생활습관 변화로 후성적 변화가 일어나는가? (한 세대 내 가능)
네, 강한 증거 있습니다.
식습관과 운동은 후성적 표지를 빠르게 바꿀 수 있으며,
이는 한 세대(개인 자신) 내에서 일어납니다.
2021년(후속 인용 많음) randomized controlled trial에서
8주간 식물 중심 식단, 운동, 수면·스트레스 관리 intervention으로
에피제네틱 연령(DNA methylation age)이 평균 3년 이상 역전되었습니다.
https://pmc.ncbi.nlm.nih.gov/articles/PMC8064200/

| 고령화 인구 증가에 따른 사회적·의료적 비용을 고려할 때, 생물학적 노화를 늦추고 건강수명(healthspan)을 연장하기 위한 개입은 중요한 관심사이다. 본 연구에서는 50~72세의 건강한 성인 남성 43명을 대상으로 무작위 대조 임상시험을 수행한 결과를 보고한다. 8주간의 중재 프로그램은 식이, 수면, 운동 및 이완에 대한 지침과 함께 프로바이오틱스 및 식물영양소 보충을 포함하였다. 대조군은 어떠한 중재도 받지 않았다. 전장 유전체 DNA 메틸화 분석은 Illumina Methylation EPIC Array를 사용하여 타액 샘플에서 수행되었으며, DNAmAge는 온라인 Horvath DNAmAge 시계(2013)를 이용해 계산하였다. 식이 및 생활습관 중재는 대조군과 비교했을 때 DNAmAge가 3.23년 감소한 것과 연관되었으며(p=0.018), 중재군 내에서는 프로그램 시작 시점과 종료 시점을 비교했을 때 평균 1.96년의 DNAmAge 감소가 관찰되어 통계적으로 유의미한 경향을 보였다(p=0.066). 혈액 생체지표의 변화 중에서는 평균 혈청 5-메틸테트라하이드로폴레이트가 15% 증가하였고(p=0.004), 평균 중성지방은 25% 감소하였다(p=0.009). 본 연구는 우리가 아는 한, 특정한 식이 및 생활습관 중재가 건강한 성인 남성에서 Horvath DNAmAge(2013) 후성유전학적 노화를 되돌릴 수 있음을 시사한 최초의 무작위 대조 연구이다. 이러한 결과를 확인하기 위해서는 더 대규모이고 장기간의 임상시험이 필요하며, 다른 인간 집단을 대상으로 한 추가 연구도 요구된다. |
운동만으로도
몇 시간~며칠 내에 DNA 메틸화 변화가 관찰됩니다.

| 후성유전적 재프로그래밍은 건강 관련 대사 질환이 장기적인 기능적 건강에 미치는 영향을 예측한다. 이러한 후성유전적 재프로그래밍은 외인성 또는 내인성 자극에 의해 활성화되며, 그 결과 건강한 상태의 변화와 다양한 질병 상태로 이어진다. 이러한 후성유전적 및 환경적 변화에는 식이 구성 요소와 운동이 후성유전적 각인에 미치는 영향, 그리고 배아 발달 과정에서 형성된 후성유전체 패턴을 회복하는 과정 등과 같은 후성유전 네트워킹의 로드맵이 포함되며, 이는 젊은 세포 유형과 건강을 확립하는 데 핵심적이다. 영양과 운동은 적절한 발달과 기능적 생활방식에 영향을 미치는 가장 잘 알려진 환경적 후성유전 요인 중 하나로, 건강 상태에 유익하거나 해로운 영향을 모두 미칠 수 있다. 수정(수태) 시점부터 적용되는 식이 및 운동 전략은 인간 질환의 예방과 치료를 위한 혁신적인 후성유전적 표적이 될 수 있다. 본 논문에서는 건강과 질병에서 치료적 후성유전 전략으로서 식이와 운동의 잠재적 역할을 설명하고, 이 분야의 향후 전망을 제시한다. |
주요 결과 - 식이의 영향 (Key Findings: Diet)
- 메틸 공여 영양소 (folate, B12, choline): One-carbon metabolism 통해 SAM 생성 → DNA/히스톤 메틸화 유지 (Figure 2)
- 기아 노출 (Dutch Famine): 지속적 hypomethylation → 자손/손자 세대 T2DM·비만 위험 증가
- Vegan/식물성 식단: DNMT 억제 (polyphenols) → 유전자 body hypomethylation → T2DM 발병률 감소 (vegan 0.54% vs. non-veg 2.12%)
- 암 관련: EGCG, quercetin 등 bioactives → tumor suppressor hypermethylation 역전

