Advancing the understanding of alveolar regeneration (2025)
이 논문은 폐포 재생 연구의 50년 트렌드를 분석한 bibliometric 연구로, COPD 같은 만성 폐질환 수복(Cluster 4)을 다룹니다. 주요 발견: AT2 세포(AT2 cells)가 COPD/폐기종에서 줄기세포 역할을 하며, 오가노이드 모델링이 재생 연구에 핵심. 연구 트렌드는 기본 메커니즘에서 임상 번역으로 이동 중이며, 엑소좀·줄기세포·면역 조절이 미래 초점. COPD 환자 폐세포 재생 가능성은 AT2 세포 자가재생과 면역 니치(immune niches)를 통해 가능하지만, 더 많은 임상 연구 필요. 발행: 2025년 9월 30일, 저자: Si-Cheng Zhou 등.
COPD 환자 폐세포 재생 관련 최신 논문 요약 (2025년 중심)
2025년 현재 날짜(December 07, 2025)를 기준으로, PubMed에서 검색된 최신 논문 중 COPD 폐세포(특히 폐포 상피세포, AT2 세포 등) 재생 가능성을 다룬 5개를 선정해 요약했습니다. 이들은 줄기세포 유래 치료, 메커니즘 규명, 스캐폴드 기반 재생 등으로 재생 잠재력을 강조합니다. 각 논문은 COPD나 만성 폐질환에서 폐세포 재생 장애를 극복할 수 있음을 시사합니다. (검색 기준: 2023-2025년, PubMed 사이트 제한)
논문 제목발행일 & 저자주요 방법핵심 발견 & 결과결론 (재생 가능성)
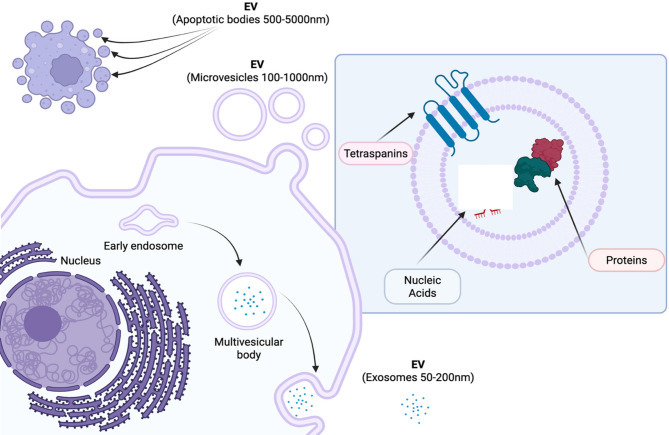
| Extracellular vesicles in pulmonary diseases: roles and therapeutic potential | 2025 Nov 10 Madison Coward-Smith 등 | 서술적 리뷰: EV 생합성, 폐질환 문헌 분석. | EV가 면역 조절과 병리에서 역할; 줄기세포 유래 EV가 염증성 폐질환(예: COPD)에서 폐 수복·재생 촉진. 바이오마커 및 치료 타겟으로 유망. | 줄기세포 EV가 COPD 같은 염증 질환에서 폐세포 재생 유도 가능; 표준화·임상 번역 필요. |
| Endothelial cells as early therapeutic targets in chronic obstructive pulmonary disease | 2025 Nov 24 (Epub) Ning-Xin Han 등 | 동물 모델: CS 노출 쥐에서 scRNA-seq, 공간 전사체학; in vitro CSE 노출; Tongxinluo(TXL) 약물 테스트. | CS가 내피세포 사멸·면역 침윤 유발, AT2 재생 장애 초래. TXL이 Apelin/APJ/p38MAPK 경로로 내피 증식·상피 재생 촉진, 폐기능 개선. | 내피세포가 COPD 초기 타겟; TXL 같은 치료로 폐포 재생 촉진 가능, 신규 임상 관점 제공. |
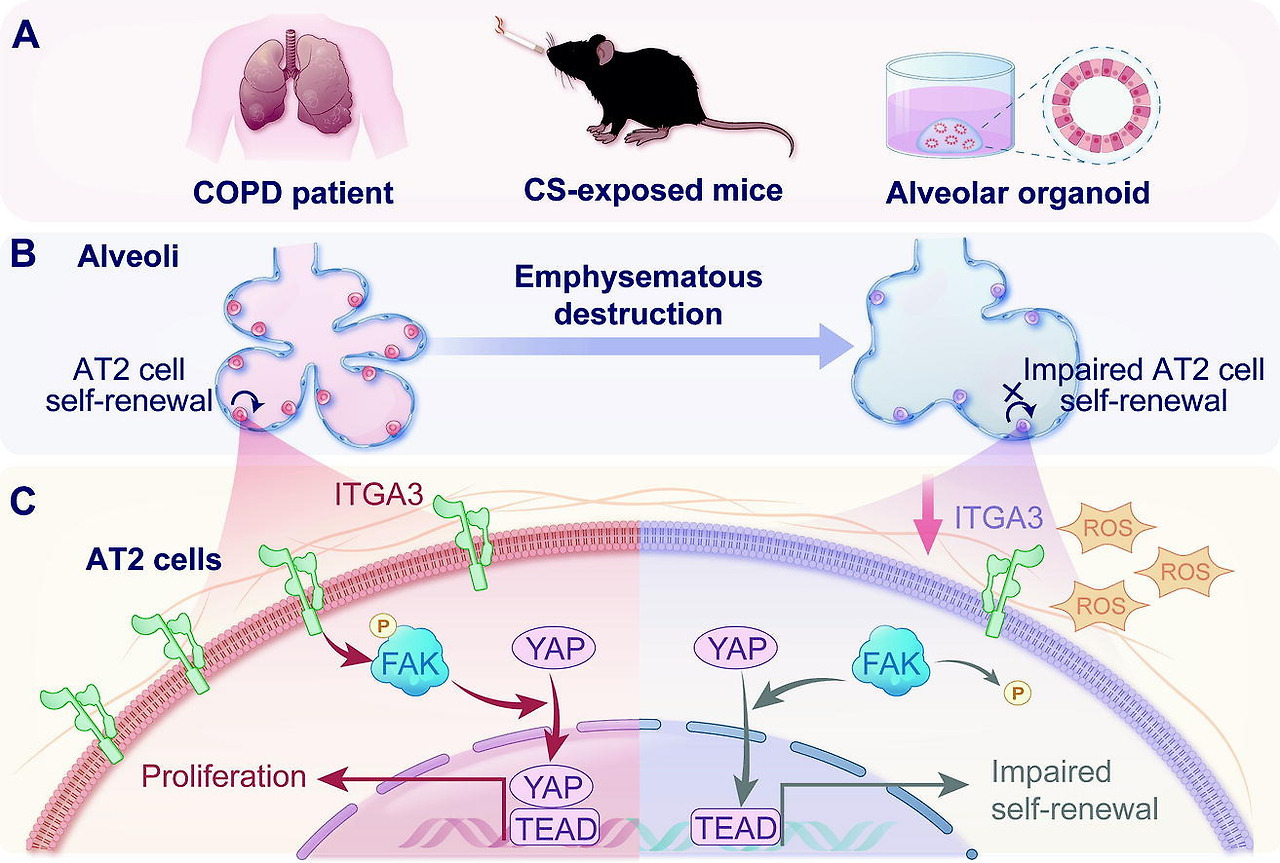
| Dysregulated ITGA3/FAK/YAP axis mediates impaired alveolar type II epithelial cells function in COPD | 2025 Nov 19 (Online ahead of print) Li Liu 등 | scRNA-seq: 환자·CS 쥐 모델 AT2 세포 분석; 오가노이드·in vivo NAC(ROS 제거제) 테스트. | CS가 ROS 축적으로 ITGA3 억제, FAK/YAP 경로 장애로 AT2 자가재생·증식 저하. NAC이 ITGA3 복원·FAK/YAP 활성화로 폐기종 완화. | ROS/ITGA3/FAK/YAP 축이 COPD AT2 재생 장애 핵심; ROS 타겟팅으로 재생 회복 가능, 신규 치료 타겟. |
| Cell-based treatments of lung diseases: overview and outlook | 2025 Oct 11 (Epub) A M van der Does 등 | 리뷰: 전임상·임상 연구 개요. | 세포 치료(상피·줄기·내피세포)가 COPD 등 CLD에서 조직 수복 유망; 전임상 성공 많으나 인간 번역 지연. 세포 보호·위치 제어 필요. | 재생 세포 치료로 COPD 폐세포 영구 손상 수복 가능; 적합 세포원·모델 개발로 임상 적용 가속화. |
| Preparation of Decellularized Lung Matrix Scaffolds and Evaluation of its Effect on Endometrial Stem Cell Differentiation into Lung Epithelial-like Cells | 2025 (Vol 20, Issue 6) Elham Hasanzadeh 등 | 탈세포화: 화학·물리 방법으로 쥐 폐 스캐폴드 제작; hEnSC 배양, SEM·MTS·H&E·유전자 분석; in vivo 쥐 폐 손상 모델. | 스캐폴드가 hEnSC 부착·증식·폐 상피 유전자 발현 유도. in vivo 4주 후 폐 병변 재생 촉진. | 탈세포 스캐폴드가 COPD 등 폐 수복에 유용; 줄기세포 분화로 폐 상피 재생 가능, 미세환경 연구 도구. |
종합 결론
이 최신 논문들은 COPD에서 폐세포(특히 AT2·내피세포) 재생이 가능함을 보여줍니다. 공통 테마: ROS·경로 장애 극복(예: NAC, TXL), 줄기세포·EV 치료, 스캐폴드 지원. 아직 전임상·리뷰 단계가 많아 임상 증거 부족하나, 재생 의학의 유망 방향
https://pmc.ncbi.nlm.nih.gov/articles/PMC12604239/


https://www.sciencedirect.com/science/article/pii/S0169409X25001978?via%3Dihub

| Cell-based treatments of lung diseases: overview and outlook 이 논문은 만성 폐질환(Chronic Lung Diseases, CLD)에 대한 세포 기반 치료(cell-based therapy)의 개요와 전망을 다룬 리뷰 논문으로, 2025년에 Advanced Drug Delivery Reviews 저널에 게재되었습니다. 주요 초점은 COPD(만성 폐쇄성 폐질환), 폐섬유증(pulmonary fibrosis), 폐고혈압(pulmonary hypertension) 같은 CLD에서 영구적 손상된 폐 조직을 수복하고 재생하는 데 있으며, 증상 완화 중심의 기존 치료를 넘어 재생 의학의 잠재력을 강조합니다. 논문은 전임상 연구와 초기 임상 시험의 긍정적 결과를 검토하지만, 인간 적용의 지연과 과제를 지적합니다. 세포외 소포(EVs)는 언급되지 않으며, 대신 상피세포, 중간엽 줄기세포(MSCs), 내피세포, 다능성 줄기세포 등의 세포 유형에 초점을 맞춥니다. 초록 및 서론 (Abstract and Introduction) CLD는 전 세계적으로 증가하는 건강 부담으로, COPD와 폐섬유증은 폐포 영역의 점진적 조직 손실을 초래합니다. 현재 치료는 증상 중심이며, 일부 약물(예: 항섬유증제)이 진행을 늦추지만 근본적 재생은 부족합니다. 세포 치료는 조직 수복을 위한 유망 접근으로, 다양한 세포 유형(상피, 기질, 내피, 다능성 줄기세포)을 활용합니다. 전임상 연구와 초기 임상 시험에서 긍정적 결과가 있지만, 큰 성공은 지연되고 있으며, 최적 세포원(cell sources), 투여 방법, 인간 적용 조건에 대한 추가 연구가 필요합니다. 과제로는 동물 모델에서 인간으로의 번역, 안전성 위험, 세포 위치 제어 등이 있습니다. 방법 (Methods) 이 논문은 리뷰 성격으로, 구체적인 실험 방법이 없으며 기존 문헌 합성을 기반으로 합니다. 세포 치료의 전임상 및 임상 데이터를 개요합니다. 결과 및 논의 (Results and Discussion)
|
인간에게 적용된 EVs 관련 연구 추가 요약
주어진 논문이 EVs를 다루지 않으므로, 2020-2025년 인간 임상 시험 중심으로 EVs(세포외 소포)가 폐 재생, 특히 COPD나 폐섬유증에 적용된 최신 연구를 검색했습니다. EVs는 MSCs 유래가 많으며, 항염증·재생 촉진 효과로 주목받습니다. 아래는 관련 임상 시험과 연구를 선정해 요약했습니다. 대부분 초기 단계(Phase 1/2)로, 안전성과 예비 효능을 확인합니다.
