similar levels of FA uptake compared with control NK cells (Figure 4I), suggesting that although Cpt1a-deficient NK cells cannot perform mitochondrial FA oxidation, FA uptake is not disrupted. However, Cpt1a-deficient NK cells did show a marked reduction in effector function during a CFD (Figures 4J and S5D), suggesting that an inability to efficiently process intracellular FAs during fasting hampers NK cell survival and anti-tumor function, disrupting NK cell-mediated control of metastatic disease.

Figure 4 CFD-induced FA oxidation improves NK cell anti-tumor immunity
Show full captionFigure viewer
S1PR5 and CXCR4 govern redistribution of NK cells to the BM during fasting
Because acute periods of fasting resulted in decreased NK cell numbers in the spleen and blood without any significant changes in cell death (Figures 2A–2C and S3C), we reasoned that they could be accumulating in other organs in this setting (Figure S5E). Indeed, a significant increase in NK cells was observed in the bone marrow (BM) after 48 h of fasting (Figure 5A). The accumulation of NK cells in the BM during fasting coincided with a loss in blood and peripheral tissues and was followed by a restoration of NK cell numbers within 24 h of unrestricted refeeding (Figure 5B). These rapid and fluid changes suggested that a proportion of peripheral NK cells were redistributed to the BM during periods of fasting. To confirm this, we transferred mature CD45.1-expressing splenic NK cells into CD45.2-expressing recipient mice that were either fasted for 48 h or fed ad libitum. The transferred NK cells were recovered at reduced numbers in the spleen and blood and increased numbers in the BM (Figure 5C). In addition, we used the iNkp46tdTomato mouse model to “time stamp” NK cells by inducing expression of tdTomato (Tom+), permanently marking mature peripheral NK cells.33 Similar to the adoptive transfer experiment, Tom+ NK cells were redistributed to the BM during periods of fasting (Figure 5D). Together, these data support that while a population of NK cells remains in peripheral organs such as the spleen during fasting, some mature NK cells are preferentially redistributed to the BM.

Figure 5 Fasting promotes NK cell redistribution to the BM
Show full captionFigure viewer
In contrast to the metabolically reprogrammed NK cells recovered from the spleen of mice subjected to 48 h of fasting, NK cells from the BM showed a reduction in the uptake of FAs and CPT1A expression (Figures 5E and 5F). These changes coincided with reduced levels of GCs in the BM compared with serum and splenic levels when compared with ad libitum-fed controls (Figure 5G). Similarly, NK cells from the BM of mice subjected to a CFD showed reduced FA uptake but maintained glucose uptake (Figures S5F and S5G). The BM has previously been suggested as a niche that supports lymphocyte survival during periods of nutrient deprivation,18,19,20 and, altogether, our data suggest that the tissue-specific localization of NK cells during fasting may have alternate effects on metabolic and functional capacity.
To understand the consequences of blocking entry into the BM niche during a CFD, we determined which signal(s) were driving NK cells into the BM during periods of fasting. We observed increased numbers of Ter119-expressing red blood cells (RBCs) in the BM during fasting compared with ad libitum-fed controls (Figure S6A). Increased RBCs in the BM are a phenomenon previously reported during dietary restriction and may be a consequence of increased GCs in the periphery.18,34,35 RBCs are a major source of sphingosine-1-phosphate (S1P), a molecule important for lymphocyte trafficking.36,37 NK cells express high levels of S1PR5, a receptor required for efficient NK cell trafficking through lymph nodes and the BM.38,39 We used a knockin S1pr5eGFP-T2A-iCre reporter mouse40 and found that S1pr5eGFP expression increased in the BM over 48 h of fasting (Figure 6A), suggesting that an increase of RBCs might also increase the local S1P concentration in the BM.