| 간헐적 단식(Intermittent Fasting, IF)은 다양한 종에서 강력한 항노화 효과와 연령 관련 질환의 완화를 달성하는 데 있어 가장 효과적인 중재로 여겨지고 있다. 후성유전적 변형은 여러 환경 요인이 유전자 발현에 미치는 생물학적 효과를 매개하지만, IF가 후성유전체(epigenome)에 미치는 영향에 대해서는 아직 알려진 바가 없다. 본 연구에서 우리는 3개월간의 IF가 소뇌에서 H3K9 삼중 메틸화(H3K9me3)의 변화를 유도하며, 이것이 IF 동안 흔히 관찰되는 강력한 대사 전환 과정에 관여하는 다양한 전사체 수준의 변화를 조율한다는 사실을 처음으로 발견하였다. 또한 IF에 의해 유도된 후성유전체 및 전사체 변화의 일부가 IF 종료 후 재급식(refeeding) 기간 동안에도 최소 3개월 이상 현저하게 유지되어, IF로 유도된 후성유전적 변화에 대한 ‘기억’이 보존됨을 확인하였다. 그러나 주목할 점은 IF를 중단하면 전사체에 대한 H3K9me3의 조절이 소실된다는 사실이다. 종합적으로, 본 연구는 수많은 대사 과정을 조절하는 후성유전–전사체 축에 대한 IF의 새로운 영향을 규명하였다. 본 연구에서 수행된 포괄적인 분석은 IF가 뇌의 대사–후성유전 축(metabolo-epigenetic axis)에 어떠한 영향을 미치는지를 이해하기 위한 분자적 틀을 제시하며, 향후 연구를 위한 중요한 자료가 될 것이다. |
주요 결과 - 운동의 영향 (Key Findings: Exercise)
- 급성 고강도 운동: 근육 promoter hypomethylation (PGC-1α, TFAM, MEF2A, PDK4 등) → 미토콘드리아 기능·포도당 흡수 향상 (Figure 3)
- 지구력 훈련 (6개월): GLUT4 상향, T2DM 위험 유전자 hypomethylation
- 심장 조직: miRNA 상향 (miR-222, miR-29c, miR-17-3p) → 항비대·항섬유화 효과
- 가역성: 운동 후 빠른 복원
주요 결과 - 식이 + 운동 결합 & 세대 간 효과
- MBC2 trial (9개월): IRF2 hypomethylation (항염증), PCCA hypermethylation (염증 감소)
- NAFLD 모델: 저탄수화물 식단 + 운동 → GAB2 promoter hypomethylation → 지방 축적 억제
- 세대 간: 부모 고지방/저활동 → 정자/난자 메틸화 변화 → 자손 비만/T2DM 전달 (Swedish cohort 예)
- 역전 가능성: 건강한 생활습관으로 methylation 패턴 복원
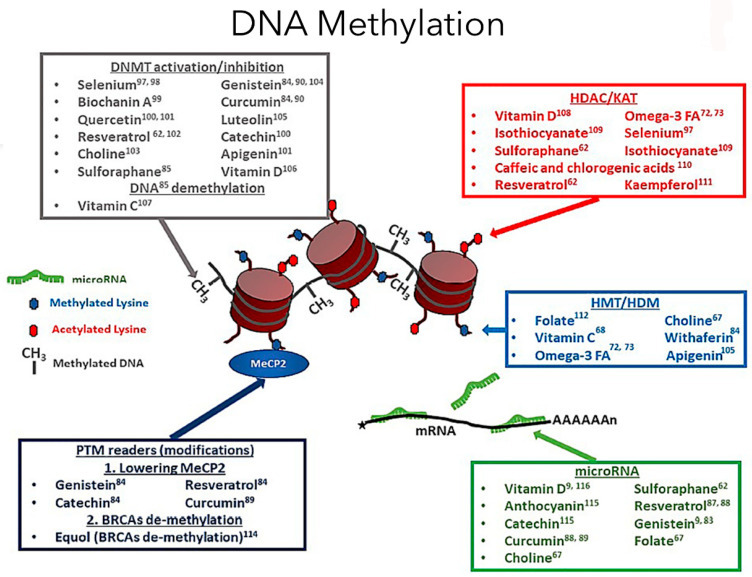
2025년 리뷰에서도
건강한 식단·운동이 에피제네틱 시계를 늦추거나 역전시킬 수 있다고 결론짓습니다.

세대 간 전달(transgenerational inheritance)은 가능한가?
동물(주로 쥐·생쥐) 연구에서는 가능합니다.
어미의 고지방 식단이나 영양 부족이
후성적 표지를 통해 자손·손자 세대까지 비만·당뇨 위험을 증가시킵니다.
일부 연구에서 정상 식단으로 3세대 유지하면
이런 phenotypes(비만 등)과 후성적 변화가 사라진다고 보고되었습니다 (예: Masuyama et al., 2015~2016 참조).
https://pmc.ncbi.nlm.nih.gov/articles/PMC5357455/
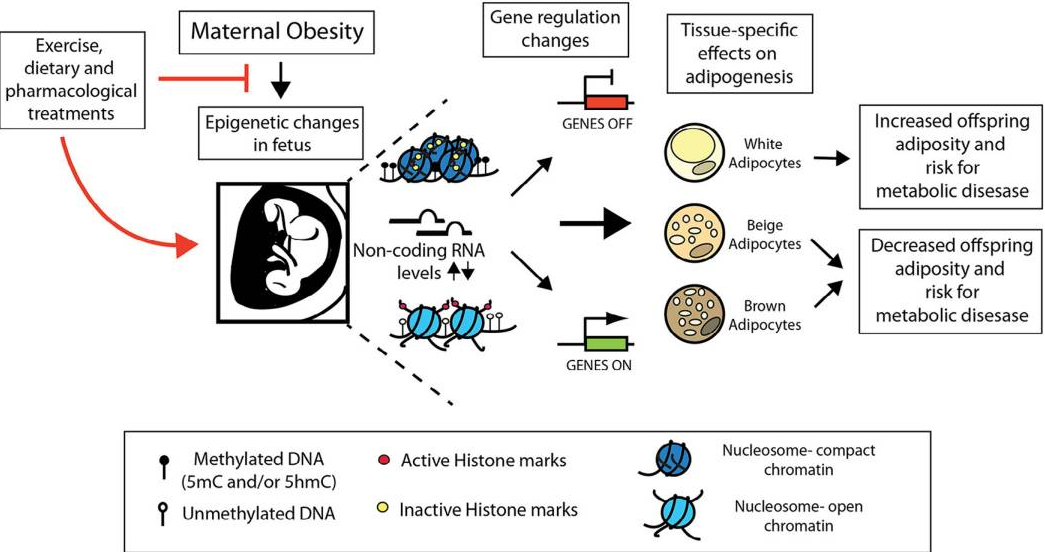

| 모성 비만, 식단, 운동이 지방세포(adipocytes)의 후성유전(epigenetic) 조절에 미치는 영향, 그리고 이를 통해 자손에게 비만 위험이 전달되는 메커니즘을 다룹니다. 이 논문은 실험 연구가 아닌 리뷰로, 동물 모델(주로 쥐와 생쥐) 연구를 종합합니다. 초록(Abstract) 요약모성 비만과 고지방 식단(HFD)이 자손의 지방세포 대사와 후성유전 변화를 통해 비만과 대사증후군을 유발할 수 있으며, 이는 세대 간 전달될 수 있습니다. DNA 메틸화, 히스톤 수정, 비코딩 RNA(miRNA, lncRNA) 등의 후성 메커니즘이 핵심이며, 운동이나 영양 보충(예: 폴리페놀)이 이를 역전시킬 수 있습니다. 서론(Introduction)
|
다른 연구에서는
영양 부족의 ‘기억’이 손자 세대까지 지속되다
증손(great-grandchildren) 세대 전에 사라집니다.
인간에서는 어떠한가? (가장 중요한 부분)
인간에서
**true transgenerational epigenetic inheritance
(환경 요인으로 유발된 후성적 표지가 생식세포를 통해 여러 세대 전달)**에 대한 증거는
매우 약하거나 부족합니다.
2024년 리뷰:
동물에서는 증거 강하지만,
인간에서는 여전히 결여되어 있습니다.
https://pmc.ncbi.nlm.nih.gov/articles/PMC10795545/

2024년 critical perspective:
인간 TEI는 150년 넘게 논쟁 중이지만, 증거가 약하고 재현되지 않으며,
많은 경우 문화적·사회적 전달로 설명 가능합니다.