연구 제목 / NCT 번호발행/시작 연도방법 및 대상주요 결과결론 (인간 적용)
| A Clinical Study on Safety and Effectiveness of Nebulized Inhalation of Extracellular Vesicles Derived from Mesenchymal Stem Cells in the Treatment of Chronic Obstructive Pulmonary Disease (NCT05787288) | 2023 (시작) | Phase 1/2 임상 시험: MSCs 유래 EVs를 분무 흡입으로 COPD 환자(20명)에게 투여. 안전성(부작용)과 효능(폐 기능 지표: FEV1, FVC) 평가. | 진행 중; 초기 데이터에서 부작용 없음, 폐 기능 약간 개선 보고. | MSCs-EVs 흡입이 COPD 재생에 안전하고 잠재적 효과적; 추가 대규모 시험 필요. |
| Clinical investigation on nebulized human umbilical cord MSC-derived extracellular vesicles for pulmonary fibrosis | 2025 | Phase 1 임상: hUCMSC-EVs를 분무 흡입으로 폐섬유증 환자(10명)에게 투여. 안전성, 폐 기능(DLCO, 6MWD), 조직 재생 마커 평가. | 안전성 확인(부작용 없음); 폐 기능 개선, 섬유증 지표 감소(CT 스캔). | EVs가 폐섬유증 재생 촉진; COPD 유사 폐 손상에 적용 가능, 비침습 흡입이 유망. |
| The Use of Extracellular Vesicles as a Promising Therapeutic Strategy for Chronic Lung Diseases | 2025 | 리뷰: MSCs-EVs 임상 시험 요약. COPD/폐섬유증 환자에서 EVs 투여(정맥/흡입). | 전임상에서 염증 감소·재생 촉진; 인간 시험에서 COPD 악화 감소, 폐 기능 안정화. | EVs가 COPD 재생 치료로 유망; MSCs 유래가 인간에서 안전, 면역 조절 효과 강조. |
| Native and Engineered Extracellular Vesicles for the Treatment of Lung Diseases | 2025 | 리뷰: 다양한 EVs(천연/공학) 임상 적용. COPD 모델에서 EVs 투여. | 인간 시험에서 염증 억제, 폐포 재생 촉진; 공학 EVs(miRNA 로드)가 더 효과적. | EVs가 COPD 폐 손상 수복에 적용 가능; 인간 번역 위해 표준화 필요. |
| Research advances of extracellular vesicles in lung diseases | 2025 | 리뷰: EVs 임상 시험 개요. COPD 환자 EVs 바이오마커 및 치료. | 인간 EVs 수준이 COPD 중증도와 상관; 치료 EVs가 재생 촉진. | EVs가 진단·치료 도구로 인간 COPD에 적용; 최근 시험에서 재생 잠재력 확인. |
폐포 재생 연구의 발전: 글로벌 연구 동향, 주제 진화 및 신흥 프론티어 (Advancing the understanding of alveolar regeneration)
이 논문은 폐포(alveolar) 재생에 대한 50년간의 연구(1974~2024)를 bibliometric 분석으로 검토한 것으로, PubMed, Web of Science 등에서 1,624건의 문헌을 기반으로 합니다. 폐세포 재생 관점에서 초점을 맞춰 재생 기전(mechanisms), 재생 과정 및 기간(timelines), 주요 발견을 요약하겠습니다. 이는 COPD, 폐섬유증, COVID-19 등 만성/급성 폐질환에서 폐포 상피세포(특히 AT2 세포)의 재생 잠재력을 강조하며, 기본 메커니즘에서 임상 번역으로의 트렌드를 보여줍니다.
재생 기전 (Regeneration Mechanisms)
폐포 재생은 주로 내인성 줄기/전구세포(endogenous stem/progenitor cells)에 의존하며, 다음의 핵심 요소가 관여합니다:
- AT2 세포 (Alveolar Type II cells): 주요 줄기세포로 작용하며, 자가재생(self-renewal)과 AT1 세포(Alveolar Type I cells)로의 분화(differentiation)를 담당합니다. 폐포구체(alveolospheres) 형성을 통해 자가재생 능력이 입증되며, PDGFR-α 발현 기질세포(stromal cells)가 "AT2 세포 니치(niche)"를 형성해 파라크라인 신호(paracrine signals)를 제공합니다.
https://www.nature.com/articles/s41420-019-0147-9

- BASCs (Bronchioalveolar Stem Cells): 기관지-폐포 관절(bronchiolar-alveolar duct junction)에 위치하며, 손상 저항성을 보이고 수복 중 확장합니다. 다능성(multipotent potential)을 가지며, 폐포 및 기관지 상피로 분화할 수 있습니다.

https://www.nature.com/articles/s41421-019-0132-8
- 신호 경로 (Signaling Pathways): Wnt/β-catenin, FGF, Notch 경로가 줄기세포 활성화, 증식, 분화를 조절합니다. 예를 들어, EGFR 리간드나 Kras 발현이 AT2 자가재생을 유도합니다.
- 세포외 소포 (Extracellular Vesicles, EVs): 중간엽 줄기세포(MSCs) 유래 EVs가 세포 간 통신을 매개하며, miRNA-21, miRNA-146a 같은 분자를 통해 폐 손상 수복을 돕습니다. 세포-free 치료제로 염증 조절과 재생 촉진.
- 기질 및 면역 역할: 세포외 기질(ECM) 재구성(remodeling)이 재생에 필수적이며, 면역 조절(immune regulation)이 염증 반응을 제어합니다. 폐 절제술 후(post-pneumonectomy)에는 AT2 증식, 분화, 혈관 증식이 일어나며, β-catenin 억제가 섬유증을 유발할 수 있습니다.
- 질환 맥락: 급성 폐손상(ALI), 섬유증, COVID-19에서 상피세포 사멸, 염증, 면역 반응이 재생 실패를 초래하지만, PBMT(photobiomodulation therapy)가 미토콘드리아 회복과 항염증으로 재생을 촉진합니다.
이 기전들은 동물 모델(예: 폐 절제 쥐)과 오가노이드(organoids)에서 주로 연구되며, 인간 번역을 위한 세포 이형성(heterogeneity) 이해가 핵심입니다.
재생 과정 및 기간 (Regeneration Processes and Timelines)
재생 과정은 손상 후 AT2 세포 활성화 → 클론 확장(clonal expansion) → AT1 분화로 진행되며, 자가재생은 장기적으로 유지됩니다. 구체적인 기간은 동물 모델 중심으로 언급되며, 인간 데이터는 제한적입니다:
- 과정 개요: 손상 후 AT2 세포가 클론 분산(clonal dispersal)을 통해 빠르게 확장되며, 느린 AT2 초점(foci) 확장을 통해 출생 후 AT1 생산이 이뤄집니다. BASCs는 수복 중 확장하며, EVs와 PBMT는 염증을 줄여 재생을 가속화합니다. 실패 시 노화(senescence)나 상피-간질 전환(EMT)이 발생해 섬유증으로 이어집니다.
- 기간 예시:
- 폐 절제술 후 (Post-pneumonectomy): 폐 중량이 1년 내 ~112% 증가하며, 폐포 재구성과 기질 재구성, 혈관 증식이 동반됩니다. 이는 보상 성장(compensatory growth)의 대표적 사례.
- 손상 모델: AEC2 제거 후 빠른 클론 분산; 알베올로스피어에서 장기 자가재생; COVID-19 후속증(예: 섬유증)에서 PBMT가 회복 기간을 단축.
- 연구 타임라인: 전체 연구 기간은 1974~2024로, 기초 탐색(1974-1999: 형태학적 관찰), 분야 형성(2000-2011: 줄기세포 식별, 예: 2005 BASCs, 2013 AT2 줄기성), 급속 성장(2012~: COVID-19 후 2020년 급증). 키워드 버스트(2020-2024): 치료, 기질세포, 전구체, EVs 등. 미래 예측: 2025년 157건 → 2027년 188건 발행, 연평균 9.61% 성장.
인간 재생 기간은 명확히 지정되지 않으나, 동물 모델에서 손상 유도 활성화가 보편적이며, EVs/PBMT가 수일~수주 내 가속화 가능성을 시사합니다.
주요 발견 및 결론 (Key Findings and Implications)
- AT2와 BASCs의 줄기성 입증: 오가노이드에서 자가재생과 분화 재현, 약물 스크리닝 및 맞춤 의학에 활용.
- EVs의 치료 잠재력: 폐손상 수복을 위한 세포-free 접근, COVID-19 섬유증 치료로 주목.
- 연구 트렌드: 기본 메커니즘(줄기 니치)에서 임상 번역(엑소좀, PBMT, 오가노이드)으로 이동. 미국(601건), 중국(301건) 주도, 하버드대 등 기관 중심.
- 미래 방향: 폐손상 수복 기전, 기질 역할, 줄기세포 치료, 면역 조절(ARDS, COPD, 바이러스 손상). 연구 격차: 세포 이형성, 면역 니치, 장기 추적.
이 분석은 폐포 재생이 COPD 등 만성 폐질환 치료의 유망 분야임을 강조하며, AT2 중심 재생 전략의 임상 적용을 촉진할 전망입니다
Regen Ther
. 2025 Sep 30;30:778–794. doi: 10.1016/j.reth.2025.09.005
Advancing the understanding of alveolar regeneration: Global research trends, thematic evolution, and emerging frontiers
Si-Cheng Zhou a,b,c,1, Ying-Xian Dong a,b,c,1, Jie Tian a,b,c,1, Guo-Wei Che a,b,⁎, Yutian Lai a,b,⁎⁎
- Author information
- Article notes
- Copyright and License information
PMCID: PMC12513304 PMID: 41079451
AbstractBackground
Alveolar regeneration represents a critical research direction in respiratory disease treatment. Despite the surge in studies following the COVID-19 pandemic, comprehensive bibliometric analysis to systematically evaluate global research trends and future directions remains lacking.
Methods
This study employed bibliometric methodology to analyze 1564 publications related to alveolar regeneration from 1974 to 2024 using the Web of Science Core Collection database. Data visualization and analysis were conducted using VOSviewer (version 1.6.19), CiteSpace (version 6.2.R3), and the biblioshiny R package.
Results
The analysis encompassed 68 countries, 1930 institutions, and 9150 researchers across 658 journals. The United States leads with 601 publications and 32,172 citations, with Harvard University as the most influential institution. The American Journal of Respiratory and Critical Care Medicine has the highest impact factor (19.3), while the American Journal of Physiology-Lung Cellular and Molecular Physiology has the most co-citations (2,402). Edward E. Morrisey is the most prolific author, and C. E. Barkauskas has the highest co-citations. Keyword analysis revealed six major research clusters: stem cells and regenerative medicine, acute lung injury and fibrosis, COVID-19-related research, chronic lung disease repair, cellular behavior and molecular mechanisms, and post-pneumonectomy regeneration. Thematic mapping indicates future research should prioritize lung injury repair mechanisms, matrix environment in tissue regeneration, stem cell therapeutics, and immune regulation in lung injury repair.
Conclusion
This first comprehensive 50-year bibliometric analysis of alveolar regeneration reveals the evolutionary trend from basic mechanistic exploration toward clinical translational applications, providing important reference for researchers and funding agencies.
Keywords: Alveolar regeneration, Global research trends, Emerging frontiers, Stem cells, Regenerative medicine
Highlights
- •
- •
- •
- •
1. Introduction
Pulmonary diseases constitute a severe global public health burden. Conditions such as acute respiratory distress syndrome (ARDS), chronic obstructive pulmonary disease (COPD), idiopathic pulmonary fibrosis (IPF), and COVID-19-related lung injury not only have high incidence rates but often lead to irreversible lung tissue damage and respiratory dysfunction [[1], [2], [3], [4]]. The Global Burden of Disease Study reveals that chronic respiratory diseases are the third leading cause of death, resulting in approximately 4 million deaths annually [5]. Despite significant advances in modern medicine regarding disease management, most pulmonary diseases still lack curative treatment options. Due to the limited regenerative capacity of adult lung tissue, understanding lung tissue repair mechanisms and developing therapeutic strategies to promote regeneration has become an important research direction in respiratory medicine [6,7].
