연구 목적 및 배경
안정기 COPD(만성 폐쇄성 폐질환) 환자에서 전통 중의학(TCM, 주로 중약(CHM)과 침술 조합)을 기존 서양의학 치료(Conventional Treatment, CT)에 보조적으로 사용했을 때의 임상 효과와 안전성을 평가하기 위한 체계적 문헌고찰 및 메타분석. 이전 연구들은 CHM이나 침술을 단독으로 평가했으나, 실제 TCM 임상처럼 CHM과 침술을 결합한 통합 접근을 중점으로 분석. COPD는 전 세계 사망 원인 상위권 질환으로, 증상 완화와 급성 악화 예방이 치료 목표.
방법
- 연구 디자인: PRISMA 지침 준수 체계적 고찰 및 메타분석 (PROSPERO 등록: CRD42019161324).
- 포함 기준: 이중맹검 RCT만 포함, 안정기 COPD 환자(≥18세, GOLD 기준 진단), TCM(CHM ± 침술) + CT vs. CT 단독.
- 검색: PubMed, EMBASE, CNKI 등 다수 데이터베이스 (2019년 11월까지).
- 포함 연구: 100개 RCT (총 8,291명 참가자), 대부분 중국 단일 센터 연구, 샘플 크기 작음(15~83명/arm).
- 주요 평가 지표:
- Primary: 폐기능(FEV₁), 급성 악화율, CAT 점수, 부작용.
- Secondary: 삶의 질(QoL), TCM 증후 점수 및 유효율, 6분 보행거리(6MWD).
- 분석: Random-effects 모델, GRADE로 증거 질 평가.
주요 결과
- 폐기능(FEV₁): 40개 연구 → TCM 보조군에서 평균 0.18L 증가 (MD 0.18, 95% CI 0.08~0.28, p<0.001).
- 급성 악화율: 13개 연구 → 감소 경향 (MD -0.29, 95% CI -0.61~0.03, p=0.075, 비유의).
- CAT 점수: 20개 연구 → 유의한 개선 (MD -2.16, 95% CI -3.44~-0.88, p<0.001).
- TCM 증후 점수: 35개 연구 → 유의한 감소 (MD -3.96, 95% CI -5.41~-2.51, p<0.00001).
- TCM 유효율: 62개 연구 → 증가 (RR 0.89, 95% CI 0.80~0.93, p<0.00001).
- 6분 보행거리(6MWD): 25개 연구 → 평균 37.81m 증가 (MD 37.81, 95% CI 20.90~54.73, p<0.001, I²=14.6%). 이는 운동 내성 향상을 시사하며, COPD 예후 지표로 중요한 의미.
- 삶의 질(QoL): 17개 연구 → 유의한 개선 없음 (SMD -0.01, p=0.859).
- 안전성: 심각한 부작용 없음 (경미한 소화불량 등 보고 적음).
토론 및 결론
중등도~저등급 증거로, 안정기 COPD에서 TCM(주로 CHM)을 CT에 보조적으로 사용하면 폐기능, 증상(CAT), 운동 능력(6MWD), TCM 증후가 개선되며 안전하다. 급성 악화 감소는 경향성만 보임. 강점: 이중맹검 RCT만 포함, 통합 TCM 접근. 한계: 연구 대부분 중국산(편향 가능), 샘플 크기 작음, 위험 편향 불명확(배정 은폐, 맹검 등), 다양한 처방으로 이질성, 출판 편향 의심. 대규모 고품질 RCT 필요. 임상적으로 TCM 보조 사용 고려 가능하나, 증거 한계 인지해야 함.
이 논문은 COPD에서 한약(중의학) 보조 요법의 잠재적 이점을 보여주며, 특히 6MWD 38m 가까이 증가는 폐재활과 유사한 수준의 임상적 의미를 가짐 (MCID 기준 25~35m).

|
| COPD(만성 폐쇄성 폐질환) 환자에서 **6분 보행거리(6-minute walk distance, 6MWD)**는 운동 능력, 예후 예측(사망률, 입원 위험), 치료 효과 평가에 중요한 지표로 사용됩니다. 주요 연구(RCT, 메타분석 중심) 결과를 요약하면 다음과 같습니다. 1. 예후 예측 관련
baseline 평가 시 2회 검사 권장. 중증 COPD에서 특히 예후 지표로 강력하며, 폐재활이나 보조 요법(한약 포함)으로 개선 가능. |
COPD(만성 폐쇄성 폐질환) 환자에서 한약재의 효과를 평가한 주요 RCT 및 메타분석 결과를 요약하면, 대부분 서양의학(흡입제 등) 병용 시 증상 완화, 삶의 질 개선, 급성 악화 감소, 폐기능 향상 등의 효과가 관찰되었습니다. 증거 수준은 중등도(편향 위험 있음)로, 대규모 고품질 연구가 더 필요합니다. 안전성은 대체로 양호하나, 장기 사용 시 모니터링 권장.
주요 한약 처방 및 RCT 요약
- Bufei Granule / Bufei Yishen Granule (보폐과립 / 보폐익신과립)
- 구성: 인삼, 황기 등 폐기 보강, 기허 보충 중심.
- RCT 및 메타분석: 안정기 COPD 환자에서 폐기능(FEV1, FVC) 개선(SMD 0.43~0.70), 삶의 질(SGRQ 점수) 향상, 급성 악화 빈도 감소. 중증(GOLD 3-4) 환자에서 급성 악화 및 입원 감소 확인. 병용 시 효과 우수.
- 증거: 다수 RCT(599명 이상) 및 메타분석에서 유의미한 개선.