Dutch Hunger Winter나 Överkalix 연구처럼
조부모 세대 기아가 손자 세대에 영향을 준다는 보고도 있지만,
이는 엄격한 transgenerational이 아니라 intergenerational(직접 노출 영향)으로 해석되거나
재현 실패 사례가 많습니다
Aging (Albany NY)
. 2021 Apr 12;13(7):9419–9432. doi: 10.18632/aging.202913
Potential reversal of epigenetic age using a diet and lifestyle intervention: a pilot randomized clinical trial
Kara N Fitzgerald 1,✉, Romilly Hodges 2, Douglas Hanes 3, Emily Stack 4, David Cheishvili 5, Moshe Szyf 6, Janine Henkel 7, Melissa W Twedt 7, Despina Giannopoulou 7, Josette Herdell 7, Sally Logan 7, Ryan Bradley 7,8
- Author information
- Article notes
- Copyright and License information
PMCID: PMC8064200 PMID: 33844651
This article has been corrected. See Aging (Albany NY). 2022 Jul 27;14(14):5959.
This article has been corrected. See Aging (Albany NY). 2024 Mar 15;16(5):4943.
Abstract
Manipulations to slow biological aging and extend healthspan are of interest given the societal and healthcare costs of our aging population. Herein we report on a randomized controlled clinical trial conducted among 43 healthy adult males between the ages of 50-72. The 8-week treatment program included diet, sleep, exercise and relaxation guidance, and supplemental probiotics and phytonutrients. The control group received no intervention. Genome-wide DNA methylation analysis was conducted on saliva samples using the Illumina Methylation Epic Array and DNAmAge was calculated using the online Horvath DNAmAge clock (2013). The diet and lifestyle treatment was associated with a 3.23 years decrease in DNAmAge compared with controls (p=0.018). DNAmAge of those in the treatment group decreased by an average 1.96 years by the end of the program compared to the same individuals at the beginning with a strong trend towards significance (p=0.066). Changes in blood biomarkers were significant for mean serum 5-methyltetrahydrofolate (+15%, p=0.004) and mean triglycerides (-25%, p=0.009). To our knowledge, this is the first randomized controlled study to suggest that specific diet and lifestyle interventions may reverse Horvath DNAmAge (2013) epigenetic aging in healthy adult males. Larger-scale and longer duration clinical trials are needed to confirm these findings, as well as investigation in other human populations.
고령화 인구 증가로 인한 사회적·의료적 비용을 고려할 때,
생물학적 노화를 늦추고 건강수명을 연장하기 위한 개입은
큰 관심의 대상이다.
본 연구에서는
50~72세의 건강한 성인 남성 43명을 대상으로 무작위 대조 임상시험을 수행하였다.
8주간의 중재 프로그램은
식이, 수면, 운동, 이완에 대한 지침과 함께 프로바이오틱스 및 식물영양소 보충을 포함하였다.
대조군은 어떠한 중재도 받지 않았다.
전장 유전체 DNA 메틸화 분석은 Illumina Methylation Epic Array를 이용하여 타액 샘플에서 수행되었으며, DNAmAge는 Horvath DNAmAge clock(2013) 온라인 계산기를 통해 산출하였다.
식이 및 생활습관 중재군은 대조군에 비해 DNAmAge가 3.23년 감소하였으며, 이는 통계적으로 유의하였다(p = 0.018). 중재군 내에서는 프로그램 시작 시점과 비교했을 때 종료 시점에서 DNAmAge가 평균 1.96년 감소하였고, 통계적 유의성에는 근접한 강한 경향을 보였다(p = 0.066).
혈액 바이오마커 변화 중에서는 평균 혈청 5-메틸테트라하이드로폴레이트가 15% 증가(p = 0.004)하였고, 평균 중성지방은 25% 감소(p = 0.009)하여 유의한 변화를 나타냈다.
본 연구는 건강한 성인 남성에서 특정 식이 및 생활습관 중재가 Horvath DNAmAge(2013)로 측정한 후성유전학적 노화를 역전시킬 수 있음을 시사한 최초의 무작위 대조 연구로 알려져 있다. 다만 이러한 결과를 확증하기 위해서는 더 큰 규모와 장기간의 임상시험이 필요하며, 다른 인구 집단에 대한 추가 연구 또한 요구된다.
Keywords: DNA methylation, epigenetic, aging, lifestyle, biological clock
INTRODUCTION
Advanced age is the largest risk factor for impaired mental and physical function and many non-communicable diseases including cancer, neurodegeneration, type 2 diabetes, and cardiovascular disease [1, 2]. The growing health-related economic and social challenges of our rapidly aging population are well recognized and affect individuals, their families, health systems and economies. Considering economics alone, delaying aging by 2.2 years (with associated extension of healthspan) could save $7 trillion over fifty years [3]. This broad approach was identified to be a much better investment than disease-specific spending. Thus, if interventions can be identified that extend healthspan even modestly, benefits for public health and healthcare economics will be substantial.
DNA methylation is the addition of a methyl group to cytosine residues at selective areas on a chromosome (e.g. CpG islands, shelf/shore, exons, open sea). Methylation constitutes the best-studied, and likely most resilient of many mechanisms controlling gene expression [4]. Unique among epigenetic markers, DNA methylation can readily and cheaply be mapped from tissue samples. Of 20+ million methylation sites on the human genome, there are a few thousand at which methylation levels are tightly correlated with age. Currently, the best biochemical markers of an individual’s age are all based on patterns of methylation [5]. This has led some researchers to propose that aging itself has its basis in epigenetic changes (including methylation changes) over time [6–9].
As of this writing, the best-studied methylation-based clock is the multi-tissue DNAmAge clock [10]. At the time this study design was approved, there were few viable alternatives. Horvath’s DNAmAge clock predicts all-cause mortality and multiple morbidities better than chronological age. Methylation clocks (including DNAmAge) are based on systematic methylation changes with age. DNAmAge clock specifically demonstrates about 60% of CpG sites losing methylation with age and 40% gaining methylation. This is distinct from stochastic changes, “methylation drift”, unpredictable changes which vary among individuals and cell-by-cell within individuals. Systematic methylation changes include hypermethylation in promotor regions of tumor suppressor genes (inhibiting expression) and hypomethylation promoting inflammatory cytokines (promoting expression). Saliva can be considered a good source of high-quality DNA, containing both white blood cells and buccal cells, and is a suitable tissue type to be assessed for the DNAmAge clock [10, 11].