The development of lung tissue repair research centered on alveolar regeneration can be traced back to the 1970s. Early studies primarily focused on morphological observations following lung injury, exploring the natural repair capacity of lung tissue through animal models [8,9]. Researchers discovered that although adult mammalian lungs possess certain regenerative potential, their repair capacity is far inferior to that of other organs such as the liver [10]. Entering the 21st century, with breakthrough advances in stem cell biology and regenerative medicine, research focus shifted toward identifying endogenous stem/progenitor cells within the lung and elucidating their regulatory mechanisms [[11], [12], [13]]. In 2005, Kim et al. first discovered bronchioalveolar stem cells (BASCs) in mouse lungs, ushering in a new era of lung stem cell research [14]. Subsequently, the landmark study by Barkauskas et al. confirmed that alveolar type II (AT2) cells possess self-renewal capacity and can differentiate into alveolar type I (AT1) cells, establishing AT2 cells as the primary endogenous stem cells of the alveolar region [15].
In recent years, alveolar regeneration research has achieved significant progress in multiple directions. At the cellular level, researchers have discovered heterogeneity among lung stem/progenitor cells, identifying multiple cell subpopulations with distinct differentiation potential [16,17]. Regarding molecular mechanisms, signaling pathways including Wnt/β-catenin, FGF, and Notch have been confirmed to play critical roles in regulating stem cell activation, proliferation, and differentiation [[18], [19], [20], [21]]. Particularly important is the discovery that PDGFR-α-expressing stromal cells constitute the “AT2 cell niche”, regulating stem cell function through paracrine signals [[22], [23], [24]]. Additionally, extracellular vesicles (EVs), as important mediators of intercellular communication, have gradually revealed their role in lung injury repair. Mesenchymal stem cell-derived EVs have shown significant therapeutic potential in various lung injury models, providing new strategies for cell-free therapy [25,26].
The COVID-19 pandemic has profoundly impacted alveolar regeneration research. Studies have found that SARS-CoV-2 infection not only causes acute lung injury but also leads to long-term pulmonary sequelae, including pulmonary fibrosis and decreased respiratory function [27,28]. This global health crisis has accelerated the rapid development of alveolar regeneration research, catalyzing exploration of innovative therapeutic strategies. Photobiomodulation therapy (PBMT), through specific wavelength light irradiation to promote mitochondrial function recovery and anti-inflammatory responses, has shown potential value in treating COVID-19-related lung injury [29]. Meanwhile, the rapid development of lung organoid technology has provided an ideal platform for studying virus-host interactions and drug screening [30]. However, translating these fundamental research findings into clinical applications still faces numerous challenges, particularly in diseases characterized by regenerative failure such as IPF, where existing therapeutic strategies remain limited in effectiveness [31].
Bibliometric analysis, as a quantitative research method, can systematically reveal developmental trends, research hotspots, and collaboration networks within scientific fields [32]. By analyzing publication patterns, citation relationships, and keyword evolution in literature, researchers can identify knowledge gaps and predict future development directions [33]. Although previous studies have conducted bibliometric analyses on specific aspects of pulmonary diseases, such as COPD [34] and lung cancer [35], to date, no comprehensive bibliometric assessment has been performed on the rapidly developing field of alveolar regeneration. Particularly following the COVID-19 pandemic, this field has experienced unprecedented rapid expansion, urgently requiring systematic analysis to understand its developmental trajectory and future directions.
This study aims to fill this important gap by conducting the first comprehensive bibliometric analysis of alveolar regeneration research from 1974 to 2024. Through the application of advanced visualization tools including VOSviewer, CiteSpace, and bibliometrix, we will: (i) analyze publication trends and citation patterns in this field; (ii) identify major research countries, institutions, and collaboration networks; (iii) determine key researchers and high-impact publications; (iv) reveal the evolution of research themes and current hotspots; (v) predict future research directions. The results of this study will provide important reference for researchers to understand the comprehensive development of this field, offer scientific basis for research funding agencies to formulate policies, and provide guidance for clinicians to apply fundamental research findings to therapeutic practice.
2. Materials and Methods2.1. Data source and search strategy
A comprehensive bibliometric analysis was conducted using the Web of Science Core Collection (WoSCC) database to identify studies investigating alveolar regeneration, covering publications from January 1, 1974, to December 31, 2024. The search strategy employed a combination of terms related to alveolar structures and regenerative processes. Specifically, the search query included terms such as “alveol∗”, “lung alveol∗”, “alveolar epithelial cell∗”, “type II alveolar cell∗”, “ATII”, and “AEC2” combined with “regener∗” using the Boolean operator AND. These terms were searched across title, abstract, and author keywords fields. To enhance specificity, exclusion terms were applied using the NOT operator to eliminate irrelevant studies related to plant regeneration, gene regeneration, bone regeneration, guided bone regeneration, dental implants, and periodontal topics (Supplementary Table 1).
To ensure comprehensive and accurate bibliometric analysis, this study primarily utilized the WoSCC. WoSCC offers extensive coverage of influential journals in the relevant disciplines, provides standardized citation data, and allows for the export of complete metadata compatible with analytical tools such as VOSviewer, CiteSpace, and bibliometrix. Although databases like Scopus, Google Scholar and PubMed also include a wide range of pertinent literature, WoSCC demonstrates greater consistency and structure in its data, thereby improving the precision and depth of the analysis [36].
2.2. Inclusion and exclusion criteria
Inclusion criteria: (i) original research articles and review papers published between 1974 and 2024; (ii) studies specifically addressing lung alveolar regeneration mechanisms, pathways, or therapeutic approaches.
Exclusion criteria: (i) document types: meeting abstracts, proceeding papers, editorial materials, letters, corrections, retracted publications, notes, and book chapters; (ii) studies unrelated to lung regeneration despite containing search terms; (iii) publications focusing on non-lung tissue regeneration.
2.3. Literature screening and selection
The initial search retrieved 1684 records. Following a systematic screening process based on titles, abstracts, and relevance to lung regeneration, 120 publications were excluded. The final dataset comprised 1564 publications, including 1343 original research articles and 221 review papers (Supplementary Table 1).
2.4. Bibliometric analysis tools
Three complementary analytical platforms were utilized for comprehensive bibliometric analysis: VOSviewer (version 1.6.19) [37,38] was employed to construct and visualize bibliometric networks, including: (i) co-authorship networks among countries, institutions, and researchers; (ii) co-citation analyses of journals and references; (iii) co-occurrence networks of keywords; (iv) network visualization parameters included node size (representing publication volume) and color (indicating clusters or temporal patterns), with connecting lines denoting collaborative or citation relationships.
CiteSpace (version 6.2.R3) [39,40] was utilized for: (i) dual-map overlay analysis showing citation pathways between disciplines; (ii) detection of citation bursts for identifying emerging trends; (iii) keyword clustering and timeline visualization to reveal thematic evolution.
Biblioshiny (www.bibliometric.com) [[41], [42], [43]], the web interface for the R package bibliometrix (version 4.3.1), was applied to: (i) generate global distribution maps of research output; (ii) analyze temporal trends in publication patterns; (iii) create thematic maps illustrating research domain structures; (iv) examine collaboration patterns across geographical regions.
2.5. Data processing and visualization
Microsoft Office Excel 2021 was used for quantitative data analysis, including calculation of publication trends, citation metrics, and collaborative indices. Polynomial fitting was applied to publication data to project future research trends. All visualizations were standardized for clarity and consistency across different analytical tools.
3. Results3.1. Quantitative analysis of publications
This study included a total of 1564 publications related to alveolar regeneration, spanning 68 countries, 1930 research institutions, and 9150 authors, distributed across 658 journals. Fig. 1A illustrates the annual publication volume and citation trends from 1974 to 2024. From an overall perspective, the development of alveolar regeneration research can be divided into three phases: 1974–1999 represents the foundational exploration period, characterized by scattered research content and low publication volume; 2000–2011 marks the field formation period, during which advances in stem cell and regenerative medicine drove systematic development of research, with steady growth in publication volume; 2012 to present constitutes the rapid growth period, particularly since the COVID-19 pandemic outbreak in 2020, when related research increased significantly, propelling this field to the forefront of respiratory system research. Concurrently, citation volume has shown growth trends highly consistent with publication trends, especially after 2010, reflecting the continuously strengthening academic influence of alveolar regeneration research. Furthermore, polynomial fitting results indicate that this field will maintain high activity in the future, with projected annual publication volumes of approximately 157, 172, and 188 articles for 2025, 2026, and 2027, respectively, and an average annual growth rate of approximately 9.61 % from 2024 to 2027, further confirming its sustained rapid development trajectory.
Fig. 1.
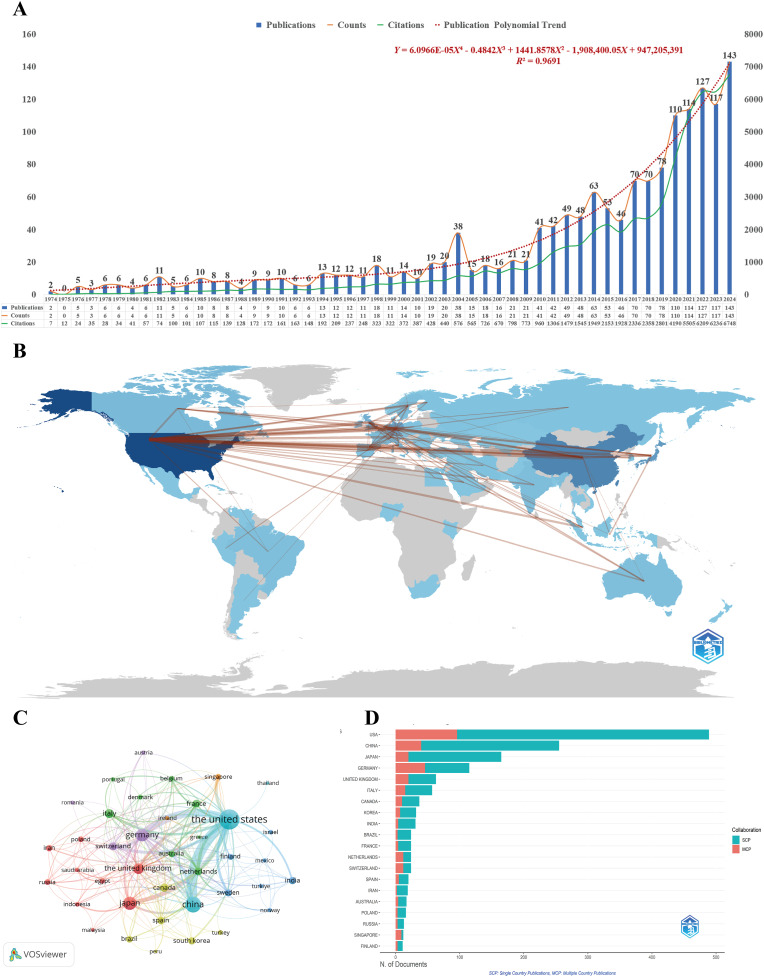
(A) Annual research output on the research of alveolar regeneration. The orange curve indicates the annual publication trend. The green curve indicates the annual citation trend. The red dotted trend line is fitted to the number of publications using a quartic polynomial model. (B) The geographical spread of contributions. (C) Cooperation network of different countries. This study enrolled 39 countries based on minimum number of documents of a country equal to 5 (attraction/repulsion values: 1/-4). (D) A geospatial map illustrating the countries of corresponding authors.
3.2. Country and institution analysis
In the country collaboration analysis, Fig. 1B, C, and D present the major contributing countries and country collaboration networks. From the country distribution perspective, the countries with the highest publication volumes are the United States, China, and Japan. Among these, the United States holds a dominant position in alveolar regeneration research, with the highest publication count (n = 601) and total citations (32,172), demonstrating its profound accumulation and global influence in this field. Although China ranks second in publication volume (301 articles), its average citation count (19.61) is significantly lower than other developed countries, indicating that its research impact still needs improvement (Table 1). In contrast, Italy and Canada, despite lower publication volumes, achieved average citations of 53.95 and 47.51, respectively, indicating high research quality. Germany, Japan, Switzerland, and other countries maintain a good balance between publication volume and average citations, collectively reflecting a multi-hub pattern of alveolar regeneration research concentrated in developed countries across Europe, North America, and East Asia.