| COPD(만성 폐쇄성 폐질환)의 주요 병인 중 하나인 **기도 상피세포 노화(cellular senescence)**를 **Bufei Yishen formula (BYF, 보폐익신방)**가 억제하는지, 그리고 그 기전(특히 AMPK-Sirt1-FoxO3a 경로 활성화와 자가포식(autophagy) 촉진)을 규명하는 것. BYF는 COPD 임상에서 증상 개선 효과가 알려져 있으나, 세포 노화 억제 기전은 불분명했다. 방법
보폐익신방은 고전 처방이 아닌 현대 한의학 처방으로, 중국 허난중의약대학(Henan University of Chinese Medicine)의 Jiansheng Li(李建生) 교수가 COPD(만성 폐쇄성 폐질환)의 폐신기허(肺腎氣虛) 증후를 타겟으로 개발했습니다. 전통 중의학 이론(補肺益腎, 化痰, 活血)에 기반하며, 다수 임상 및 실험 연구(2012년 이후)에서 COPD 치료 효과가 확인되었습니다. 특허 등록된 처방으로, 원방은 12가지 한약재로 구성되며, 이후 유효성분 호환(ECC-BYF) 버전으로 발전했습니다. 원방 처방 및 용량 (주요 연구 기준, 1첩 기준 생약재 g) 연구마다 약간의 변동이 있으나, 대표적인 구성과 용량은 다음과 같습니다 (대부분 Jiansheng Li 교수팀 연구에서 일관됨):
이 처방은 COPD 안정기 치료에 서양의학 병용으로 주로 사용되며, 안전성이 높으나 전문 한의사 처방 하에 사용하세요. 유효성분 버전(ECC-BYF III)은 icariin, astragaloside IV, nobiletin 등 5가지 성분으로 간소화된 형태 |
- San-Huang Gu-Ben Zhi-Ke (삼황고본지해 처방)
- 구성: 황금 등 열청화담 중심.
- RCT: 안정기 COPD(폐비기허증) 환자에서 증상 완화, 안전성 확인. 이중맹검 위약 대조 연구에서 유효성 평가 중.

- Yupingfeng Granule (옥평풍과립)
- 구성: 황기, 백출, 방풍 등 기허 보강.
- RCT: 안정기 COPD에서 CAT 점수 감소, 삶의 질 향상. 급성 악화 예방 효과.
- Bufei Jianpi Formula (보폐건피 처방)
- 구성: 인삼, 황기, 백출 등 폐비 건강.
- RCT: 점막 면역 개선, 장-폐 축(gut-lung axis) 조절 통해 염증 감소, 폐기능 향상.
- Panax Ginseng (인삼 단독 또는 처방 내)
- RCT: 중등도 COPD에서 폐기능(FEV1) 및 운동 능력 개선 보고 있으나, 일부 대규모 RCT(168명)에서 위약 대비 급성 악화 감소나 삶의 질 향상 미미. 염증 억제(NF-κB, MAPK 경로) 기전 확인.
전체 메타분석 요약 (주로 CHM 병용)
- 안정기 COPD: 폐기능(FEV1 MD 0.18L), 6분 보행거리 증가(MD 37m), 급성 악화 감소.
- 증상(CAT, SGRQ) 및 삶의 질 개선, 염증 지표 감소.
- 급성 악화기: 일부 처방(예: Xiaoqinglong Tang)에서 증상 완화.
- 안전성: 부작용 보고 적음, 위약과 유사.
Evid Based Complement Alternat Med
. 2021 Jun 4;2021:5550332. doi: 10.1155/2021/5550332
The Effectiveness of Traditional Chinese Medicine (TCM) as an Adjunct Treatment on Stable COPD Patients: A Systematic Review and Meta-Analysis
K H Chan 1,2,✉, Y Y S Tsoi 3, M McCall 2
- Author information
- Article notes
- Copyright and License information
PMCID: PMC8195656 PMID: 34188688
AbstractBackground
Traditional Chinese medicine (TCM), including Chinese herbal medicine (CHM) and acupuncture, exhibits beneficial effects on stable chronic obstructive pulmonary disease (COPD) such as improving lung function and reducing exacerbation. Previous research studies have examined either CHM or acupuncture alone, which are not the usual practice in TCM clinic setting. We conduct a systematic review for evaluating the clinical effectiveness and safety of TCM by combining CHM and acupuncture.
Methods
Databases are searched from inception to November 2019. Randomized controlled trials examining either acupuncture or CHM on stable COPD are included. Primary outcomes include lung functions, exacerbations, and COPD assessment test. Secondary outcomes include quality of life, TCM syndrome score and effective rate, and 6-minute walk distance. Two independent reviewers extract data and assess the quality of evidence and generate meta-analysis and risk of bias by STATA. This protocol follows the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) guidelines.
Results
100 randomized controlled trials (8291 participants) were included to compare add-on Chinese medicine treatment with conventional treatment (CT). Combining CHM with CT improves FEV1 (MD: 0.18, 95% CI: 0.08, 0.28), exacerbation rate (MD: −0.29, 95% CI: −0.61, 0.03), COPD assessment test (MD: −2.16, 95% CI: −3.44, -0.88), TCM syndrome score (MD: −3.96, 95% CI: −5.41, −2.51) and effective rate (RR: 0.89, 95% CI: 0.80, 0.93), and 6-minute walk test (MD: 37.81, 95% CI: 20.90, 54.73). No serious adverse events were reported. Risk of bias: low to unclear.
Conclusions
This review identifies sufficient moderate-to-low-quality evidence to suggest TCM as an adjunct treatment for stable COPD patients. Though heterogeneity was low among studies, the results were limited and the quality of evidence was low or very low based on small sample sizes and risk of bias. Future studies with larger sample sizes are warranted. The trial is registered with CRD42019161324.
1. Introduction
Chronic obstructive pulmonary disease (COPD) is a common, treatable, and preventable disease, which is characterized by chronic respiratory symptoms and airflow limitation owing to airway and/or alveolar abnormalities caused by persistent exposure to noxious gases or molecules. The major known pathogenesis of COPD is a complex mixture of small airway disease, parenchymal destruction, and chronic airway and/or systemic inflammation.
COPD is an important cause of chronic morbidity and mortality in the world, which ranks the fourth in the leading cause of death and is projected to be the third by 2020 [1, 2]. It is a common, preventable, and treatable disease but poses an economic burden on the society. COPD patients are usually characterized by persistent respiratory symptoms and airflow limitation. Occasionally, they may have acute exacerbation induced by respiratory infection and increase the hospitalization and readmission rate.
Current COPD prevalence data show significant differences among countries, probably because of different diagnostic criteria, survey techniques, and analytical methods [3]. The Burden of Obstructive Lung Diseases (BOLD) program has reported the prevalence and risk factors for COPD in people aged ≥40 in more than 29 countries and found that COPD is more common in men than women [4, 5]. Up to now, there are around three million deaths per year [6]. The prevalence of COPD is predicted to rise in the coming 30 years, and by 2030, there might be over 4.5 million deaths per year from COPD and comorbidities [7, 8].