The dietary recommendations employed as part of the treatment protocol for this study were based largely on biochemistry and generalized measures of health, because few dietary associations with the DNAmAge clock have yet been established. A modest, but significant, reduction in DNAmAge in individuals consuming a non-specific lean meat, fish and plant-based diet (as measured by blood carotenoids) has been observed [12]. It is possible that changes of a greater magnitude require a more targeted approach. The dietary intervention used here was also plant-centered, but including a high intake of nutrients that are substrates or cofactors in methylation biosynthetic pathways (e.g. containing folate, betaine), ten-eleven translocation (TET) demethylase cofactors and modulators (e.g. alpha ketoglutarate, vitamin C and vitamin A) [13] and polyphenolic modulators of DNA methyl transferases (DNMT) (e.g. curcumin, epigallocatechin gallate (EGCG), rosmarinic acid, quercetin, luteolin) [14]. It also included limited nutrient-dense animal proteins (e.g. liver, egg). The diet restricted carbohydrates and included mild intermittent fasting, both designed to lower glycemic cycling. The diet was supplemented daily with a fruit and vegetable powder, also rich in polyphenolic modulators of DNMT activity, and a probiotic providing 40 million CFU of Lactobacillus plantarum 299v. L. plantarum has been shown to be a folate producer in the presence of para aminobenzoic acid (PABA) [15]; it also has been demonstrated to alter gene expression [16].
Lifestyle guidance in this study included a minimum of 30 minutes of exercise per day, at least 5 days per week at an intensity of 60-80 percent of maximum perceived exertion. Exercise is well-known to be broadly beneficial for almost every aspect of health and has been shown to extend mean lifespan in animal models. Exploration of the effect of exercise on the methylome has recently begun. For example, regular tai chi practice was associated with slowing of age-related DNA methylation losses in 500 women [17]. In another study of 647 women, a lifelong history of exercise was associated with a similar endpoint [18]. These results were not reported in terms of the Horvath clock, because it had not yet been developed. One systematic review of human studies found that regular, daily physical activity was associated with lower blood levels of homocysteine, which when elevated, suggests an insufficiency of methylation capacity [19]. Excessive exercise may accelerate methylation aging, but this danger has only been observed in elite, competitive athletes [20].
Twice-daily breathing exercises that elicit the Relaxation Response were prescribed for stress reduction. It was recently demonstrated that 60 days of relaxation practice designed to elicit the Relaxation Response, 20 minutes twice per day, could significantly reduce DNAmAge as measured by the Zbieć-Piekarska clock in their group of healthy participants (though not in their ‘patient’ group) [21]. Almost a quarter of the DNAmAge CpG sites (85/353) are located in glucocorticoid response elements, pointing to a likely relationship between stress and accelerated aging. Cumulative lifetime stress has been shown to be associated with accelerated aging of the methylome [22]. Zannas et al. also reported that dexamethasone, a glucocorticoid agonist, can advance the DNAmAge clock and induce associated transcriptional changes. Dexamethasone-regulated genes showed enriched association of aging-related diseases, including coronary artery disease, arteriosclerosis and leukemias. Other findings include that PTSD contributes to accelerated methylation age [23]; and that greater infant distress (lack of caregiver contact) is associated with an underdeveloped, younger epigenetic age [24].
This study aimed to optimize sleep, with a recommendation for at least seven hours nightly. Seven hours is generally considered to be healthy [25], but the limited data on accelerated aging only relates to extremes of sleep deprivation. A (presumably transient) effect of sleep deprivation on genome-wide methylation patterns in blood has been demonstrated [26]. Acceleration of the DNAmAge clock has been associated with insomnia in a sample of 2078 women [27]. Carskadon et al [28] found an association between poor quality / fewer hours of sleep with age acceleration in a small sample of 12 female college students.
This multimodal (“systems”) intervention is reflective of a clinically-used approach that combines individual interventions, each of which carry evidence of favorable influence on the DNA methylome and of which several authors of this study have clinical experience of health benefits. Such interventions likely produce synergistic effects and reduce the possibility of negative effects from one disease-promoting input canceling out the benefits of another health-promoting input. Dietary and lifestyle interventions, as used here, target upstream influences that are generally considered safe, even over the long term.
By design, an important endpoint of this study was to be Horvath’s DNAmAge clock, to see if it could be potentially slowed. This is to say we have tentatively accepted the hypothesis that the methylation pattern from which the DNAmAge clock is computed is a driver of aging (and the chronic diseases of aging), thus we expect that attempting to directly influence the DNA methylome using diet and lifestyle to set back DNAmAge will lead to a healthier, more “youthful” metabolism. To date, three non-controlled studies have demonstrated set back of DNAmAge. One small pilot study has been reported to have set back the DNAmAge clock over the course of 12 months by 1.5 (plus the one-year duration of the study) years in healthy men, using a combination of growth hormone, metformin, DHEA and two dietary supplements [29]. Two additional studies have demonstrated age reduction from diet and/or dietary supplement interventions. A subgroup of Polish women from the NU-AGE cohort suggested a reduction in biological age of 1.47 years after 1 year of a Mediterranean diet plus 400IU vitamin D3, and a 16-week trial using 4000IU of vitamin D3 in overweight or obese African Americans with suboptimal D status demonstrated a 1.85-year reduction in biological age [30, 31]. Herein we report comparable initial results based on diet and lifestyle interventions employed for eight weeks (preceded by a one-week education period).
RESULTSDNA methylation clock
Compared to participants in the control group (n=20), participants in the treatment group scored an average 3.23 years younger at the end of the eight-week program according to the Horvath DNAmAge clock (p=0.018). Those in the treatment group (n=18) scored an average 1.96 years younger, at the end of the program compared to the same individuals at the beginning with a strong trend towards significance (p=0.066 for within group change). Control participants scored an average of 1.27 years older at the end of the study period, though this within-group increase was not statistically significant (p= 0.153). Comparison of DNAmAge change between treatment and control groups is shown in Figure 1 whereas within group changes for the treatment group are shown in Figure 2.
Figure 1.
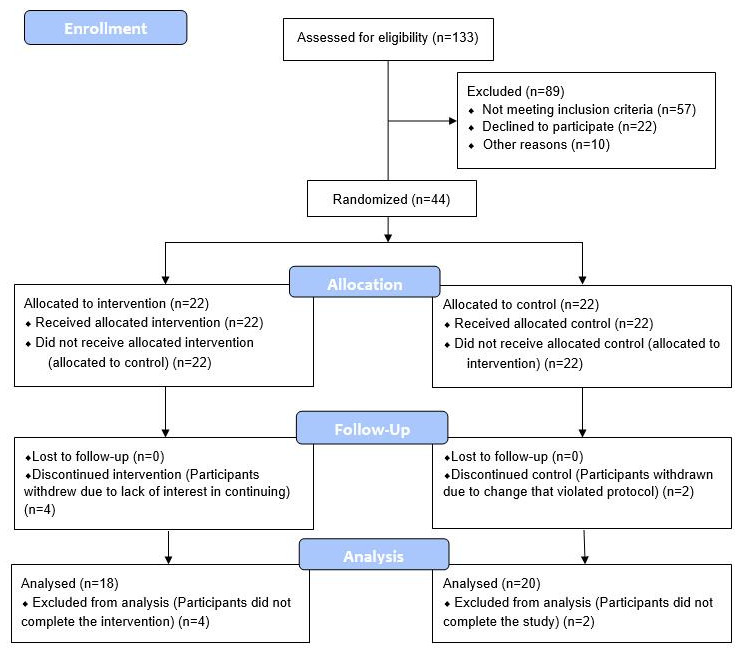
CONSORT 2010 flow diagram.