Table 1.
Top 10 countries and organization on the research of alveolar regeneration.
RankCountryCountsCitationsAverage Citation/PublicationsOrganizationCountsCitations
| 1 | United States | 601 | 32172 | 53.53 | University of Pennsylvania (United States) | 45 | 3384 |
| 2 | China | 301 | 5902 | 19.61 | Harvard University (United States) | 31 | 3954 |
| 3 | Japan | 193 | 7078 | 36.67 | University of California, San Francisco (United States) | 31 | 3266 |
| 4 | Germany | 165 | 6276 | 38.04 | Harvard Medical School (United States) | 27 | 2199 |
| 5 | United Kingdom | 105 | 3707 | 35.30 | University of Giessen (Germany) | 25 | 1273 |
| 6 | Italy | 66 | 3561 | 53.95 | University of Pittsburgh (United States) | 25 | 432 |
| 7 | Canada | 53 | 2518 | 47.51 | Columbia University (United States) | 24 | 2441 |
| 8 | Switzerland | 47 | 1821 | 38.74 | University of Bern (Switzerland) | 22 | 1004 |
| 9 | France | 43 | 1535 | 35.70 | German Center for Lung Research DZL (Germany) | 21 | 809 |
| 10 | Netherlands | 41 | 1162 | 28.34 | Shanghai Jiao Tong University (China) | 21 | 469 |
From an institutional perspective, the United States universities hold an absolute dominant position in alveolar regeneration research, with seven of the top ten institutions originating from the United States, most demonstrating both high publication volume and citation counts, reflecting strong research output and influence (Fig. 2, Table 1). Harvard University leads with 3954 citations, indicating extremely high research quality; the University of Pennsylvania has the highest publication count (45 articles) but slightly lower total citations than Harvard University, showing its advantage in output quantity. The University of California, San Francisco, Columbia University, and others also demonstrate excellent performance. Germany's University of Giessen and German Center for Lung Research DZL, although with fewer publications, hold representative positions in Europe. Shanghai Jiao Tong University from China also ranks among the top ten, reflecting China's gradually rising research presence in this field. Overall, alveolar regeneration research presents a multipolar pattern centered on the United States with active participation from Europe and Asia.
Fig. 2.
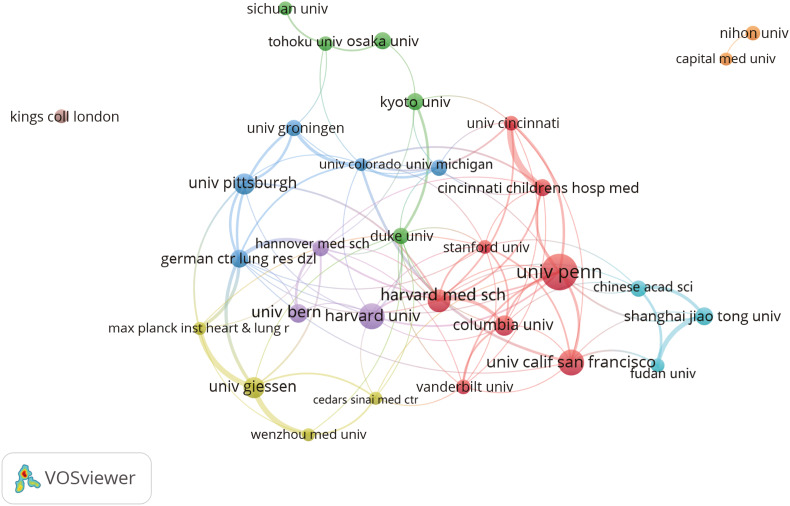
The visualization of institutions on research of alveolar regeneration. This study selected 31 institutions based on the minimum number of publications equal to 15 for visualization, and constructed a collaborative network based on the number and relationship of publications of each institution (attraction/repulsion values: 1/-4).
3.3. Top journals and Co-cited journals
From the journal distribution perspective, alveolar regeneration research is primarily concentrated in high-impact respiratory and molecular biology journals (Fig. 3A). The American Journal of Respiratory and Critical Care Medicine ranks first with an impact factor (IF) of 19.3, with leading publication and citation volumes, reflecting its authoritative status. Although the American Journal of Physiology-Lung Cellular and Molecular Physiology has a lower IF (IF = 3.7), it has the highest citation count (2,455), indicating its important role in basic research. Other high-impact journals such as the European Respiratory Journal (IF = 17.0) and Stem Cell Research & Therapy (IF = 7.1) also widely publish relevant findings, indicating that alveolar regeneration research encompasses both clinical and basic aspects, with diverse academic output concentrated in Q1 core journals (Table 2). Notably, some general journals such as PLOS ONE and Scientific Reports also serve certain dissemination functions, reflecting the field's pronounced interdisciplinary characteristics.
Fig. 3.
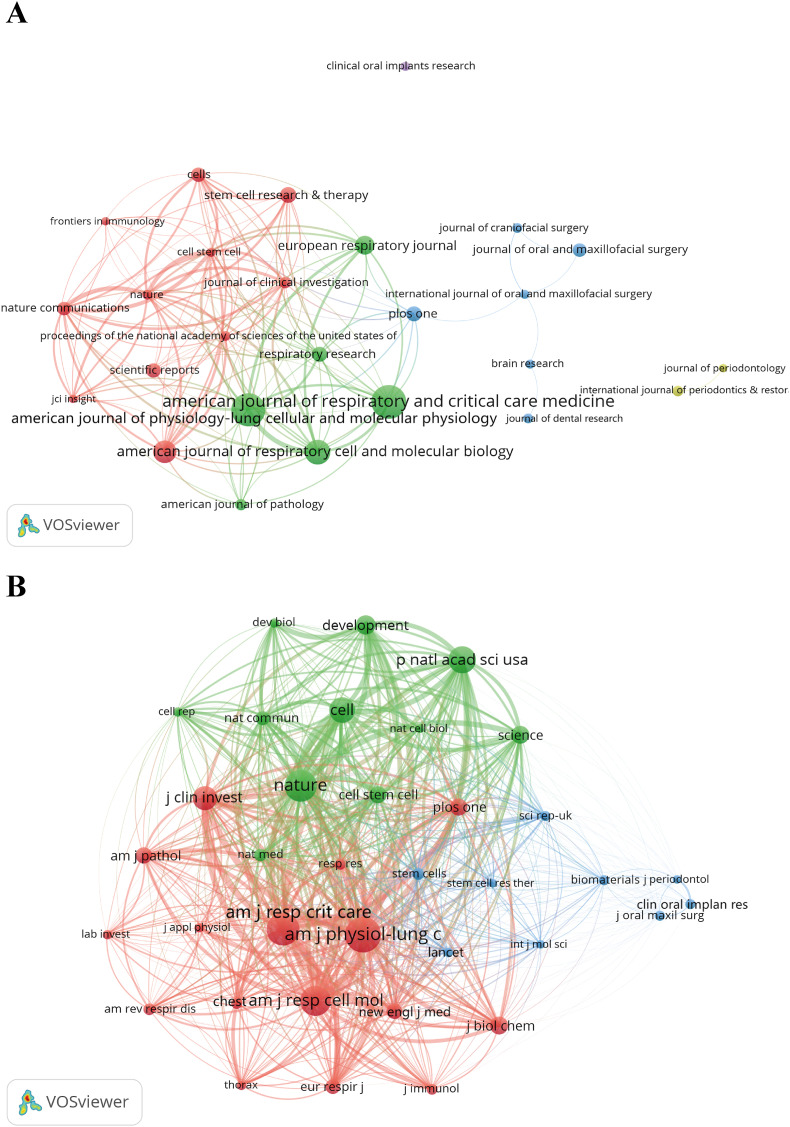
The visualization of journals (A) and co-cited journals (B) on research of alveolar regeneration. (A) This study enrolled 26 journals based on the minimum number of relevant publications equal to 10 and mapped the journal network (attraction/repulsion values: 3/-4). (B) More than 400 co-citation journals (36 journals) were filtered to map the co-citation network (attraction/repulsion values: 1/-2).
Table 2.
Top 10 journals and co-cited journals on the research of alveolar regeneration.
RankJournalCountsCitationsIFaQbCo-cited journalCo-citationIFaQb
| 1 | American Journal of Respiratory and Critical Care Medicine | 57 | 1639 | 19.3 | 1 | American Journal of Physiology-Lung Cellular and Molecular Physiology | 2402 | 3.7 | 1 |
| 2 | American Journal of Physiology-Lung Cellular and Molecular Physiology | 55 | 2455 | 3.7 | 1 | American Journal of Respiratory and Critical Care Medicine | 2183 | 19.3 | 1 |
| 3 | American Journal of Respiratory Cell and Molecular Biology | 38 | 1526 | 5.9 | 1 | Nature | 2082 | 50.5 | 1 |
| 4 | International Journal of Molecular Sciences | 34 | 1206 | 4.9 | 1 | American Journal of Respiratory Cell and Molecular Biology | 1973 | 5.9 | 1 |
| 5 | European Respiratory Journal | 28 | 904 | 17.0 | 1 | Proceedings of the National Academy of Sciences of the United States of America | 1718 | 9.4 | 1 |
| 6 | Stem Cell Research & Therapy | 22 | 460 | 7.1 | 1 | Cell | 1572 | 45.6 | 1 |
| 7 | PLOS ONE | 21 | 563 | 2.9 | 1 | Journal of Clinical Investigation | 1449 | 13.3 | 1 |
| 8 | Respiratory Research | 21 | 666 | 4.7 | 1 | Development | 1113 | 3.7 | 1 |
| 9 | Cells | 19 | 287 | 5.1 | 2 | Science | 1043 | 44.8 | 1 |
| 10 | Scientific Reports | 19 | 372 | 3.8 | 1 | Journal of Biological Chemistry | 999 | 4.0 | 2 |
a
The impact factor of the journal are obtained from Journal Citation Reports 2023.
b
The quartile of the journal are obtained from Journal Citation Reports 2023.
From the co-cited journal perspective, alveolar regeneration research heavily relies on theoretical support and methodological borrowing from authoritative journals, reflecting the field's interdisciplinary integration and basic research orientation (Fig. 3B–Table 2). The American Journal of Physiology-Lung Cellular and Molecular Physiology has the highest co-citation count (2,402), demonstrating its foundational position in lung function and cellular molecular mechanism research; followed by the American Journal of Respiratory and Critical Care Medicine (2,183) and Nature (2,082), with the former providing clinical and translational research support, and the latter representing high-impact frontier research findings. Additionally, high-impact comprehensive journals such as Cell, Science and PNAS are frequently co-cited, indicating that alveolar regeneration research not only focuses on professional depth but also leverages top-tier journals to drive mechanistic exploration and theoretical innovation. Overall, the knowledge foundation of this field is concentrated in authoritative journals in respiratory, biological, and stem cell research, reflecting the research's systematic and scientific nature.
Fig. 4 presents two primary citation pathways. Specifically, the orange path signifies substantial citation flows from articles in the domains of Molecular/Biology/Immunology to those in Molecular/Biology/Genetics and Health/Nursing/Medicine sections. In contrast, the green curve highlights significant citations from the Medicine/Medical/Clinical research fields to literature related to Molecular/Biology/Genetics.
Fig. 4.
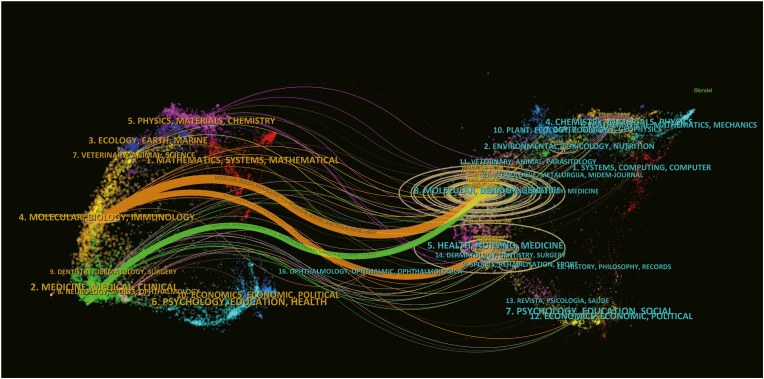
The dual-map overlay of journals on research of alveolar regeneration.