Diagnosis of COPD is primarily by spirometry which measures the patient's airflow limitation. It is the most widely accepted, easily available, and reproducible test of lung function. A ratio of postbronchodilator forced expiratory volume in first second (FEV1)/forced vital capacity (FVC) <0.70 confirms the presence of persistent airflow limitation [9]. Main symptoms include dyspnea, chronic cough, chronic sputum production, wheezing, and chest tightness. But the severity of airflow limitation is weakly correlated with symptoms in clinical context [10], and spirometry itself has a relatively low specificity [11]. So other symptom assessments are required to categorize COPD patients, which commonly include the Modified British Medical Research Council (mMRC) Questionnaire [12] and COPD Assessment Test (CATTM) [13–15].
COPD patients may suffer acute worsening of respiratory symptoms that lead to additional therapy, namely, acute exacerbations [16–19]. There are three classifications of exacerbations: mild (short-acting bronchodilators (SABDs) only), moderate (SABDs plus antibiotics and/or oral corticosteroids), and severe (hospitalization or visiting emergency room). The best indicator of frequent exacerbations (defined as two or more exacerbations per annum) is a history of earlier treated events [20]. Apart from these tests, physical exercise measurements, such as paced shuttle walk test and the unpaced 6-minute walk test, are also suggested for monitoring patient health status and predicting prognosis [21–23].
For stable COPD, the goals of pharmacological therapy are to reduce symptoms, reduce the frequency and severity of exacerbations, and improve health status and exercise tolerance. Apart from smoking cessation and vaccinations, there are two major classes of medications: bronchodilators and anti-inflammatory drugs. Bronchodilators can increase FEV1 and/or modify other spirometric values and are usually prescribed regularly to prevent or reduce symptoms. Commonly used bronchodilators include short-acting and long-acting beta2-agonists (SABA and LABA, respectively) and short-acting and long-acting anticholinergics (SAMA and LAMA, respectively) [24–27].
Traditional Chinese medicine (TCM) has been using to treat symptoms similar to those in COPD, for instance, cough, sputum, or shortness of breath and has shown beneficial effects for over hundreds of decades. However, there is no such a disease term as COPD in TCM. Instead, COPD patients are classified as having “Fei Zhang” with reference to TCM theory [28]. In a normal TCM clinical setting, either Chinese herbal medicine, acupuncture, or the combination of both is used to relieve COPD symptoms and improve lung functions and/or exercise tolerance [29–32].
TCM is very different from contemporary medicine in both diagnosis and treatment methods. Commonly used TCM treatments include herbal medicinal formula, acupuncture, moxibustion, Tuina, or the combination of them. In a daily TCM healthcare setting, patients with COPD symptoms are often given a set of treatments such as acupuncture/moxibustion, or acupuncture/medicinal formula. Most RCTs for TCM treatments were conducted only on several acupoints, or a single herb or formulae, which is not similar to the usual TCM practice. This study aims to examine the effectiveness and adverse effects of adding TCM treatments on western medicine in stable COPD, to synthesize the best available data towards recommendations of optimal treatment.
The primary objective of this study is to measure the effectiveness of TCM as an adjunct treatment on stable COPD patients in any setting and the adverse events associated with its use in clinical trials measured by lung function and exacerbation rate. The secondary objective of this study is to compare the efficacy of either herbal medicine, acupuncture, or the combination of both on treating stable COPD patients reflected by TCM syndrome score and health status.
Population: patients with stable COPD aged >18 years old, of any sex, education, and socioeconomic status
Interventions: add-on TCM treatment, either herbal medicine, acupuncture, or the combination of both, on conventional medicine
Controls/comparators: mainstream pharmacotherapy for managing stable COPD
Outcomes: lung functions as measured by FEV1 using spirometry, exacerbation rate, 6-minute walk test, and health-related quality of life (QoL)
Study design: double-blind, randomized controlled clinical trials
2. Methods
This systematic review was prepared with reference to the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) [33] and the Cochrane Handbook for Systematic Reviews of Interventions [32] and registered on the international prospective register of systematic review (PROSPERO) on 10.12.2019 (registration number: CRD42019161324). Research protocol and supplementary information are listed in Appendices 1–5.
2.1. Inclusion and Exclusion Criteria for Studies2.1.1. Types of Included Studies
Any randomized controlled trials (RCT) with double-blind assessment of patient-reported outcomes, of which both patients and assessors were blind to the treatments given, were included. RCTs published in a peer-reviewed journal with full text were requested, and unpublished clinical trials with online results available were included.
2.1.2. Types of Excluded Studies
Abstracts alone, nonrandomized trials, case reports, cohort studies, case-control studies, cross-sectional studies, retrospective surveys or chart reviews, editorials, commentaries, and clinical observations were excluded from this systematic review. Other systematic reviews were not included, but the reference lists of similar were searched.
2.1.3. Types of Included Participants
Our search was designed to include (1) patients who were 18 years old or above, regardless of sex, education, race, and socioeconomic status, and (2) patients who were diagnosed with stable COPD according to the diagnostic criteria from the Global Initiative for Chronic Obstructive Lung Disease (GOLD) [6]. Stable COPD patients were defined as patients having mild cough, expectoration, and dyspnea.
2.1.4. Types of Excluded Participants
We excluded patients with other diseases such as asthma, tuberculosis, bronchiolitis, congestive heart failure, or other severe complications because we only wanted to examine the efficacy of TCM on stable COPD.
2.1.5. Types of Interventions
We included any herbal drugs, extracted active ingredients, or formula administered orally, which could be either in a form of TCM granules or boiled soup, and compared to no treatment, placebo, or any active comparator plus conventional medicine. We also included any acupuncture treatment, or dry needling, using any acupoint combinations, and compared to no treatment, placebo, or any active comparator plus conventional medicine. Studies in any healthcare and any global setting were included. Interventions either alone or in combination with each other were included.
2.1.6. Types of Outcome Measures
Primary Outcomes. We included the following items as primary outcomes: (1) lung functions by measuring the change in FEV1 [35]; (2) exacerbations defined as time-to-first exacerbation or exacerbation rate [6]; (3) COPD assessment test [36]; and (4) adverse events of any cause.