Figure 6 Redistribution of NK cells to the BM during a CFD promotes tumor control
Show full captionFigure viewer
We compared NK cell numbers in the BM of reconstituted S1pr5+/+ and S1pr5−/− mixed BM chimeras (Figure S6B) that had been subjected to 48 h fasting and found that fasting-mediated NK cell accumulation in the BM was reduced in the absence of S1PR5 (Figure 6B). Because S1pr5−/− mice have increased numbers of NK cells in the BM under ad libitum conditions, the differences between fasted and ad libitum-fed mice were compared within genotypes and presented as fold-change (fasted/average in ad libitum). An increase in mature CD27−CD11b+ NK cells was observed in the BM after fasting, and S1pr5eGFP expression was also significantly increased in this population (Figures S6C and S6D). Deletion of S1PR5 selectively limited the entry of CD27−CD11b+ NK cells into the BM after 48 h of fasting (Figure 6C). To ensure that the loss of cells in the BM was due to defective trafficking, we transferred equal numbers of WT and S1pr5−/− NK cells into recipient mice (Figure S6E) that were either fasted for 48 h or fed ad libitum and found reduced numbers of S1pr5−/− NK cells in the BM compared with S1PR5-expressing WT NK cells (Figure S6F), altogether suggesting the S1P-S1PR5 axis is required for CFD-mediated NK cell migration into the BM during fasting.
Because NK cells and other lymphocytes enter and exit the BM through sinusoids that are connected to blood circulation,41,42 we incorporated an intravenous CD45 labeling technique43 to discriminate between sinusoid- and parenchyma-resident cells. CD45 labeling revealed a stepwise increase in parenchymal NK cells and a concomitant reduction in sinusoidal NK cells within 48 h of fasting (Figures 6D and S6G). Given that CXCR4 expression is tightly linked to NK cell retention in the parenchyma,41 we hypothesized that CXCR4 may also be integral to BM entry/exit during fasting. We compared the fold-change increase in BM NK cells from fasted mice treated with or without the CXCR4 antagonist, AMD3100, to ad libitum-fed mice and found a reduced capacity for NK cells to enter the BM (Figure 6E). Altogether, these data suggest that BM entry during fasting is mediated by an interplay of CXCR4-mediated parenchymal retention and S1P-guided NK cell accumulation.
Redistribution of NK cells to the BM during CFD promotes tumor control
To determine the effects of blocking NK cell entry into the BM on CFD-mediated tumor control, we challenged either S1pr5−/− mice or mice treated with AMD3100 with tumor cells and subjected them to either a CFD or ad libitum feeding (Figures S6H and S6I). Blocking fasting-induced BM entry of NK cells via either method abrogated CFD-mediated anti-tumor control (Figures 6F, 6G, and S6J–S6L). We next profiled the tumors of S1pr5−/− mice or mice treated with AMD3100 subjected to a CFD and found no differences in the total numbers of NK cells found in the TME (Figures 6H, 6I, and S6M). Furthermore, NK cells from the tumor and spleen no longer possessed CFD-mediated differences in IFN-γ production (Figures 6J, 6K, and S6N). Altogether, these data suggest that BM entry may promote the CFD-facilitated anti-tumor effector function observed in NK cells.
The fasted BM niche promotes NK cell priming via IL-12 production
To understand why NK cell redistribution to the BM was important for CFD-mediated anti-tumor responses, we performed scRNA-seq on NK cells from the BM of mice that were subjected to either CFD or ad libitum feeding (Figure S7A). NK cells were partially separated based on whether they had been subjected to a CFD or ad libitum feeding using UMAP projections (Figure 7A). We found several interleukin (IL)-12 pathway genes, such as Stat4, upregulated in NK cells from the BM of CFD mice (Figure 7B). We also observed upregulation of STAT4 signaling components JAK2 and TYK2, and a number of transcription factors that are induced by STAT444 (Figures S7B and S7C). Coinciding with the induction of IL-12 and STAT4 target genes in NK cells, we found increased IL-12 concentrations in the BM after 48 h fasting and reduction in the spleen and serum (Figure 7C). This coincided with an increase in IL-12-producing cells in the BM compared with the spleen (Figures 7D and S7D). IL-12+ cells in the BM expressed CD11b (Figures S7E and S7F) and were negative for lymphoid lineage markers CD19, CD3, TCRβ, and NK1.1 (Figure S7G), suggesting that the cell population is of myeloid origin. Because IL-12 is a potent stimulator of IFN-γ secretion in NK cells,45 we determined whether the increase in BM IL-12 during fasting could functionally prime NK cells. Indeed, NK cells from the BM of CFD mice showed a significant increase in IFN-γ production (Figure 7E). Furthermore, NK cells from the spleen of CFD mice that had been refed (thus redistributing NK cells that were previously in the BM back to the spleen) also secreted more IFN-γ (Figure 7F).