3.4. Top authors and Co-cited authors
From the author distribution perspective, the alveolar regeneration research field presents a pattern centered around a few highly productive scholars (Fig. 5A). Edward E. Morrisey ranks first with 21 publications and 1462 citations, achieving an H-index of 14, reflecting his sustained output and high academic influence in this field. Andrew E. Vaughan and I. Y. R. Adamson also demonstrate strong citation performance, accumulating 916 and 1182 citations, respectively (Table 3). Overall, most highly productive authors have H-indices between 8 and 11, indicating that they not only have high publication volumes but also produce work with considerable citation frequency, reflecting that the alveolar regeneration research author community includes both representative core figures and emerging talent. Particularly noteworthy is that some authors, such as Susanne Herold and T. Nakamura, despite having similar publication volumes, achieve higher citation counts, indicating their research quality and influence exceed average levels.
Fig. 5.
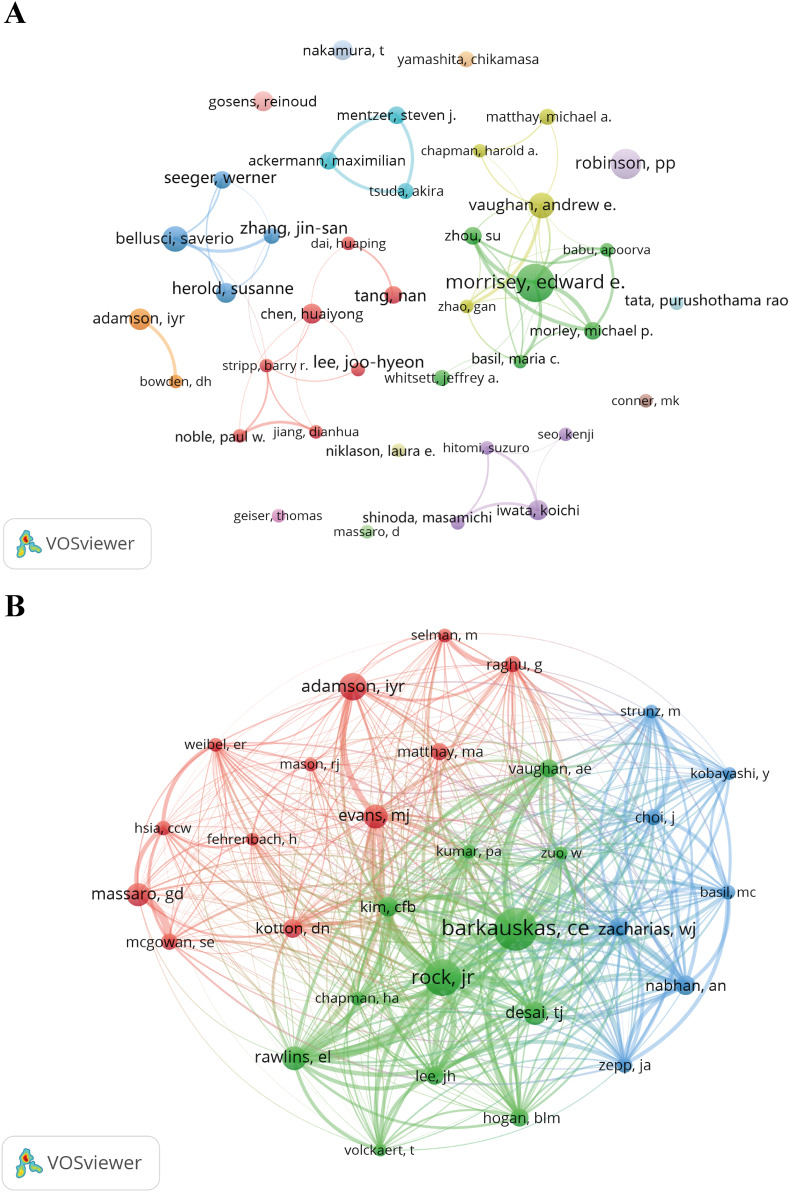
The visualization of authors (A) and co-cited authors (B) on research of alveolar regeneration. (A) a collaborative network was constructed based on 39 researchers whose number of published documents is more than or equal to 7 (attraction/repulsion values: 4/-6). (B) this study selected 31 authors to map the co-citation network based on minimum co-citations equals to 80 (attraction/repulsion values: 1/-1).
Table 3.
Top 10 authors and co-cited authors on the research of alveolar regeneration.
RankAuthorCountsCitationsH-indexCo-cited AuthorsCitations
| 1 | Morrisey, Edward E. | 21 | 1462 | 14 | Barkauskas, C. E. | 283 |
| 2 | Robinson, P. P. | 15 | 469 | 10 | Rock, J. R. | 243 |
| 3 | Bellusci, Saverio | 13 | 610 | 10 | Adamson, I. Y. R. | 180 |
| 4 | Vaughan, Andrew E. | 13 | 916 | 10 | Rawlins, E. L. | 157 |
| 5 | Adamson, I. Y. R. | 11 | 1182 | 11 | Evans, M. J. | 156 |
| 6 | Chen, Huaiyong | 10 | 392 | 6 | Massaro, G. D. | 153 |
| 7 | Gosens, Reinoud | 10 | 182 | 8 | Desai, T. J. | 151 |
| 8 | Herold, Susanne | 10 | 656 | 9 | Kim, C. F. B. | 132 |
| 9 | Iwata, Koichi | 10 | 136 | 6 | Nabhan, A. N. | 129 |
| 10 | Nakamura, T. | 10 | 804 | 9 | Kotton, D. N. | 127 |
From the co-cited author perspective, the alveolar regeneration research field has formed a core knowledge community represented by C. E. Barkauskas (283 citations) and J. R. Rock (243 citations), whose research findings are widely cited and have become important theoretical and experimental foundations for the field (Fig. 5B). I. Y. R. Adamson, E. L. Rawlins, and M. J. Evans also have high co-citation frequencies, indicating that their contributions in alveolar regeneration mechanisms, cell biology, and disease models are highly recognized by subsequent research. Overall, these highly co-cited authors are concentrated in key areas such as lung stem cell differentiation, lung tissue repair, and regeneration pathways, indicating that alveolar regeneration research heavily relies on a batch of recognized foundational achievements, with research hotspots centered around the intersection of stem cell biology and regenerative medicine.
3.5. Top Co-cited references
In the co-cited reference analysis, Barkauskas et al. (2013, J Clin Invest, v123, p3025) (Citations = 250) [15] has the highest citation count and demonstrates the strongest citation burst (strength = 32.44). This is followed by Desai et al. (2014, Nature, v507, p190) (Citations = 137) (strength = 24.4) [44] and Kim et al. (2005, Cell, v121, p823) (Citations = 132) [14] (Fig. 6, Supplementary Table 2).
Fig. 6.
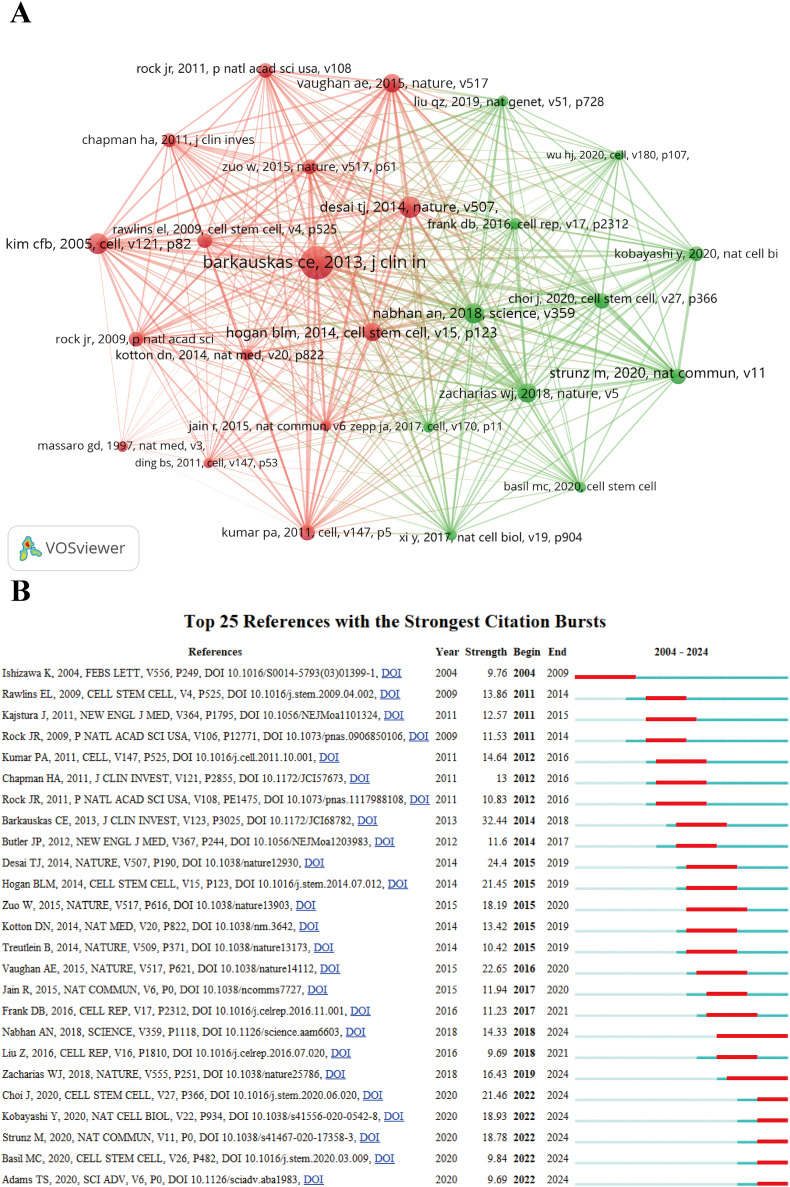
The visualization of co-cited references on research of alveolar regeneration. A collaborative network was constructed based on 26 references whose number of co-citation is more than or equal to 50. (attraction/repulsion values: 3/-1) (B) Top 25 references with strong citation bursts. A red bar indicates high citations in that year.
Recent research on epithelial cell stemness and regenerative mechanisms in the alveolar region continues to advance, progressively revealing key cellular dynamics in lung tissue homeostasis maintenance and injury repair. Existing studies have systematically explored the role of alveolar type II epithelial cells (AEC2s) in alveolar maintenance and regeneration through various approaches including in vivo clonal lineage tracing, different injury-repair models, and in vitro culture of purified cell populations [15]. Research demonstrates that AEC2s expressing surfactant protein C (SFTPC) can undergo long-term self-renewal and differentiate into type I epithelial cells (AEC1s), exhibiting typical long-term stem cell characteristics; when large numbers of AEC2s are specifically eliminated, surviving individual cells can rapidly undergo clonal expansion and widespread dispersal. Further three-dimensional culture experiments show that single labeled AEC2s can form “alveolospheres” containing both self-renewal and differentiation capabilities, and this growth and differentiation depends on co-culture support from PDGFRα+ stromal cells, suggesting that local mesenchymal cells may constitute an important component of the alveolar stem cell microenvironment.
Building upon this foundation, further research combining molecular markers, lineage tracing, and clonal analysis has described the dynamic process of alveolar epithelial cell lineage evolution across temporal scales [44]. Results show that during embryonic development, both AT1 and AT2 cells originate from a bipotent progenitor; however, postnatally, newly generated AT1 cells are primarily produced by rare mature AT2 cells through self-renewal and slowly expanding clonal foci. Notably, the stem cell function of AT2 cells is universally activated following alveolar type I cell injury, and their self-renewal capacity can be induced in vitro by EGFR ligands or significantly enhanced in vivo through expression of the oncogene Kras (G12D), forming multicentric, clonal adenomas. This discovery not only reveals the functional conversion of postnatal AT2 cells as alveolar stem cells but also suggests the dual role of the EGFR-KRAS signaling axis in stem cell self-renewal and tumorigenesis.
Simultaneously, addressing the regional heterogeneity of lung epithelial stem cells, another study isolated and identified BASCs located at the bronchiolar-alveolar duct junction [14]. This cell population demonstrates resistance to bronchiolar and alveolar region injury and significantly expands during injury repair processes, with in vitro clonal analysis showing self-renewal and multipotent differentiation capabilities. Furthermore, BASCs can expand in vitro under oncogenic K-ras stimulation and form pre-tumor foci in vivo, further supporting their role as potential cells of origin for lung adenocarcinoma.