Secondary Outcomes. As an assessment of COPD patients' quality of life, we included quality of life such as sleep patterns, mood, and mental health and physical exercise regime on a validated scale; (2) TCM syndrome score and effective rate [37]; and (3) 6-minute walk distance [38] as secondary outcomes.
Search Strategy. The lead author (KH) designed the search strategy and carry out the searches. A broad search strategy was used to cover all Chinese herbal medicine and acupuncture RCTs to include as many relevant and potentially included trials as possible, from studies inception to November 2019.
Electronic Searches. The following databases were searched mainly in English and Chinese languages and filtered for humans:
PubMed
MEDLINE
EMBASE
Cochrane Central Register of Controlled Trials (CENTRAL)
Chinese National Knowledge Infrastructure (CNKI)
WANFANG Database
Chinese Scientific and Technological Periodical Database (VIP)
Chinese Biomedical Database (CBM)
Cochrane Library Database
The search strategies were tailor-made to each database with a combination of text words and medical subject headings (MeSH), or an equivalent, and search terms are listed in Table 1.
Table 1.
Search terms used in PubMed.
NumberSearch terms
| 1 | Randomized controlled trial |
| 2 | RCT |
| 3 | Randomized |
| 4 | Randomly |
| 5 | Trial |
| 6 | Groups |
| 7 | Controlled clinical trial |
| 8 | 1 or 2-7 |
| 9 | Chronic obstructive pulmonary disease |
| 10 | COPD |
| 11 | Chronic obstructive airway disease |
| 12 | Chronic obstructive respiratory disease |
| 13 | Chronic bronchitis |
| 14 | Emphysema |
| 15 | Chronic airflow obstruction |
| 16 | 9 or 10-15 |
| 17 | Chinese Medicine |
| 18 | Chinese Herbal Medicine |
| 19 | CHM |
| 20 | Traditional Chinese Medicine |
| 21 | TCM |
| 22 | Traditional medicine |
| 23 | Herb∗ |
| 24 | Herb∗medicine |
| 25 | Plant medicine |
| 26 | Herb formula |
| 27 | Herb decoction |
| 28 | 17 or 18-27 |
| 29 | Acupuncture |
| 30 | Acupoint∗ |
| 31 | Needling |
| 32 | Dry needling |
| 33 | 29, 30–32 |
| 34 | 8 and 16 and 28 and 33 |
Moreover, the following online registries were searched in English and Chinese language and filtered for humans:
ClinicalTrials.gov
The metaRegister of controlled trials (mRCT)
The World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP)
Searching Other Resources. Bibliographies and reference lists of related publications which match the eligibility criteria were hand searched, such that we did not miss any important references during the selection process.
2.2. Data Collection and Analysis2.2.1. Data Extraction and Management
Two reviewers (KH and YYS) independently extracted study information and outcome data using a standardized data extraction table for RCTs only [39] that includes title, first author, publication year, country, sample size, age and sex of participants, intervention, treatment duration, follow-up period, outcomes, and adverse events. Extracted data were cross-checked and entered into STATA (version 16). Any disagreements about extracted data were adjudicated by the third reviewer (MM) and were resolved by discussion and consensus.
2.2.2. Risk of Bias Assessment
Two authors (KH and YYS) independently assessed the risk of bias for each record using the Cochrane Risk of Bias Tool as reported in the Cochrane Handbook for Systematic Reviews of Interventions [32].
A risk of bias table was included as part of each characteristic of included studies table. When facing disagreements about the risk of bias, a third reviewer (MM) adjudicated and disagreements were resolved by discussion. The risk of bias was assessed at the individual study level and the risk of bias was also considered when assessing Grading of Recommendations, Assessment, Development, and Evaluation system (GRADE) [40].
These seven domains were assessed for each included study as outlined by the Cochrane Handbook for Systematic Reviews of Interventions [32]:
Random sequence generation (examine potential selection bias): studies were assessed for the methods used to generate the allocation sequence
Allocation concealment (examine potential selection bias): studies were assessed for the methods used to conceal allocation to interventions before the study starts
Blinding of participants and personnel (examine potential performance bias): studies were assessed for methods used to blind the participants and personnel from knowing which intervention a participant would receive
Blinding of outcome assessment (examine potential detection bias): studies were assessed for methods used to blind the outcome assessors from knowing which intervention a participant would receive
Incomplete outcome data (examine potential attrition bias): studies were assessed for the nature, number, and handling of incomplete outcome data
Selective reporting (examine potential reporting bias): studies were assessed whether all planned outcomes were reported in the results
Other bias: studies were assessed for any additional sources of bias as low, unclear, or high and provided rationale
2.2.3. Assessment of Heterogeneity
To evaluate clinical heterogeneity, only studies with similar conditions and treatments were compared to get a clinically useful result [32]. Statistical heterogeneity was assessed visually [41] with the I2 statistic and p value. If I2 was larger than 50%, possible reasons were discussed [32].
2.2.4. Data Synthesis
The meta-analysis was conducted on extracted data using STATA (version 16) using a random-effects model. Binary data were expressed as risk ratio with 95% confidence intervals (CIs) and were analyzed by Mantel–Haenszel method. For continuous variables, mean difference (MD) with 95% CIs are calculated. Heterogeneity was examined by I2 tests.
Quality of Evidence. GRADE was used to assess the quality of evidence related to each outcome measure and to provide recommendations for clinical practice [32, 40]. A GRADE rating was assigned for each primary and secondary outcome using the four key levels: high, moderate, low, or very low quality, with reasons provided to upgrade or downgrade [40]. Under certain circumstances, the overall GRADE rating might require adjustment. For instance, a study reported very small sample sizes and results were at risk of being down to play of chance [42]. On the other hand, if no data were reported for an outcome, the term “no evidence” or “lack of evidence” could imply there were data and that the results might show no evidence of effect.
2.2.5. Subgroup Analysis
Subgroup analyses were performed to assess factors such as different TCM dosage, forms, duration of treatment, acupoints used, and measurements of results to see whether they have any impact on the effect estimate. Sensitivity analysis was conducted to examine heterogeneity. The effect of methodological quality, sample size, or missing data was also considered. Analysis was repeated after removing methodologically low-quality studies.
2.2.6. Publication Bias
If more than ten studies were selected, the Egger regression test was used to assess any possible publication bias [43].