Figure 7 The fasted BM niche promotes NK cell priming via IL-12 production
Show full captionFigure viewer
To assess whether a link between fatty acid oxidation and IL-12 priming exists, we tested whether NK cells lacking fatty acid oxidation capacity still showed BM-specific reprogramming. After 48 h of fasting, we found NK-Cpt1a−/− NK cells redistributed to the BM in a similar manner to WT NK-Cpt1a+/+ NK cells (Figure S7H). In agreement with intact redistribution during fasting, NK cells from NK-Cpt1a−/− mice subjected to a CFD showed similar increases in IFN-γ production in the BM when compared with ad libitum-fed controls (Figure S7I). These findings suggest that the inability of NK cells in the BM to perform fatty acid oxidation does not impair IL-12 priming and subsequent IFN-γ production. Altogether, these data suggest that, during periods of fasting, the BM niche is specifically reshaped to provide increased local concentrations of IL-12, priming NK cells that have preferentially redistributed to the BM and inducing local and peripheral IFN-γ secretion by NK cells important for anti-tumor immunity.
Discussion
Dietary restriction in the form of fasting and limiting caloric intake redistributes both adaptive and innate immune cell subsets such as B cells, T cells, and myeloid cells.18,19,20 Because NK cells are often placed at the intersection of innate and adaptive immunity, they represent a unique cell type that possesses features of both.46 Unlike adaptive T and B cells, NK cells act as a first line of defense against infections and tumor metastasis, and unlike innate myeloid cells, which can become detrimental in the TME as myeloid-derived suppressor cells, NK cell presence within tumors is almost always associated with better prognosis.6,47,48 Here, we show evidence that tissue repositioning during periods of fasting is advantageous for peripheral NK cell function, poising them for enhanced metabolic status and survival in the TME.
A significant advantage that NK cells have over T cells in the context of fasting is their response to GCs. Unlike T cells, which are sensitive to high levels of endogenous GCs that cause excessive cell death,18,49 we found that NK cells did not undergo greater death in response to GC exposure in vivo and in vitro. Instead, GCs promoted upregulation of CPT1A for increased FA oxidation capability, a phenomenon also observed in cardiomyocytes and muscle.27,50 Like M2 macrophages, NK cells increase lipid uptake, OXPHOS, and FA oxidation.51 Their ability to metabolically adapt in conditions of high GCs and excess FAs may be a contributing factor that distinguishes the innate functionality of NK cells from T or B cells in fasting conditions.
Metabolic rewiring in several cell types can be induced by nutrient restriction, shifting cellular metabolism from reliance on glycolysis to dependence on mitochondrial FA oxidation.26 However, this specific adaptation has not been mechanistically characterized in NK cells. Although FA oxidation was recently shown to be crucial for NK cell antiviral responses,32 much of our understanding of lipid uptake has been in the context of obesity and NK cell dysfunction, and thus the use of FAs as an energy source during cyclic fasting to improve functional responses against cancer has not been previously appreciated. In obese individuals, NK cells show an accumulation of lipids, which is correlated with reduced functionality.52 Similarly, in a mouse model of lymphoma, NK cell dysfunction is correlated with lipid accumulation and increased lipid metabolism.53 Unlike these previous studies, our work shows that cyclic fasting results in increased NK cell FA oxidation without lipid accumulation, suggesting that fasting might be an optimal approach to utilize excess lipids.