Overall, existing research consistently indicates that the alveolar region contains multiple epithelial cell types with stem cell characteristics, including AEC2s and BASCs, which play important roles in homeostasis maintenance, injury repair, and tumorigenesis processes. Simultaneously, signaling regulation from mesenchymal cells in the local microenvironment, EGFR-KRAS pathway activity, and regional cellular heterogeneity collectively constitute a complex regulatory network for alveolar regeneration and disease progression.
3.6. Keyword analysis, thematic development, and future research directions
The keyword network analysis results of this study show that research hotspots in the alveolar regeneration field can be divided into six major directions (Fig. 7A, Supplementary Table 3). Cluster 1 focuses on the application of stem cells, tissue engineering, and regenerative medicine in lung development and injury repair processes, emphasizing basic mechanisms and clinical translation potential; Cluster 2 primarily explores the dynamic changes of alveolar epithelial cell death, inflammatory responses, and tissue regeneration during acute lung injury and pulmonary fibrosis; Cluster 3 concentrates on severe lung injury caused by viral infections such as COVID-19, and emerging biological therapeutic strategies including exosomes and mesenchymal stem cells; Cluster 4 focuses on the key role of AT2 cells in chronic lung disease repair (such as COPD, emphysema) and lung organoid modeling; Cluster 5, based on mouse animal models, applies immunohistochemistry and other methods to study cellular behavior and molecular mechanisms in alveolar regeneration processes; Cluster 6 focuses on the role of AT2 cell proliferation, differentiation, and extracellular matrix remodeling in regeneration processes following lung resection surgery. Overall, these six clusters are interconnected, collectively outlining the complete developmental trajectory of alveolar regeneration research from basic mechanism exploration to disease model applications, and then to cellular and exosome therapeutic prospects, establishing a theoretical foundation for further understanding alveolar regeneration mechanisms and their clinical translation applications.
Fig. 7.
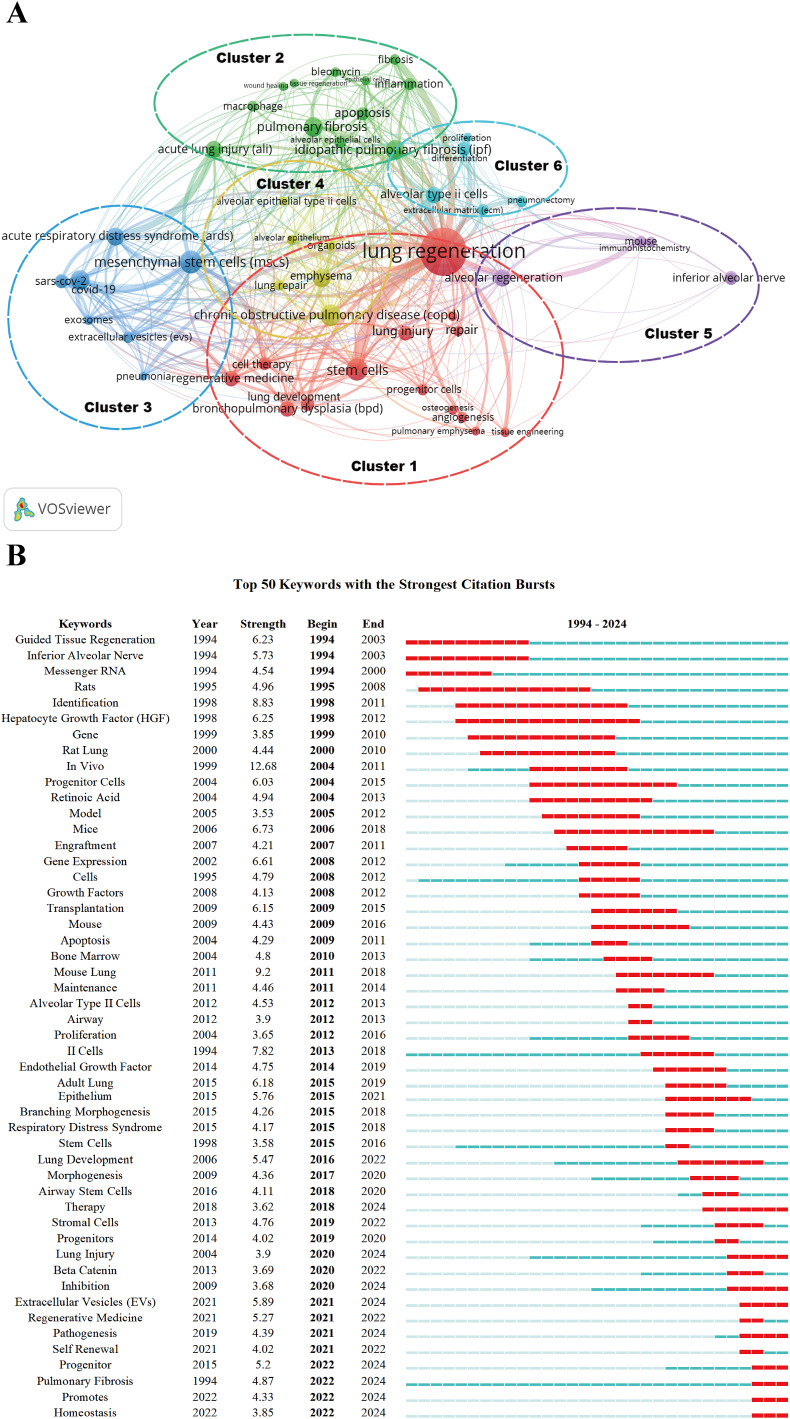
Keywords visualization analysis using VOSviewer. (A) Keyword cluster analysis {more than 13 occurrences (50 keywords) were filtered to map the network (attraction/repulsion values = 1/0)}. (B) Top 50 keywords with strongest citation bursts.
In the analysis of keywords with strongest citation bursts, we found that during 2020–2024, keywords such as therapy, stromal cells, progenitors, lung injury, beta catenin, inhibition, extracellular vesicles, regenerative medicine, pathogenesis, self-renewal, progenitor, pulmonary fibrosis, promotes, and homeostasis showed significant burst trends (Fig. 7B). This change reflects the shift in research focus within the alveolar regeneration field. First, lung injury and fibrosis mechanisms remain fundamental questions, receiving widespread attention especially in the context of the COVID-19 pandemic; second, progenitor cell self-renewal and tissue homeostasis maintenance have become emerging hotspots, prominently highlighting the importance of stem cell behavior in regeneration processes; in terms of therapeutic strategies, the development of stem cell and tissue engineering approaches, as well as the rise of EVs as cell-free therapeutic interventions, demonstrate new breakthrough directions in lung injury repair. Simultaneously, the role of the Wnt/β-catenin signaling pathway in progenitor cell regulation and disease progression has gradually become a research focus. Overall, the alveolar regeneration field is evolving from single injury mechanism studies toward multidimensional exploration of cellular regulation, signal intervention, and cellular/cell-free therapeutic approaches, providing new theoretical support and application prospects for future lung disease treatment.
Furthermore, keyword clustering analysis further reveals that alveolar regeneration research constructs a multi-layered thematic map around dimensions such as “cellular foundations (stem/progenitor cells, immune cells)”, “regulatory factors (PDGFR, EVs)”, “model technologies (organoids, PBMT)”, and “disease contexts (such as IPF)” (Fig. 8A).
Fig. 8.
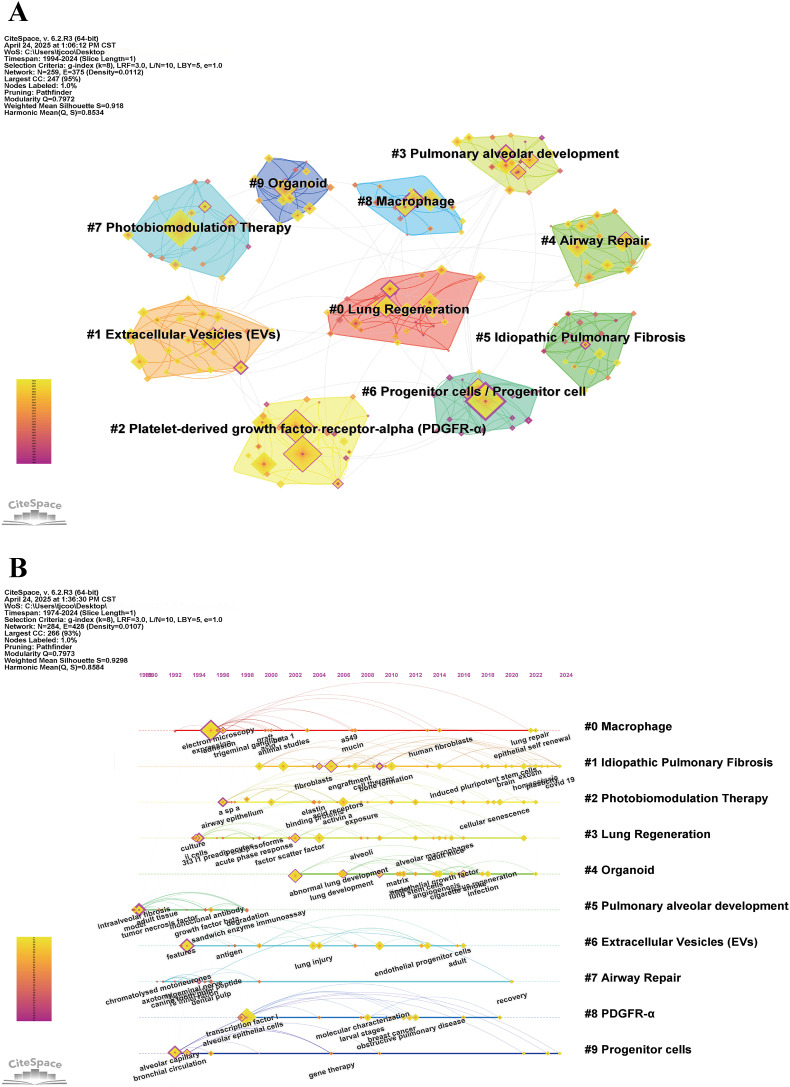
Keywords cluster analysis using CiteSpace. (A) Cluster analysis. (D) timeline analysis.
Keywords Timeline analysis (Fig. 8B) shows that alveolar regeneration research can be traced back to the 1970s, with early focus primarily on lung developmental morphology and observation of tissue repair capabilities. During the 1970s–1980s, animal model-based research focused on spontaneous repair of lung tissue following physical or chemical injury (such as gas inhalation, lobectomy), with attention on alveolar structural reconstruction and respiratory function recovery, but understanding of cellular origins and regulatory mechanisms was limited at that time. Entering the 1990s, with advances in developmental biology, research began analyzing key cells and signaling pathways in alveolar development processes, establishing the status of AT2 cells as important progenitor cells, while molecular mechanisms of airway repair and pulmonary fibrosis also made significant progress. In the 2000s, stem cell biology and lineage tracing technologies promoted research on lung stem/progenitor cell proliferation, differentiation, and homing, using IPF as a clinical model to systematically validate the pathological basis of regenerative imbalance. After the 2010s, research further expanded to immune microenvironment and systemic regulation, with the role of macrophages in inflammation and regeneration balance receiving widespread attention, and the functions of PDGFR-α, Wnt, FGF, and other signaling pathways in epithelial-mesenchymal interactions being progressively clarified. Simultaneously, EVs were established as important intercellular signaling mediators, and organoid model technology also promoted precise modeling and drug screening applications in human lung regeneration research. In recent years, to overcome limitations in lung regenerative capacity, researchers have proposed novel intervention strategies such as PBMT, advancing alveolar regeneration research toward clinical translation.
In summary, alveolar regeneration research has undergone an evolutionary process from anatomical observation to cellular molecular mechanism analysis, and then to interdisciplinary technology integration and clinical translation exploration. Current research themes encompass multiple domains including stem/progenitor cell biology, tissue microenvironment, signal regulation, organoid modeling, and novel therapeutic strategies (see theme nodes 0–9), collectively constructing the overall developmental framework of alveolar regeneration research.