Figure 2.
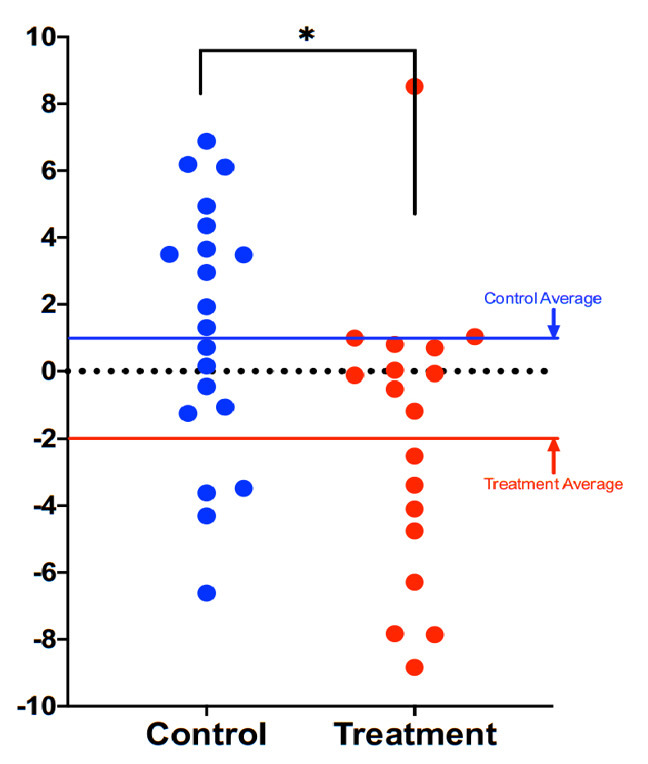
Comparison of DNAmAge change between treatment and control groups. Each dot is a subject, and the vertical axis represents difference in DNAmAge from the beginning to the end of the eight-week term. Participants scored an average 1.96 years younger, controls an average 1.27 years older. The age reduction of the treatment group strongly trended towards significance (p=0.066), while the age increase of the control group itself was not significant (p=0.153). The difference between control and treatment groups was significant at the level p=0.018 (unpaired two-tailed t-test). Long red and blue lines represent group averages (mean).
In both treatment and control groups, there was no net increase or decrease in methylation of the 353 sites that compose the Horvath clock. This finding suggests that the intervention did not lead to an overall increase in methylation of the Horvath clock sites, but rather it prompted a repositioning of clock’s CpG methylation patterns consistent with a younger biological age.
Metabolic measures
In blood markers, the most significant change was a 25% decrease in mean triglycerides from 112 to 89 mg/dL (p=0.009) over the eight-week study period. As expected from a diet rich in folate, mean serum 5-methyltetrahydrofolate (5-MTHF) rose 15% from 78 to 88 nmol/L (p=0.004).
None of the other blood markers measured changed significantly compared to controls (glucose, hemoglobin A1C, total cholesterol, HDL cholesterol, LDL cholesterol, methionine, s-adenosylmethionine (SAM), s-adenosylhomocysteine (SAH) the ratio SAM:SAH and homocysteine), but within-group the participants in the treatment group showed significant decrease in total cholesterol (-22.8 mg/dL, p=0.004) and LDL cholesterol (-16.8 mg/dL, p=0.01).
Emotional measures
Among the PROMIS markers of emotional health, there were no statistically significant changes between treatment and control groups after adjustment for baseline values. There was some trend towards reduced anxiety scores in the treatment group, however, these changes were not statistically significant.
DISCUSSIONSignificance of results
The significance of these findings is multi-factorial, but primarily as the first demonstration of potential reversal of epigenetic age in a randomized, controlled clinical trial, accounting for any normal variability in epigenetic methylation. This is the second report of a diet and lifestyle intervention reducing biologic aging in individuals otherwise known to be healthy. Notably, the shorter timeframe of this study and the scale of potential reduction, while modest in magnitude, may correlate with meaningful socioeconomic benefits, and appears to have the potential to be broadly achievable.
Vitamin D3 at a dose of 4,000 IU/d for 16 weeks has previously been shown to decrease the DNAmAge clock measurement by 1.85 years in overweight/obese African Americans with a serum 25-hydroxyvitamin D [25(OH)d] <50 nmol/L [31]. Subsequently, a one-year regimen of daily injection of growth hormone plus one prescription drug and three nutritional supplements was shown to set back the DNAmAge clock by 1.5 years in 9 middle-aged men (plus the 1-year study duration = 2.5 years) [29]. More recently, a 1-year non controlled pilot trial involving 120 participants aged 65-79 years (including 60 Italians, 60 Poles) drawn from the larger NU-AGE cohort found a non-significant trend towards reversal of the DNAmAge clock after 1 year of a Mediterranean diet plus 400IU of vitamin D3 [30]. However, subgroup analysis did reveal a significant 1.47-year age decreases in female Polish participants (n=36) and in individuals with a baseline higher epigenetic age. It was noted in the study that Poland is a country with a non-Mediterranean baseline diet. In the present study, biological age set-back was achieved in eight weeks, using similarly non-invasive, and otherwise generally beneficial interventions known to have mechanistic plausibility for affecting methylation pathways.
The increase in circulating folate demonstrates that dietary sources and folate-producing probiotics can be an effective method of nutrient repletion. The reduction in serum triglycerides might be expected with a diet that lowered carbohydrate intake and glycemic response, plus exercise [32]. While we expected to see a decrease in homocysteine with an intervention that supplied additional dietary B vitamins and betaine, as well as exercise, the average starting homocysteine value of the treatment group was 10.9 umol/L, already within a range typically identified as “normal” (<15 umol/L).