2.2.7. Ethical Considerations
There were no ethical issues or approvals needed for this type of study as it used aggregate data already anonymized.
3. Results3.1. Study Identification
The PRISMA study flowchart of search results is shown in Figure 1.
Figure 1.
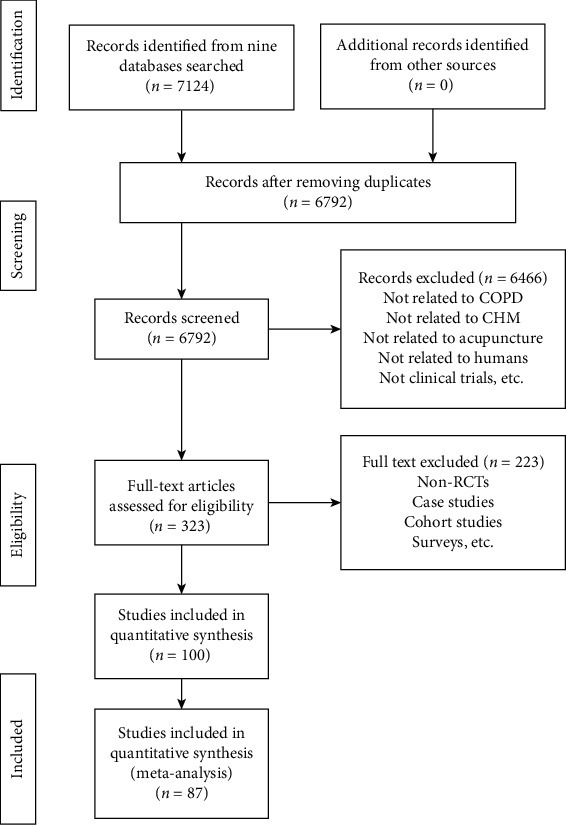
PRISMA study flowchart of search results.
Updated on 1 March 2020, a total of 7124 records are identified from databases and 0 records are found from other sources. After removing duplicates, 6792 titles and abstracts are screened and 323 full-text articles are obtained. Among them, 100 articles are included and 223 are excluded with reasons provided in Appendix 7. No studies are ongoing or under assessment.
3.2. Description of Included Studies
Table 2 summarizes the basic characteristics of 100 included studies. We report sample sizes, ages, course of the disease, and gender for control and intervention groups. Types of Chinese medicine, treatment duration, baseline difference, and quality control are also listed.
Table 2.
Basic characteristics of included studies.
AuthorYearSample size (intervention/control)Age (years) (course of disease (years))Gender (male/female)Intervention#ControlTreatment duration (weeks)Baseline differenceQuality controlControlInterventionControlIntervention
| Wang | 2019 | 48/48 | 63.04 ± 8.39 (12.17 ± 3.51) | 62.87 ± 9.35 (12.63 ± 3.84) | 25/23 | 27/21 | MSZYQD + CT | CT | 12 | NSD | NR |
| Zhu | 2019 | 45/45 | 63.6 ± 8.4 (13.5 ± 7.4) | 61.6 ± 8.5 (13.4 ± 7.3) | 23/22 | 25/20 | YQYYTBLMZY + CT | CT | 4 | NSD | NR |
| Chen | 2019 | 64/64 | 67.89 ± 9.58 (3.12 ± 0.45) | 66.99 ± 10.77 (3.21 ± 0.36) | 31/33 | 32/32 | MXBFD + CT | CT | 12 | NSD | NR |
| Zhou | 2019 | 50/50 | 67.50 ± 6.51 (NR) | 63.74 ± 6.5 (NR) | 31/19 | 32/18 | BSNQG + CT | CT | 8 | NSD | NR |
| Liu | 2019 | 51/52 | 61.75 ± 8.2 (6.05 ± 2.33) | 62.40 ± 0.31 (5.93 ± 2.47) | 34/17 | 36/16 | BFHXD + CT | CT | 12 | NSD | NR |
| Zhang | 2019 | 30/30 | 64.02 ± 3.49 (8.02 ± 2.64) | 63.82 ± 3.67 (7.85 ± 2.71) | 16/14 | 17/13 | MSZJQD + CT | CT | 8 | NSD | NR |
| Zhang | 2019 | 72/72 | 45.18 ± 6.30 (7.25 ± 1.99) | 45.30 ± 6.12 (7.38 ± 1.99) | 41/31 | 45/27 | SMZYYFD + CT | CT | 12 | NSD | NR |
| Lin | 2019 | 33/33 | 68.24 ± 9.76 (22.30 ± 2.75) | 70.62 ± 9.38 (23.52 ± 2.89) | 20/13 | 21/12 | YSBFG + CT | CT | 24 | NSD | NR |
| Wang | 2019 | 73/73 | 62.05 ± 5.13 (6.40 ± 1.25) | 63.32 ± 4.59 (6.02 ± 1.14) | 47/16 | 44/29 | SMQFWSHTD + CT | CT | 12 | NSD | NR |
| Jin | 2019 | 30/30 | NR | NR | NR | NR | SLBZDAAS + CT | CT | 12 | NSD | NR |
| Li | 2019 | 40/40 | 68.25 ± 15.21 (7.82 ± 2.53) | (8.15 ± 2.41) | 25/15 | 23/17 | FZHTQYM + CT | CT | 12 | NSD | NR |
| Liang | 2019 | 40/40 | 62 ± 10 (6 ± 5) | 64 ± 10 (6 ± 5) | 23/17 | 22/18 | GBQFD + CT | CT | 12 | NSD | NR |
| Wang | 2019 | 30/30 | NR | NR | NR | NR | SLBZDAAS + CT | CT | 12 | NSD | NR |
| Chen | 2019 | 38/38 | 67.52 ± 3.22 (3.03 ± 0.38) | 67.56 ± 3.24 (3.05 ± 0.37) | 19/19 | 20/18 | SQBFD + CT | CT | 8 | NSD | NR |
| He | 2019 | 38/38 | 62.5 ± 0.5 | 62.4 ± 0.6 | 21/17 | 20/18 | SQBFD + CT | CT | 8 | NSD | NR |
| Yun | 2019 | 42/42 | 63.5 ± 11.2 (12.5 ± 4.6) | 67.2 ± 14.1 (10.5 ± 5.2) | 19/23 | 22/20 | SQGBD + CT | CT | 24 | NSD | NR |