Additionally, the thematic map (Supplementary Fig. 1) further depicts the relevance and developmental trends of various key themes. Analysis indicates that currently underdeveloped research directions are primarily concentrated in repair and regeneration mechanisms following lung injury, the role of matrix environment in tissue regeneration, the potential of stem cells and their derivatives in lung disease treatment, and the function of immune regulation in acute and chronic lung injury repair processes. These directions have important theoretical significance and application prospects in the context of ARDS, COPD, and virus infection-related lung injury, urgently requiring in-depth mechanistic research and innovative therapeutic strategy exploration to establish foundations for promoting clinical translation of alveolar regenerative medicine.
4. Discussion4.1. General information
This study represents the first systematic analysis of global research trends in the alveolar regeneration field (1974–2024). Results demonstrate that alveolar regeneration research has undergone a continuous evolutionary development process. The early phase was primarily characterized by observational exploration, focusing mainly on lung development and post-injury repair potential, but with insufficient understanding of cellular origins and regulatory mechanisms. Following 2000, with breakthroughs in stem cell biology and regenerative medicine, alveolar regeneration gradually established itself as an independent research direction, with research focus shifting toward analysis of stem/progenitor cells, tissue engineering, and signal regulatory mechanisms. Since 2012, particularly following the COVID-19 pandemic outbreak, alveolar regeneration research has entered a rapid expansion period, reflecting a significant increase in global attention to respiratory system repair and regeneration mechanisms. Multiple trend analyses predict that this field will maintain sustained growth momentum in the coming years.
From a country distribution perspective, the United States continues to maintain a dominant position in both publication volume and citation impact, while China, as an emerging scientific force, has rapidly risen. Although there remains a gap in average citation levels, overall research output has grown significantly, with China's contributions becoming increasingly prominent in recent years (strength = 18.05) (Supplementary Fig. 2). Countries such as Italy and Canada, despite lower output volumes, demonstrate strong influence through high citation rates. At the institutional level, the United States universities such as Harvard University and the University of Pennsylvania have played key driving roles, while the rise of Chinese universities such as Shanghai Jiao Tong University demonstrates the enhancement of Asian research capabilities. Journal distribution reveals that alveolar regeneration research has integrated into high-impact platforms, encompassing multiple disciplinary fields including respiratory medicine, stem cell biology, and regenerative engineering, reflecting strong interdisciplinary characteristics. Co-cited author analysis reveals professional differentiation among numerous scholars around different directions such as stem cell dynamics, injury repair mechanisms, and developmental biology. Overall, alveolar regeneration research has gradually evolved from basic exploration toward multidisciplinary integration and clinical translation applications, providing new scientific foundations and intervention strategies for addressing complex respiratory diseases.
In recent years, research in the alveolar regeneration field has gradually formed a diversified development pattern centered on cellular biological foundations, regulatory mechanism exploration, model technology innovation, and disease-context applications. Different research themes are interwoven with each other, constructing a complex and three-dimensional knowledge structure network. In this process, although stem cell-mediated tissue repair, immune microenvironment regulatory mechanisms, and the role of matrix remodeling in disease progression have become focal points, systematic analysis of regeneration mechanisms following lung injury remains relatively weak. Particularly in the context of acute and chronic lung injury, how to efficiently promote regeneration, reconstruct functional lung tissue, and develop clinical application strategies for stem cells and their derivatives have become critical issues requiring breakthrough. Future research should, based on deepening mechanistic understanding, further integrate emerging technological approaches and explore intervention pathways with translational potential to advance alveolar regenerative medicine toward a more application-promising developmental stage.
4.2. Research hotspots and mechanistic insights in lung regeneration
Based on keyword clustering and burst word analysis in this study, the alveolar regeneration field has formed six major research clusters, reflecting the field's evolution from basic mechanism exploration toward clinical translation. The following section provides in-depth analysis of the scientific implications behind each research hotspot and their developmental drivers, combining bibliometric findings.
4.2.1. Central position of endogenous stem cell research
Our co-citation analysis shows that the study by Barkauskas et al. [15] has the highest citation frequency (250 times) with the strongest citation burst (strength = 32.44), marking endogenous stem cell research as the cornerstone of this field. This phenomenon reflects a fundamental shift in the scientific community's understanding of AT2 cells as endogenous stem/progenitor cells. This study confirmed through cell lineage tracing that SFTPC-expressing AT2 cells exhibit typical stem cell characteristics in both homeostasis and post-injury conditions, capable of forming “alveolospheres” with self-renewal and differentiation capabilities, establishing theoretical foundations for subsequent research.
Keywords timeline analysis further reveals that “stem cells” and “progenitor cells” have continuously appeared since 2000, with “progenitors” and “self renewal” becoming burst words during 2020–2024. This evolutionary trajectory indicates that research focus has shifted from early stem cell identification toward in-depth exploration of stem cell behavioral regulatory mechanisms. The highly cited studies by Desai et al. [44] and Kim et al. [14] respectively elucidated the differentiation potential of AT2 cells and BASCs, explaining why stem cell heterogeneity has become a current research hotspot.
4.2.2. Explosive growth in exosome research
Citation burst analysis shows that “EVs” demonstrated significant bursts during 2020–2024, closely related to urgent demands for cell-free therapy following the COVID-19 pandemic. Our journal co-citation network shows that journals publishing EVs-related research have expanded from traditional respiratory medicine journals to comprehensive journals such as Nature Communications, indicating that this field is gaining broader attention from the scientific community.
MSC-derived exosomes have become a research hotspot primarily because they overcome limitations of cell therapy—avoiding transplant rejection and cell survival issues [45]. Literature analysis indicates that miRNA-21, miRNA-146a, and other molecules carried by MSC-EVs can regulate multiple lung injury repair processes [46,47], explaining why “therapy” and “regenerative medicine” simultaneously became burst words. Interdisciplinary citation pattern analysis shows that EVs research presents significant citation flow from Molecular/Biology/Immunology toward Medicine/Medical/Clinical, indicating that this technology is rapidly advancing toward clinical translation.
4.2.3. Emergence and development of microenvironment research
Although “PDGFR-α” did not appear among high-frequency keywords, “stromal cells” as a 2020–2024 burst word reflects the scientific community's enhanced recognition of the importance of cellular microenvironment. Co-author network analysis shows that scholars studying PDGFR-α+ stromal cells have formed close collaborative networks with those studying AT2 cells, reflecting the integration trend of stem cell biology and microenvironment research.
The rise of this research direction stems from recognition of the “AT2 cell niche” concept—that PDGFR-α+ stromal cells support AT2 cell function through secretion of Wnt ligands, FGF factors, and other molecules [17,48]. Journal citation analysis shows that related research is primarily published in high-impact journals such as Cell Stem Cell and Nature Cell Biology, indicating that microenvironment regulation has become a frontier direction in the stem cell field.
4.2.4. Research value of fibrosis as a regeneration failure model
“Pulmonary fibrosis” became a burst word during 2020–2024, while IPF appeared in multiple keyword clusters, reflecting the importance of this disease as a model for studying regeneration failure mechanisms. Institutional collaboration networks show close cooperation between major institutions studying IPF (such as University of Michigan, Yale University) and institutions studying normal alveolar regeneration, reflecting a research strategy from pathology to physiology.
Literature co-citation analysis indicates that IPF-related research extensively cites basic research on AT2 cell dysfunction and epithelial-mesenchymal transition [49]. This citation pattern reveals that the scientific community has recognized that the essence of IPF is dysregulated regeneration mechanisms—AT2 cell senescence [50], mitochondrial dysfunction [51], and abnormal TGF-β signaling activation [52] collectively lead to fibrosis replacing normal regeneration. “Beta catenin” and “inhibition” simultaneously becoming burst words suggests that therapeutic strategies targeting signaling pathways are becoming research priorities.
4.2.5. Emergence of novel therapeutic technologies
Keyword clustering analysis identified PBMT as an independent research direction. Although its publication volume is relatively low, its citation growth rate is high, reflecting the field's continuous exploration of innovative therapeutic approaches. PBMT promotes regeneration through activating mitochondrial function and regulating immune responses [53]. Its mechanistic research is primarily published in journals such as IEEE J Sel Top Quantum Electron, while application research appears in mainstream respiratory medicine journals, showing translation trends from basic to clinical research.
Timeline analysis shows that PBMT research has grown rapidly since 2018, related to the maturation of light-emitting diode technology and increased demand for non-invasive treatments following the COVID-19 pandemic [54]. International collaboration networks show that this technology is primarily driven by researchers from Brazil, Italy, and other countries, and is expanding toward major research forces such as the United States and China.
4.2.6. Organoid technology driving personalized medicine
The development of lung organoid technology has provided a revolutionary experimental platform for lung regeneration research. Researchers have successfully established three-dimensional lung organoid models through human pluripotent stem cells or adult lung stem cells, and these organoids can recapitulate key structural features and functions of alveoli and airways [55]. Particularly noteworthy is that scientists have been able to construct individualized lung organoids from patient-specific induced pluripotent stem cells, providing unique model systems for studying hereditary lung diseases such as cystic fibrosis and IPF [56]. These patient-derived organoids not only retain disease characteristics of original tissues but also demonstrate individualized responses to drug treatments, becoming ideal tools for precision medicine. Previous studies have shown that lung organoids contain multiple lung epithelial cell types, including AT2 cells, Club cells, and ciliated cells, and the differentiation trajectories of these cells are highly similar to in vivo developmental processes (definitive endoderm → foregut endoderm → lung bud organoids) [[57], [58], [59], [60]].
In clinical translation applications, lung organoid technology is driving the development of individualized treatment strategies. Researchers have established high-throughput drug screening platforms, utilizing patient-specific lung organoids to evaluate the efficacy of different treatment regimens [30]. For example, Sachs et al. successfully predicted therapeutic responses to CFTR modulators through lung organoids from cystic fibrosis patients, providing evidence for individualized medication [61]. Additionally, lung organoids have been used to study SARS-CoV-2 infection mechanisms and screen antiviral drugs. Han et al. found significant differences in viral infection susceptibility among lung organoids from different patients, and this difference aligns with the clinical observation of COVID-19 severity heterogeneity [62]. More importantly, researchers are exploring the combination of organoid technology with gene editing, correcting pathogenic mutations in patient organoids through CRISPR/Cas9 technology, laying foundations for future autologous transplant therapy [63,64]. These advances indicate that lung organoid technology is becoming a key bridge connecting basic research and clinical applications, with potential to accelerate the individualized treatment process in lung regenerative medicine.
4.2.7. Compensatory regeneration research following lung resection
Post-lung resection regeneration processes are reflected in keyword clustering analysis, and the formation of this independent research direction reflects the driving effect of clinical surgical needs on basic research. In surgical treatment of lung cancer, severe lung infections, and other diseases, partial lobectomy is a common approach, and post-operative compensatory growth and functional recovery of lung tissue are crucial for patient prognosis. For example, residual lung tissue achieves compensation through alveolar expansion, vascular proliferation, and lung volume increase. Studies show that lung weight increases by an average of 112.4 % one year post-surgery [65], with significant volume increases in the non-operated lung and ipsilateral remaining lobes [66,67].
Our analysis shows that keywords such as “pneumonectomy”, “proliferation”, “alveolar type II cells”, and “extracellular matrix remodeling” form tight networks in this cluster. This indicates that researchers have recognized that post-lung resection regeneration processes involve multiple levels: (i) AT2 cell activation and proliferation as initiating events [68]; (ii) dynamic extracellular matrix remodeling providing scaffolds for tissue regeneration [69]; (iii) formation and maturation of new alveoli restoring respiratory function [70,71].
Timeline analysis shows that post-lung resection regeneration research significantly increased after 2010, related to the development of minimally invasive surgical techniques and expansion of lung cancer surgical indications. Institutional collaboration networks indicate that institutions engaged in this research often possess both strong thoracic surgery clinical capabilities and basic research capacity, reflecting translational medicine research characteristics.
Furthermore, research in this field extensively cites basic research findings from developmental biology and stem cell biology, while also referencing regeneration patterns from other organs such as liver regeneration [72]. This cross-organ knowledge borrowing reflects common principles in regenerative medicine. Particularly noteworthy is the recent application of single-cell sequencing technology in lung resection models, revealing dynamic changes in cell populations at different time points post-surgery, providing new perspectives for understanding cellular and molecular mechanisms of compensatory growth [73].