Effects of non-methyl donor factors on DNA methylation
The seminal work of Waterland and Jirtle in the Agouti mouse model marked a defining point in our understanding that nutrition elements could so affect DNA methylation marks as to silence gene expression and dramatically alter phenotype [33]. The power of nutrition to bring about transformative phenotypic changes has held up over the intervening years, most strongly in animal studies, but also in some limited human trials [33]. All of the aforementioned human trials (NU-AGE, TRIIM, Vitamin D3 study) and the present study were able to effect changes on the DNA methylome without extra-dietary supplementation of known methyl donor nutrients (e.g. folate, vitamin B12, choline, SAMe or betaine), supporting the concept of a far-reaching regulatory network on DNA methylation and representing a departure from previous studies that manipulated DNA methylation more directly with extra-dietary supplemental folate, B12 and other methyl donor nutrients [33–36].
The DNAmAge clock is computed from some sites that increase and others that decrease methylation with age, so a net increase in methylation is not the therapeutic target, compared to modifying methylation at appropriate sties. Since this study targeted a healthy methylation pattern, not limited to increased methylation, the prescribed diet contained TET demethylase-associated nutrients, such as vitamins A and C [13] and specific plant polyphenols such as curcumin and EGCG known to inhibit DNMT activity, in addition to high quantities of food-sourced methyl donor nutrients. As evidenced by younger DNAmAge (ie, “improved” methylation patterns) in the treatment group without overall increase (or decrease) in methylation, it suggests that these compounds collectively assist in elevating methylation and demethylation enzymatic support and thereby potentially regulate where methyl groups are applied and removed. In addition, the combination of polyphenols, rather than the use of singular phytonutrients, has been shown to deliver enhanced favorable effects on epigenetic changes [14, 37].
Rationale for not using supplemental methyl donor nutrients
In designing the present study, extra-dietary supplementation of methyl donor nutrients was specifically avoided because a growing body of epidemiological evidence indicates potential long-term risks, to which the short-term studies were not sensitive. Although overall data are mixed, and certain conditions (e.g. pregnancy, macrocytic anemia, hyperhomocysteinemia, dietary limitations) often require extra-dietary supplementation, several trials have found a positive association between methyl donor supplementation and increased cancer risk: Published long-term follow up on 2,524 participants in the B-PROOF trial which assessed the effect of 2-3 years of daily supplementation with 400 mcg folic acid and 500 mcg vitamin B12 found an increased risk of overall cancer (HR 1.25, 95% CI 1.00-1.53), p=0.05) and colorectal cancer in particular (HR 1.77, 95% CI 1.08-2.90, p=0.02) [38]. A meta-analysis of 2 trials in Norway similarly reported that 800 mcg folic acid plus 400 mg vitamin B12 daily was associated with increased cancer outcomes and all-cause mortality [39]. In contrast, dietary folate intake from food was found to be inversely associated with non-muscle-invasive bladder cancer progression in a study that also found higher recurrence for folic acid intake [40], and baseline dietary folate intake was inversely associated with prostate cancer risk in a trial that subsequently identified an increased risk of prostate cancer in the treatment arm that received 1 mg folic acid per day for 10 years [41]. Also relevant is the demonstration, albeit in a small study, adding dietary supplements of folic acid, vitamin B6 and vitamin B12 to a vitamin D plus calcium intervention increased biological aging (sex-adjusted odds ratio 5.26 vs vitamin D plus calcium alone) during a 1-year intervention [42].
Cautions and future directions
One significant limitation of this pilot trial is limited statistical power due to the relatively small sample size. Confirmation of these results is therefore needed in larger study groups and populations beyond middle-aged men.
It is not yet fully established whether interventions that slow any of the “methylation clocks” necessarily curtail risks of age-related disease. This unknown remains an important area of investigation by epidemiologists working to validate predictors of age-related morbidity and mortality, which would otherwise require very long clinical trials. The use of a multimodal intervention has advantages, as discussed above, however it also means it is not possible to attribute improved outcomes to any one element of the intervention. The combination of interventions used in this study may yet be improved upon and may be more impactful when further personalized. Future iterations of the intervention in continued clinical trials will attempt to optimize the program for efficacy, efficiency, scalability and affordability. An ever-evolving understanding of personalized application of such dietary and lifestyle interventions will likely lead to refinements to this kind of intervention that may further extend indicators of biological age.
Finally, it may be that emerging “omics” approaches continue to evolve our understanding of biological age prediction and reversal beyond DNA methylation alone [43]. Integration of our future understanding of multi-omics data should therefore be considered in the future trials of candidate age-delaying interventions.
MATERIALS AND METHODSStudy design and conduct
The trial design was approved by the Institutional Review Board of the National University of Natural Medicine (IRB number: RB100217) and registered at ClinicalTrials.gov (Identifier: NCT03472820). All trial procedures (visits, consent, randomization, and study visits) were conducted at the Helfgott Research Institute, 2220 SW 1st Ave, Portland, OR 97201.
Participants were voluntarily recruited on a rolling basis from the general community surrounding Portland, OR, using flyers, online and newspaper advertising, and electronic newsletters posted at locations intended to attract more health-conscious individuals, such as gyms. Volunteers completed telephone pre-screening followed by an initial onsite screening visit including confirmation of eligibility, willingness to follow the study protocol, and informed consent. Full inclusion and exclusion criteria are provided as Supplementary Material.
43 eligible adult males without history of recent or chronic disease, between the ages of 50-72, were recruited, consented, enrolled and randomized between March 2018 and August 2019. A CONSORT flow diagram is shown in Figure 3. Baseline characteristics are shown in Table 1. The age range of 50-72 was selected as a time when age-related vulnerabilities typically manifest, and the limitation to only male participants was to avoid the potential confounding factor of pre-, peri-, and post-menopausal sex hormone levels of the same age range in women.
Figure 3.
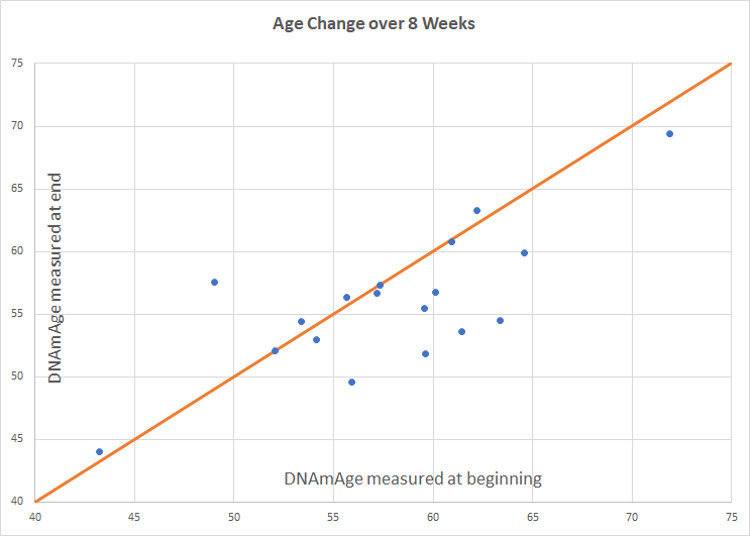
Intervention group age change. Participants scored an average of 1.96 years younger than baseline (p=0.066). Of 18 participants included in the final analysis, 8 scored age reduction, 9 were unchanged, and 1 increased in methylation age.