| Zeng | 2019 | 30/30 | 64.43 ± 6.95 (9.5 ± 2.74) | 66.20 ± 7.27 (9.0 ± 2.03) | 19/11 | 16/14 | LWBQG + CT | CT | 32 | NSD | NR |
| Ke | 2019 | 58/58 | 45.6 ± 7.1 (3.14 ± 0.96) | 44.8 ± 6.9 (3.27 ± 0.88) | 31/27 | 35/23 | JPYFD + CT | CT | 3-4 | NSD | NR |
| Feng | 2018 | 60/60 | 60.12 ± 2.76 (8.14 ± 2.23) | 59.42 ± 2.56 (7.58 ± 1.24) | 36/24 | 38/22 | QTJFD + CT | CT | 12 | NSD | NR |
| Huang | 2018 | 40/40 | 65.56 ± 5.83 (5–20) | 64.76 ± 6.46 (6–17) | 29/11 | 30/10 | JSLJD + THSWD + CT | CT | 8 | NSD | NR |
| Xu | 2018 | 30/30 | 61.4 ± 4.9 (12.3 ± 1.7) | 62.6 ± 5.5 (12.6 ± 1.5) | 15/15 | 14/16 | BFHXD + CT | CT | 28 | NSD | NR |
| Liu | 2018 | 55/55 | 51.97 ± 17.34 (9.2 ± 5.3) | 52.61 ± 16.99 (9.4 ± 4.8) | 35/20 | 34/21 | BFHXD + CT | CT | 12 | NSD | NR |
| Kang | 2018 | 75/75 | 61.25 ± 11.57 | 62.2 ± 12.65 | 44/31 | 52/23 | BZYQD + CT | CT | 24 | NSD | NR |
| Wang | 2018 | 38/38 | 65.44 ± 6.85 (6.23 ± 2.35) | 65.96 ± 6.57 (6.43 ± 2.24) | 19/19 | 20/18 | YQBFD + CT | CT | 4 | NSD | NR |
| Lu | 2018 | 29/29 | 58.3 ± 1.3 | 59.2 ± 1.2 | 16/13 | 15/14 | LJZD + CT | CT | 8 | NSD | NR |
| Yang | 2018 | 30/30 | 57.14 ± 10.67 (9.92 ± 6.02) | 56.75 ± 11.14 (10.36 ± 5.74) | 21/9 | 20/10 | FFGSD + CT | CT | 12 | NSD | NR |
| Li | 2018 | 40/40 | 61.2 ± 2.1 (5.4 ± 1.1) | 63.4 ± 1.8 (5.1 ± 1.0) | 28/12 | 29/11 | MZFWDD + CT | CT | 16 | NSD | NR |
| Yang | 2018 | 40/40 | 75.0 ± 4.5 (5.0 ± 2.2) | 74.5 ± 5.0 (5.3 ± 2.1) | 28/12 | 27/13 | PKHTD + CT | CT | 12 | NSD | NR |
| Gao | 2018 | 80/80 | 59.7 ± 6.4 (16.4 ± 2.1) | 58.8 ± 5.9 (15.9 ± 2.7) | 54/26 | 51/29 | SGDHP + CT | CT | 8 | NSD | NR |
| Zhang | 2018 | 60/60 | 62.39 ± 5.43 (13.95 ± 3.79) | 64.36 ± 5.65 (13.19 ± 3.99) | 37/23 | 34/26 | SGBFD + CT | CT | 12 | NSD | NR |
| Wu | 2018 | 15/15 | 51.46 ± 8.42 (10.01 ± 7.19) | 53.25 ± 7.51 (9.85 ± 8.15) | 8/7 | 6/9 | SLBZD + CT | CT | 12 | NSD | NR |
| Lu | 2018 | 40/40 | 64.49 ± 8.22 (7.82 ± 0.92) | 65.28 ± 8.31 (7.79 ± 0.90) | 25/15 | 26/14 | SQBFD + CT | CT | 4 | NSD | NR |
| Feng | 2018 | 42/42 | 63.1 ± 8.7 (NR) | 64.7 ± 9.2 (NR) | 24/18 | 25/17 | SQBFD + CT | CT | 12 | NSD | NR |
| Yue | 2018 | 43/42 | 528.±4.9 (12.3 ± 3.7) | 53.6 ± 3.2 (11.4 ± 2.2) | 25/18 | 23/19 | SQBFD + CT | CT | 12 | NSD | NR |
| Xu | 2018 | 60/60 | 65.65 ± 1.36 (10 ± 0.27) | 66.35 ± 2.16 (10 ± 0.65) | 38/22 | 36/24 | MBXXXD + CT | CT | 8 | NSD | NR |
| Tu | 2018 | 30/30 | 60.00 ± 6.80 (11.53 ± 4.90) | 62.60 ± 5.74 (11.92 ± 5.02) | 29/1 | 28/2 | MLJZD + CT | CT | 12 | NSD | NR |
| Li | 2018 | 44/44 | 69.58 ± 1.02 (12.1 ± 3.4) | 69.42 ± 1.03 (13.2 ± 4.5) | 26/18 | 28/16 | YFBJD + CT | CT | 4 | NSD | NR |
| Dai | 2017 | 36/36 | 64 ± 7.2 (18.64 ± 10.61) | 63.33 ± 6.11 (15.61 ± 8.05) | 19/17 | 18/18 | SSSQD + CT | CT | 12 | NSD | NR |
| Zhou | 2017 | 30/30 | 72.05 ± 7.62(6.03 ± 1.09) | 71.89 ± 7.56(6.57 ± 1.14) | 17/12 | 18/12 | MZFWDF + CT | CT | 12 | NSD | NR |
| Liu | 2017 | 40/40 | 64.21 ± 6.91(13.56 ± 8.62) | 66.17 ± 6.02(12.89 ± 7.94) | 27/13 | 25/15 | ZYZYF + CT | CT | 4.29 | NSD | NR |
| Yu | 2017 | 42/41 | 63.34 ± 5.19 (NR) | 63.34 ± 5.19 (NR) | 20/21 | 21/21 | BZYQD + CT | CT | 24 | NSD | NR |
| Zhang | 2017 | 50/50 | 52.02 ± 3.10(12.80 ± 2.11) | 53.23 ± 3.02(13.30 ± 2.01) | 27/23 | 28/22 | SZYQD + CT | CT | 2 | NSD | NR |