Future post-lung resection regeneration research may develop in the following directions: (i) developing drugs or biological agents to promote post-operative lung regeneration; (ii) optimizing surgical approaches to maximally preserve regenerative potential; (iii) establishing biomarker systems to predict post-operative lung function recovery; (iv) exploring tissue engineering technology applications in functional reconstruction following partial lung resection.
4.2.8. Overall trends in research hotspot evolution
Comprehensive analysis reveals that alveolar regeneration research presents a clear developmental trajectory: from simple cell identification and mechanism research toward systematic understanding of regeneration processes and therapeutic strategy development. Multi-dimensional bibliometric analysis reveals several key trends: (i) research focus shifting from single cell types toward intercellular interaction networks; (ii) therapeutic strategies shifting from cell transplantation toward cell-free therapy and tissue engineering; (iii) research models shifting from animal models toward humanized systems; (iv) clinical translation shifting from empirical attempts toward mechanism-guided precision therapy.
The formation of these trends reflects both the driving effect of technological advancement and the pulling effect of clinical needs. Particularly, the COVID-19 pandemic outbreak accelerated the translation process from basic research to clinical applications, making “therapy” one of the strongest burst words. In the future, with further integration of multidisciplinary technologies, alveolar regeneration research is expected to achieve breakthrough progress in individualized treatment and clinical translation.
4.3. Future research directions
Based on bibliometric analysis and key burst word analysis in this study, alveolar regeneration research is at a critical turning point of rapid development, with future directions primarily focusing on the following key areas.
4.3.1. Advancing from mechanism exploration to clinical translation
Although understanding of basic alveolar regeneration mechanisms continues to deepen, clinical translation still faces enormous challenges. Future research needs to: (i) establish standardized stem cell/exosome treatment protocols, optimizing delivery routes, dosages, and treatment timing [74,75]; (ii) develop more targeted drug delivery systems to improve treatment efficiency and reduce side effects [76]; (iii) explore combination therapy strategies, such as synergistic applications of anti-fibrotic drugs with pro-regenerative treatments.
4.3.2. Individualized precision medicine strategy development
With the maturation of patient-specific organoid models, individualized treatment will become a priority: (i) establishing biomarker-based patient stratification systems to predict treatment responses [77]; (ii) developing individualized organoid drug screening platforms to formulate optimal treatment regimens for each patient; (iii) integrating multi-omics data (genomics, transcriptomics, proteomics) to construct predictive models guiding clinical decisions [78].
4.3.3. Integrated application of emerging technologies
Technological innovation will drive breakthroughs in alveolar regeneration research: (i) deep integration of spatial transcriptomics with single-cell sequencing to precisely analyze spatiotemporal dynamics of lung injury repair [79,80]; (ii) application of artificial intelligence and machine learning in predictive modeling, drug screening, and treatment optimization [81,82]; (iii) exploration of gene editing technologies (CRISPR/Cas9) in correcting genetic defects and enhancing regenerative capacity [83,84].
4.3.4. Targeted research on priority diseases
Precise interventions for specific lung diseases: (i) long-term repair mechanisms and intervention strategies for COVID-19-related lung injury; (ii) development of early diagnostic and combination treatment regimens for idiopathic pulmonary fibrosis; (iii) breakthroughs in alveolar regeneration barriers in chronic obstructive pulmonary disease; (iv) research on relationships between lung cancer and regenerative dysregulation.
4.3.5. Innovation in interdisciplinary collaboration models
Promoting deep multidisciplinary integration: (i) combination of biomedical engineering with regenerative medicine to develop new biomaterials and tissue engineering scaffolds; (ii) intersection of immunology with stem cell biology to optimize immune regulation for promoting regeneration; (iii) integration of nanotechnology with drug delivery to improve treatment precision and efficiency.
4.3.6. Research gaps and opportunities
Based on thematic map analysis, the following areas still require in-depth exploration: (i) systematic analysis of endogenous repair mechanisms following lung injury; (ii) dynamic regulatory roles of extracellular matrix in tissue regeneration; (iii) precise regulation of immune microenvironment in acute and chronic lung injury repair; (iv) standardized preparation and quality control of stem cell derivatives (such as exosomes).
In summary, lung regeneration research is developing toward more precise, individualized, and translational applications. Through multidisciplinary integration, new technology applications, and clinical needs orientation, major breakthroughs are expected in the future, bringing new therapeutic hope to lung disease patients.
4.4. Limitation and future directions
This study, as a comprehensive bibliometric analysis of the alveolar regeneration field, has the following limitations: (i) Database coverage limitations: This study only used the WOS database. Although multi-database integration faces technical challenges in unifying export file formats, making it difficult for analysis software such as CiteSpace and VOSviewer to conduct comprehensive analysis, and WOS has sufficient data advantages in bibliometrics to meet current analysis needs [36], a single database may still lead to omission of some important literature. (ii) Time window restrictions: Citation data for recent research (particularly 2023–2024) may not have fully accumulated, leading to lag in evaluating the latest research hotspots and breakthrough findings. (iii) Literature quality assessment limitations: Bibliometric analysis is primarily based on quantitative indicators (such as citation counts, H-index, etc.), which may not fully reflect actual research quality and innovation; some breakthrough research that has not yet been widely recognized may be underestimated. (iv) Interdisciplinary research identification challenges: Alveolar regeneration research involves multidisciplinary intersections, and existing disciplinary classifications and keyword analyses may not adequately identify interdisciplinary innovations. (v) Insufficient clinical translation assessment: This study primarily focuses on basic research and mechanism exploration, with relatively weak analysis of clinical trials and translational applications. (vi) Missing dynamic network analysis: This study provides static collaborative network and citation relationship analysis but lacks in-depth exploration of network dynamic evolution. (vii) Industrial translation data limitations: This study is primarily based on academic literature, with less involvement of patent data and industrialization information.
To overcome the above limitations, future research may consider the following improvement measures: (i) Data integration technology development: Develop standardized data conversion tools to solve multi-database file format compatibility issues, achieving integrated analysis of PubMed, Scopus, and other databases with WOS. (ii) Dynamic update mechanism establishment: Establish regularly updated analysis systems, combining data from preprint platforms (such as bioRxiv, medRxiv) to track the latest research dynamics in real-time. (iii) Hybrid evaluation method application: Combine quantitative indicators with qualitative analysis, introduce expert review mechanisms for more comprehensive evaluation of research quality and innovation. (iv) Interdisciplinary identification algorithm optimization: Develop more refined interdisciplinary identification algorithms, construct dynamic thematic evolution models to more accurately capture interdisciplinary innovations. (v) Clinical trial data integration: Strengthen integrated analysis of clinical trial registration databases (such as ClinicalTrials.gov) to systematically evaluate translation rates from basic research to clinical translation. (vi) Temporal network analysis application: Adopt dynamic network analysis methods to reveal evolution of research team collaboration patterns and changes in knowledge flow pathways. (vii) Patent data fusion analysis: Integrate patent databases for comprehensive evaluation of innovation trends and industrialization potential in the lung regeneration field. (viii) Artificial intelligence technology introduction: Utilize machine learning and natural language processing technologies to improve efficiency and accuracy of large-scale literature analysis, achieving intelligent knowledge discovery.
Through these improvement measures, future bibliometric research will be able to more accurately and comprehensively present the complete development picture of the lung regeneration field, providing more valuable references for researchers and decision-makers.
5. Conclusion
Through systematic analysis of 1564 alveolar regeneration-related publications from 1974 to 2024, this study presents the first comprehensive overview of 50 years of development in this field. The research found that the alveolar regeneration field has experienced three phases from basic exploration to rapid development, particularly showing explosive growth following the COVID-19 pandemic in 2020. The United States maintains absolute dominance in this field, while emerging scientific forces such as China are rapidly rising. Identification of six major research clusters reveals the field's evolutionary trend from stem cell biology basic research toward clinical application translation, with exosomes, progenitor cell self-renewal, and regenerative medicine becoming current research frontiers.
Consent to participate
Not applicable, as this study did not involve human participants.
Consent to publish
Not applicable, as this study did not involve human participants.
Code availability
Not applicable.
Ethical approval
The conducted research is not related to either human or animals use.
Authors’ contributions
Si-Cheng Zhou: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review & editing. Ying-Xian Dong: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review & editing. Jie Tian: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review & editing. Guo-Wei Che: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review & editing. Yutian Lai: Software, Supervision, Validation, Visualization, Writing-original draft, Writing-review & editing.
Clinical trial number
Not applicable.
Funding
Project supported by the Natural Science Foundation for Young Scientists of Sichuan Province, China (Grant No. 2025ZNSFSC1541); "Qimingxing" Research Fund for Young Talents (Grant No. HXQMX0026); Noncommunicable Chronic Diseases—National Science and Technology Major Project(Grant No 2023ZD0501706&2023ZD0501702); and the 1.3.5 project for Disciplines of Excellence, West China Hospital, Sichuan University (ZYAI24001).
Declaration of competing interest
All the authors declare that they have no conflict of interest.
Acknowledgements
Not applicable.
Footnotes
Peer review under responsibility of the Japanese Society for Regenerative Medicine.
Appendix A
Supplementary data to this article can be found online at https://doi.org/10.1016/j.reth.2025.09.005.
Contributor Information
Si-Cheng Zhou, Email: zhou.kaizukainaho@foxmail.com.
Ying-Xian Dong, Email: dongyingxian0911@163.com.
Jie Tian, Email: tianjiehxxwk@163.com.
Guo-Wei Che, Email: cheguoweixw@126.com.
Yutian Lai, Email: 896828079@qq.com.
Appendix A. Supplementary data
The following is the supplementary data to this article:
Multimedia component 1
mmc1.pdf (192.3KB, pdf)
Multimedia component 2
mmc2.pdf (162.2KB, pdf)
Multimedia component 3
mmc3.docx (15.9KB, docx)
Multimedia component 4
mmc4.docx (14.8KB, docx)
Multimedia component 5
mmc5.docx (18.1KB, docx)
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
- 1.Ramji H.F., Hafiz M., Altaq H.H., Hussain S.T., Chaudry F. Acute respiratory distress syndrome; A review of recent updates and a glance into the future. Diagnostics. Apr 24 2023;13(9) doi: 10.3390/diagnostics13091528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chen S., Kuhn M., Prettner K., et al. The global economic burden of chronic obstructive pulmonary disease for 204 countries and territories in 2020-50: a health-augmented macroeconomic modelling study. Lancet Glob Health. Aug 2023;11(8):e1183–e1193. doi: 10.1016/s2214-109x(23)00217-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Podolanczuk A.J., Thomson C.C., Remy-Jardin M., et al. Idiopathic pulmonary fibrosis: state of the art for 2023. Eur Respir J. Apr 2023;61(4) doi: 10.1183/13993003.00957-2022. [DOI] [PubMed] [Google Scholar]
- 4.Global age-sex-specific mortality, life expectancy, and population estimates in 204 countries and territories and 811 subnational locations, 1950-2021, and the impact of the COVID-19 pandemic: a comprehensive demographic analysis for the Global Burden of Disease Study 2021. Lancet. May 18 2024;403(10440):1989–2056. doi: 10.1016/s0140-6736(24)00476-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Global burden of chronic respiratory diseases and risk factors, 1990-2019: an update from the Global Burden of Disease Study 2019. eClinicalMedicine. May 2023;59 doi: 10.1016/j.eclinm.2023.101936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hogan B.L., Barkauskas C.E., Chapman H.A., et al. Repair and regeneration of the respiratory system: complexity, plasticity, and mechanisms of lung stem cell function. Cell Stem Cell. Aug 7 2014;15(2):123–138. doi: 10.1016/j.stem.2014.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kotton D.N., Morrisey E.E. Lung regeneration: mechanisms, applications and emerging stem cell populations. Nat Med. Aug 2014;20(8):822–832. doi: 10.1038/nm.3642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Adamson I.Y., Bowden D.H. The type 2 cell as progenitor of alveolar epithelial regeneration. A cytodynamic study in mice after exposure to oxygen. Lab Invest. Jan 1974;30(1):35–42. [PubMed] [Google Scholar]
- 9.Evans M.J., Cabral L.J., Stephens R.J., Freeman G. Transformation of alveolar type 2 cells to type 1 cells following exposure to NO2. Exp Mol Pathol. Feb 1975;22(1):142–150. doi: 10.1016/0014-4800(75)90059-3. [DOI] [PubMed] [Google Scholar]