Both the obesity and lymphoma environments raise the interesting observation that NK cells in these conditions are continuously exposed to lipid-rich environments. By contrast, our model of a CFD, where a proportion of NK cells are acutely and intermittently exposed to high concentrations of FAs, represents a scenario where NK cells are constantly being shifted from glucose-depleted, FA-rich environments back to baseline during each period of fasting/re-feeding. Whether cyclic fasting can improve NK cell function in the continuously lipid-laden environments found in obese mice or humans requires further investigation. Of note, many prior and current studies, including our own here, do not assess NK cell function and FA oxidation capacity in adipose tissue. Our recent work suggests that NK cells have the ability to conditionally alter their metabolism based on the tissue in which they reside54; thus, future studies will seek to understand such additional tissue-specific roles of fasting, particularly because adipose-residing innate lymphocytes have metabolic and functional adaptations in obesity-induced disease settings.55,56,57,58
Acute myeloid leukemia (AML) is an aggressive hematological malignancy where improved outcomes are strongly associated with increased NK cell cytolytic activity.59,60,61 As such, adoptive NK cell therapies in the clinic have primarily been targeted to AML.8 Thus, whether AML maintains a lipid-rich BM environment similar to lymphoma is of interest, as the BM can also represent a site of tumor growth. In AML, the effects of cyclic fasting may be opposing. On the one hand, NK cells are preferentially redistributed to the BM during CFD conditions, where they are primed to produce more IFN-γ, which could promote leukemia tumor control. On the other hand (and as we have also observed in our study), cyclic fasting does not appear to increase FA uptake or LD abundance in NK cells in the BM, even after multiple cycles of fasting. Because our study does not address cancers of BM origin, future studies will investigate how fasting-induced NK cell responses to leukemias may differ from the solid tumor and metastasis models used in this study.
We observed that cyclic fasting improved NK cell functionality in a tissue-specific manner via parallel but disparate processes occurring in the BM versus the spleen. In mice, the half-life of an NK cell is ∼19.4 days, and thus adherence to 3 weeks of a CFD would assume that the NK cells profiled at tumor endpoint are heterogeneous in age33,62 and certainly harboring a proportion of cells that have cycled through more than one round of fasting and refeeding. Although we showed that NK cells are redistributed from the spleen and blood to the BM, we recognize that there are technological limitations with the current tools available to determine whether the same NK cell population has preferentially redistributed to the BM during each fasting cycle and, thus, whether an individual NK cell has been exposed to one or both tissue-specific processes. We provide evidence in our study that disrupting either process (i.e., FA oxidation via CPT1A deletion or BM trafficking via S1PR5 deletion or CXCR4 antagonism) has profound consequences on NK cell anti-tumor function, emphasizing the contribution of both BM entry and splenic reprogramming. Future cell-tracing studies may be able to determine whether NK cells found in the tumor have experienced fasting-induced signals from one or both tissue sites.
IL-12-producing CD45+CD11b+ cells were increased in the BM after adherence to cyclic fasting, suggesting that the effects of fasting can impose indirect consequences on NK cells. Although we have not established which subset of CD11b-expressing cells is producing IL-12, we can speculate that it may be derived from a subgroup of the myeloid lineage, as DCs and macrophages are potent producers of IL-12 and express CD11b. The BM is a complex tissue as it not only contains these subgroups but also their progenitors and a complex set of downstream putative populations. However, whether dietary restriction directly redistributes macrophages or DCs into the BM is still unknown, as most dietary studies have focused on the relocation and function of adipose tissue macrophages, suggesting that they accumulate in areas with increased lipids.63,64 Dietary restriction increases adipogenesis in the BM of mice18,65; thus, it is interesting to consider whether this increased adipogenesis may be responsible for an induction of IL-12-producing macrophages. Furthermore, a potential role of inflammatory macrophages is to metabolize lipids63; thus, increased IL-12-producing macrophages may be associated with increased lipid content within the BM. Additionally, fasting can modulate the gut bacteria flora and enrich for lactobacillus species,66 some strains of which can induce IL-12 production from macrophages.67,68,69 Because changes in gut microbiota can influence BM production of inflammatory cell types,70 we can speculate that these alterations may be responsible for the increased IL-12 production we observe in our model.
Altogether, our work recognizes a critical role for the innate immune system in the improved anti-tumor response induced by fasting. This work will be important in dissecting the immune landscape in patients from clinical trials using fasting and fasting-mimicking diets, given that NK cell infiltration and function have yet to be fully appreciated. Furthermore, we show that cyclic fasting can remodel different tissues and thus represents a novel method of improving tissue-specific NK cell function and enhancing innate immunity for the treatment of solid and metastatic cancers.