Table 1. Baseline characteristics.
| Characteristics | Treatment group n = 21 | Control group n = 22 | |||
| Value | % | Value | % | ||
| Age, years (mean±SD) | 58.5 ± 6.12 | 60.3 ± 6.68 | |||
| Race | |||||
| Black or African American | 0 | 0 | 2 | 9.1 | |
| American Indian or Native Alaskan | 0 | 0 | 0 | 0 | |
| Native Hawaiian or Other Pacific Islander | 0 | 0 | 0 | 0 | |
| Asian or Asian American | 3 | 13.6 | 1 | 4.5 | |
| White, Caucasian or European American | 18 | 81.8 | 18 | 81.8 | |
| Caribbean Islander or African National | 0 | 0 | 0 | 0 | |
| More than one race | 0 | 0 | 1 | 4.5 | |
| Unknown | 0 | 0 | 0 | 0 | |
| Education Level | |||||
| Some high school | 0 | 0 | 0 | 0 | |
| High school | 0 | 0.00 | 3 | 0.14 | |
| Some university | 1 | 0.05 | 1 | 0.05 | |
| 2 year university | 2 | 0.09 | 1 | 0.05 | |
| 4 year university | 6 | 0.27 | 4 | 0.18 | |
| Some graduate school | 2 | 0.09 | 4 | 0.18 | |
| Graduate degree | 11 | 0.50 | 9 | 0.41 | |
A 3-week washout period (with written instruction) was initiated for all participants involving discontinuation of any nutrition supplements or herbal products not prescribed by a licensed healthcare provider for a medical condition. Allowable exceptions for dietary supplementation included low dose supplements, such as a common "1-a-day" multivitamin/mineral (i.e. high potency, high dose multivitamin/mineral products were not allowable), and/or other supplements taken for prevention: fish oil (up to 1 gram/day), vitamin D (up to 6000 IU/day), vitamin C (up to 1g/day), vitamin E (up to 400 IU/day). Notably, these supplements were not recommended or prescribed, but participants already taking these products were allowed to continue them during the trial, thus any effects would be captured in their baseline assessments. Participants agreed to avoid/discontinue any recreational drugs/substances, as well as to consume no alcohol, nicotine, marijuana or cannabinoids at least 1 week before scheduled study visits.
Study participants were randomized at the baseline visit according to a randomization sequence (randomization.com). Allocation concealment was accomplished by opening sealed, signed envelopes prepared by research staff not associated with the trial which were only opened at randomization.
Initial instructions, including a recorded instructional webinar and electronic technology webinar were provided at visit 1. To allow time for participant education to occur, participants in the treatment group were instructed to begin the 8-week intervention protocol (including dietary, supplement, and lifestyle changes) starting one week after the baseline visit. Saliva samples were collected at each of the three study visits (baseline, week 5 and week 9).
An overview of the intervention is provided in Table 2. Two nutritional supplements (PhytoGanix® and UltraFlora® Intensive Care, Metagenics Inc., 25 Enterprise Aliso Viejo, CA 92656 USA) were distributed at visits 1 and 2. Unused doses were collected, counted and recorded at visits 2 and 3. Dosing adherence was verified by the retrospective review of returned doses, direct queries about each component of the intervention during study visits, and by frequent communication with trial participants.
Table 2. Summary of dietary and lifestyle interventions*.
| Intervention category | Details |
| Dietary Prescription | Guidance per week: 3 servings of liver •(1 serving = 3 oz) •Preferably organic 5-10 eggs •Ideally free-range, organic, omega-3 enriched Guidance per day: 2 cups of dark leafy greens •Measured raw, chopped, and packed •Including kale, Swiss chard, collards, spinach, dandelion, mustard greens •Does not include salad greens such as romaine, iceberg, Spring mix 2 cups cruciferous vegetables •Measured raw, chopped, and packed •Includes broccoli, cabbage, cauliflower, Brussels sprouts, bok choy, arugula, kale, mustard greens, watercress, rutabaga, kohlrabi, radish, Swiss chard, turnip 3 additional cups colorful vegetables of your choosing (excluding white potatoes, sweetcorn) 1-2 medium beet 4 tbsp (1/4 cup) pumpkin seeds (or pumpkin seed butter) 4 tbsp (1/4 cup) sunflower seeds (or sunflower seed butter) 1+ serving methylation adaptogens, choose from: •1/2 cup berries (wild preferred) •1/2 tsp rosemary •1/2 tsp turmeric •2 medium cloves garlic •2 cups green tea (brewed 10 minutes) •3 cups oolong tea (brewed 10 minutes) 6 oz animal protein •Grass-fed, pastured, organic and hormone/antibiotic-free 2 servings of low glycemic fruit General guidance: Organic preferred over conventional Stay hydrated Don’t eat between 7pm and 7am Include “healthy” oils •Balance types of fat •E.g. coconut, olive, flaxseed and pumpkin seed oil Avoid added sugar/candy, dairy, grains, legumes/beans Minimize plastic food containers |
| Supplement Prescription | PhytoGanix®, a combination of organic vegetables, fruits, seeds, herbs, plant enzymes, prebiotics and probiotics at a dose of 2 servings daily, divided UltraFlora® Intensive Care, containing Lactobacillus plantarum 299v at a dose of 2 capsules daily, divided |
| Exercise Prescription | Minimum of 30 minutes of exercise per day for at least 5 days per week, at an intensity of 60-80% of maximum perceived exertion |
| Sleep Prescription | Average a minimum of 7 hours of sleep per night |
| Stress Management Prescription A | Breathing exercise Steps to Elicit the Relaxation Response developed by Herbert Benson MD, twice daily |
AStress Management Recommendations were updated from the original Study Protocol as listed on ClinicalTrials.gov. All updates were IRB approved. *Patent pending.
Adherence to the program was supported by regular coaching sessions, delivered weekly during the first four weeks, and then at least every other week thereafter. Coaching sessions followed a pre-defined script that covered adherence to intervention guidelines and any changes to medications. A HIPAA-compliant electronic technology coaching tool was offered (MBody360, 640 Broadway 5A, New York, NY 10012) which contained reference instructions, meal planning ideas, optional recipes, and a shopping list. Participants could also use this tool to communicate with their assigned coach in between scheduled coaching sessions. Email, web platform and/or phone communication were other options for participants unable or unwilling to use MBody360.