| Kong | 2017 | 30/30 | 56.5 ± 13.5(9.5 ± 5.5) | 55 ± 13(9 ± 6) | 19/11 | 17/13 | BFFCD + CT | CT | 12 | NSD | NR |
| Wang | 2017 | 30/30 | 69.43 ± 4.897(5.80 ± 2.709) | 68.00 ± 6.119(6.10 ± 2.746) | 18/12 | 21/9 | ILANKM + CT | CT | 8.57 | NSD | NR |
| Wang | 2017 | 42/41 | 66.43 ± 7.76(5.73 ± 1.64) | 67.08 ± 7.43(6.02 ± 1.58) | 24/17 | 26/17 | BFHXD + CT | CT | 24 | NSD | NR |
| Zhao | 2017 | 42/42 | 61.32 ± 10.03 (NR) | 62.99 ± 9.10 (NR) | 24/18 | 23/19 | BFD + CT | CT | 8 | NSD | NR |
| Gong | 2017 | 45/44 | 59.62 ± 5.97(19.61 ± 4.85) | 59.63 ± 5.96(19.62 ± 5.97) | 24/20 | 24/21 | BFJPYSD + CT | CT | 4 | NSD | NR |
| Dong | 2017 | 33/32 | 66.3 ± 8.3 (NR) | 66.5 ± 8.1 (NR) | 15/17 | 16/17 | GJDCP + CT | CT | 12 | NSD | NR |
| Zhou | 2017 | 35/35 | 53.7 ± 10.9 (NR) | 53.7 ± 10.9 (NR) | 34/36 | 34/36 | SMYFPCD + CT | CT | 12 | NSD | NR |
| Xiao | 2017 | 25/25 | 64.3 ± 5.7(8.5 ± 2.3) | 64.5 ± 5.3(8.3 ± 2.1) | 18/7 | 16/9 | SMWBPSD + CT | CT | 12 | NSD | NR |
| Li | 2017 | 50/50 | 70.06 ± 3.09(6.12 ± 1.12) | 70.61 ± 3.11(6.02 ± 1.05) | 26/24 | 27/23 | SMYYQFD + CT | CT | 1 | NSD | NR |
| Wen | 2017 | 33/34 | 56.85 ± 5.47(8.36 ± 3.63) | 56.85 ± 5.47(8.36 ± 3.63) | NR | NR | FLK + CT | CT | 12 | NSD | NR |
| You | 2017 | 83/83 | 63.75 ± 5.91(7.08 ± 1.51) | 63.37 ± 5.87(6.88 ± 1.53) | 46/37 | 50/33 | YQHTQYTLD + CT | CT | 8.57 | NSD | NR |
| Lu | 2017 | 38/38 | 64.20 ± 13.40 (NR) | 64.20 ± 13.40 (NR) | 22/16 | 19/19 | WBFSM + CT | CT | 24 | NSD | NR |
| Yang | 2017 | 40/40 | NR | NR | 34/6 | 31/9 | FFGSD + CT | CT | 12 | NSD | NR |
| Lu | 2017 | 44/44 | NR | NR | NR | NR | PCGBD + CT | CT | 24 | NSD | NR |
| Lu | 2017 | 31/31 | 63.5 ± 4.1(4.2 ± 0.8) | 64.2 ± 4.2(4.0 ± 0.6) | 17/14 | 18/13 | SLBZPAAS + CT | CT | 12 | NSD | NR |
| Li | 2017 | 38/38 | 40.32 ± 3.12 (NR) | 40.65 ± 3.08 (NR) | 21/17 | 22/16 | MSGP + CT | CT | 8 | NSD | NR |
| Guo | 2017 | 46/46 | 59.1 ± 5.9 (NR) | 58.±5.7 (NR) | 27/19 | 29/17 | YYQFD + CT | CT | 4 | NSD | NR |
| Zhao | 2017 | 50/50 | 67.45 ± 3.14(4.8 ± 1.1) | 65.32 ± 2.25(4.7 ± 1.2) | 34/16 | 38/12 | SMLRD + CT | CT | 2 | NSD | NR |
| Lin | 2017 | 34/34 | 60.49 ± 8.26(7.01 ± 2.39) | 60.49 ± 8.26(6.89 ± 2.60) | 19/15 | 19/15 | MLJZD + CT | CT | 12 | NSD | NR |
| Lou | 2016 | 60/59 | 71.2 ± 5.7(19.23 ± 4.53) | 72.35 ± 4.77(18.42 ± 3.37) | 36/23 | 35/35 | JQNSM + CT | CT | 12 | NSD | NR |
| Ye | 2016 | 42/42 | 63.14 ± 12.08(6.84 ± 2.99) | 62.47 ± 11.08(7.01 ± 3.68) | 24/18 | 22/20 | TBFSM + CT | CT | 12 | NSD | NR |
| Bian | 2016 | 25/25 | 60.3 ± 5.7(16.4 ± 3.6) | 60.3 ± 5.7(16.4 ± 3.6) | NR | NR | BSFCD + CT | CT | 12 | NSD | NR |
| Li | 2016 | 30/30 | 61.33 ± 11.50(14.12 ± 5.3) | 62.2 ± 12.67(12.5 ± 4.88) | 16/14 | 19,11 | ZLFSQXDGFTJ + CT | CT | 8 | NSD | NR |
| Tang | 2016 | 34/34 | 65.87 ± 9.08 (NR) | 63.77 ± 8.64 (NR) | 20/14 | 23/11 | ZCG + CT | CT | 8 | NSD | NR |
| Guo | 2016 | 49/49 | 69.3 ± 2.5(5.4 ± 0.7) | 70.2 ± 2.3(5.6 ± 0.5) | 28/21 | 27/22 | MZFWDF + CT | CT | 12 | NSD | NR |
| Fang | 2016 | 59/59 | 64.02 ± 11.15(6.79 ± 2.14) | 63.87 ± 10.62(6.32 ± 2.02) | 36/23 | 35/24 | SLBZPAAS + CT | CT | 12 | NSD | NR |
| Chen | 2016 | 30/30 | 66.1 ± 5.8(6.3 ± 1.6) | 65.6 ± 5.1(6.5 ± 1.8) | 17/13 | 19/11 | LJF + CT | CT | 8 | NSD | NR |