Determination of epigenetic age
Sample Handling: Saliva samples were stored at -70° C and remained frozen throughout the duration of the trial. Frozen samples were batch shipped overnight on dry ice to Yale University Center for Genome Analysis at the conclusion of clinical operations. Prior to shipment, sample IDs were assigned to plates such that each plate included a representative collection of samples from both allocation groups (treatment and control) and a distribution of samples from each trial visit, i.e. plates were not homogenous by group or by visit type, in an effort to equally distribute any random variability resulting from measurement across plates.
DNA extraction
Oragene Saliva tubes were submitted and DNA extracted using the Perkin Elmer Chemagic 360 Instrument (kit# CMG-1081) following the manufacturer’s recommended protocol. An RNASE A digestion was added using 80μL of 4mg/μg Amercian Bioanalytical RNASE A (Part#AB12023-00100) after the 50 degree Celsius Oragene Saliva Incubation, before loading samples onto instrument.
Genomic DNA and RNA
The quality of the RNA/DNA was evaluated by: A260/A280 and A260/A230 ratios (as supplied by the NanoDrop 1000 Spectrophotometer), both of which should be > 1.8. The gel electrophoresis pattern was consistent with non-degraded samples.
Methods
Sample DNA was normalized to the recommended starting concentration, 1μg, for the Zymo EZ-96 DNA Methylation Kit (Cat No D5004). The samples underwent an overnight bisulfate conversion and were purified using the Zymo Methylation protocol. Samples proceeded directly to the Illumina Infinium HD assay (Illumina Methylation Epic Array Cat. No. WG-317-1001) to overnight whole genome amplification at 37 degrees Celsius. The following day samples were fragmented for 1hour at 37 degrees, precipitated at 4 degrees for 30 minutes, and pelleted at 4 degrees for 20 minutes at 3000xg. Samples were dried in a hood at room temperature for 1 hour and re-suspended in the recommended volume of RA1 following Illumina’s Infinium HD assay at 48 degrees Celsius for 1 hour. Samples were then denatured at 95 degrees Celsius for 20 minutes. Samples had a 10-minute cool down and then directly hybridized to Illumina Methylation Epic Array Cat. No. WG-317-1001. Sample placement in each chip was randomized. Hybridization was for 18 hours at 48 degrees Celsius using a stabilized hybridization oven. The following day arrays were washed and stained automatically using the Tecan Freedom Evo technology. Arrays were then be dipped in the UV protectant (Illumina’s XC3) for ten minutes and any excess removed. Arrays were dried for 1 hour in a vacuum desiccator. Arrays were scanned using Illumina’s iScan array scanner and raw files generated. Raw data files were imported into Illumina’s GenomeStudio software and a project created as well as QC parameters checked to ensure project went as expected. Scanned output files were analyzed and call rates calculated using Illumina’s GenomeStudio. The quality of the data was evaluated by both sample dependent and sample independent controls. Specifically, efficiency of target removal, non-specific binding, and appearance of cross-contamination was examined. All raw Data and the GenomeStudio project were uploaded to the password protected Keck Microarray Database.
Data analysis
DNAmAge was calculated using the online Horvath clock available at https://dnamage.genetics.ucla.edu/. Analysis of epigenetic age was performed, blinded, on the final 18 participants in the treatment group and 20 participants in the control group. P values were computed as an unpaired 2-tailed t-test between the experimental group and control group, using the individual score differences (after treatment minus before) as a random variable.
Data sharing
The data that support the findings will be available in Gene Expression Omnibus at https://www.ncbi.nlm.nih.gov/geo/, submission number GSE 149747, from 04-14-23 following an embargo from the date of publication to allow for commercialization of research findings.
Supplementary Material
Supplementary Materials
aging-13-202913-s001.pdf (209.6KB, pdf)
ACKNOWLEDGMENTS
We would like to acknowledge and thank the following for their assistance in this research study: The team at the National University of Natural Medicine Helfgott Research Institute for conducting the clinical trial. Kari Thostensen and MBody360 for their collaboration and the use of their coaching platform. Marquelle Brown, Janine Henkel, Alison Parkerson and Brianne Pugh for their contributions to diet-compatible recipe development. Yale University for measuring CpG site methylation according to the Illumina Infinium array. Dr. Moshe Szyf’s team at the Department of Pharmacology and Therapeutics at McGill University for their input on data analysis. Josh Mitteldorf for his extensive assistance with data analysis. Steve Horvath PhD ScD, for input on the calculation of epigenetic age.
Footnotes
AUTHOR CONTRIBUTIONS: KF, RB and MS designed the study. RH and KF designed the study diet. RB and ES conducted the clinical trial at Helfgott Research Institute. JH, SL, JH, DG and MT coached the participants through the intervention. DH performed the data analysis and reporting with MS, DC and DH in an advisory capacity. KF, RB and RH developed the manuscript. All authors approved the manuscript.
CONFLICTS OF INTEREST: KF and RH declare that they use the intervention described here in clinical practice, are named in a related patent application, and receive earnings from educational products associated with its use. Notably, KF and RH were not involved in the day-to-day conduct of the trial, collection of samples, or data analysis.
FUNDING: This study was generously supported through an unrestricted grant from Metagenics, Inc.
REFERENCES
- 1.Jin K, Simpkins JW, Ji X, Leis M, Stambler I. The critical need to promote research of aging and aging-related diseases to improve health and longevity of the elderly population. Aging Dis. 2014; 6:1–5. 10.14336/AD.2014.1210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sen P, Shah PP, Nativio R, Berger SL. Epigenetic mechanisms of longevity and aging. Cell. 2016; 166:822–39. 10.1016/j.cell.2016.07.050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kirkland JL, De Rooij SE, Goldman DP. Proof-of-concept clinical trials of interventions that target fundamental aging processes how can interventions be translated from the laboratory into clinical practice the economic returns to delayed aging: promise and pitfalls. Innov Aging. 2017. (Suppl 1); 1:1082. 10.1093/geroni/igx004.3967 [DOI] [Google Scholar]
- 4.Li E, Zhang Y. DNA methylation in mammals. Cold Spring Harb Perspect Biol. 2014; 6:a019133. 10.1101/cshperspect.a019133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Horvath S, Raj K. DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nat Rev Genet. 2018; 19:371–84. 10.1038/s41576-018-0004-3 [DOI] [PubMed] [Google Scholar]