| Hu | 2016 | 30/30 | 67.90 ± 7.34(16.47 ± 6.95) | 68.63 ± 6.49(14.87 ± 9.07) | 19/11 | 18/12 | BFDAAS + CT | CT | 8 | NSD | NR |
| Liang | 2015 | 42/42 | 63.14 ± 12.08(6.84 ± 2.99) | 62.47 ± 11.08(7.01 ± 3.68) | 24/18 | 22/20 | TBFSM + CT | CT | 12 | NSD | NR |
| Wang | 2015 | 37/37 | 67.13 ± 6.95(3.92 ± 0.39) | 66.72 ± 7.92(3.85 ± 0.44) | 15/22 | 18/19 | BFYSD + CT | CT | 1 | NSD | NR |
| Si | 2015 | 40/40 | 70.43 ± 8.73(16.30 ± 5.19) | 72.04 ± 9.36(18.30 ± 5.84) | 22/18 | 25/15 | BFJPYSD + CT | CT | NR | NSD | NR |
| Wang | 2015 | 36/36 | 60.51 ± 11.03(14.52 ± 5.96) | 60.81 ± 8.18(14.56 ± 6.32) | 22/14 | 21/15 | BZGWHJ + CT | CT | 8 | NSD | NR |
| Li | 2015 | 38/38 | 57.18 ± 9.92 (NR) | 56.63 ± 10.23 (NR) | 23/15 | 20/18 | YQBSHXF + CT | CT | 12 | NSD | NR |
| He | 2015 | 25/25 | NR | NR | NR | NR | SLBZG + CT | CT | 36 | NSD | NR |
| Huang | 2015 | 40/41 | 67.28 ± 4.30(8.9 ± 7.8) | 65.36 ± 2.10(8.8 ± 7.1) | 25/16 | 23/17 | KFZY + CT | CT | 36 | NSD | NR |
| Zhang | 2015 | 45/30 | 63.2 ± 11.5 (NR) | 63.8 ± 10.7 (NR) | 18/22 | 29/16 | SXDAAS + CT | CT | 12 | NSD | NR |
| Luo | 2014 | 80/80 | 66.8 (15.49) | 64.8 (16.32) | 62/18 | 64/16 | BFPCD + CT | CT | 12 | NSD | NR |
| Wen | 2014 | 40/40 | 68.3 ± 9.0(15.4 ± 3.6) | 68.6 ± 9.2(15.7 ± 3.8) | 28/12 | 30/10 | BFDAAS + CT | CT | 8 | NSD | NR |
| Li | 2014 | 49/49 | 61.37 ± 4.18(10.6 ± 8.9) | 63.52 ± 3.67(11.4 ± 7.3) | 30/19 | 32/17 | BFNSF + CT | CT | 3 | NSD | NR |
| Qi | 2014 | 80/80 | 55.9 ± 3.9(8.2 ± 3.1) | 56.3 ± 4.2(7.9 ± 4.2) | 46/34 | 44/36 | WSBFHTT + CT | CT | 8 | NSD | NR |
| Zeng | 2014 | 43/42 | 66.9 ± 7.9(14.8 ± 3.9) | 67.4 ± 7.2(15.3 ± 4.5) | 29/13 | 28/15 | MSZYQD + GZLMD + CT | CT | 12 | NSD | NR |
| Zhang | 2014 | 30/30 | 40.46 ± 16. (NR) | 45.35 ± 15.32 (NR) | 12/18 | 7/23 | QZYQDG + CT | CT | 2 | NSD | NR |
| Chen | 2013 | 30/30 | 63.51 ± 11.24(10.86 ± 7.54) | 62.54 ± 12.56(11.61 ± 8.44) | 18/12 | 16/14 | BFYSQTD + CT | CT | 42 | NSD | NR |
| Tan | 2013 | 45/34 | NR | NR | NR | NR | BZYQM + CT | CT | 2.14 | NSD | NR |
| Zhang | 2013 | 33/33 | 63.57 ± 11.47(33.57 ± 10.97) | 62.03 ± 10.94(33.26 ± 9.41) | 19/14 | 20/13 | QYJDF + CT | CT | 4 | NSD | NR |
| Liang | 2013 | 40/40 | NR | NR | 38/2 | 33/7 | RFJPBSD + CT | CT | 12 | NSD | NR |
| Jiang | 2013 | 30/30 | 61.7 ± 3.2 (NR) | 64.0 ± 2.1 (NR) | 19/11 | 17/13 | SLBZG + CT | CT | 24 | NSD | NR |
| Wang | 2013 | 30/30 | NR | NR | NR | NR | SLBZDAAS + CT | CT | 12 | NSD | NR |
| Dai | 2013 | 40/40 | NR | NR | NR | NR | SMBPYQD + CT | CT | 12 | NSD | NR |
| Chen | 2012 | 30/30 | 60.81 ± 8.57 (NR) | 65.63 ± 7.43 (NR) | 21/9 | 23/7 | GBQTHYD + CT | CT | 24 | NSD | NR |
| Chen | 2011 | 50/50 | 67.73 ± 6.11(9.75 ± 3.56) | 69.51 ± 5.41(9.45 ± 4.12) | 41/9 | 38/12 | FGFZM + CT | CT | 12 | NSD | NR |
| Gong | 2011 | 30/30 | 68.4 ± 6.2 (NR) | 67.4 ± 6.8 (NR) | 19/11 | 17/13 | SETGM + CT | CT | 24 | NSD | NR |
| Tang | 2010 | 34/35 | 71.00 ± 10.53(15.46 ± 10.89) | 72.18 ± 10.78(14.53 ± 9.15) | 26/9 | 23/11 | JPYSD + CT | CT | 12 | NSD | NR |
| He | 2010 | 49/49 | NR | NR | NR | NR | SETGM + CT | CT | 24 | NSD | NR |
| Wang | 2009 | 36/28 | 61.5 ± 4.8(15.6 ± 4.7) | 62.1 ± 5.3(16.3 ± 5.1) | 21/7 | 25/11 | SMBFTFD + CT | CT | 24 | NSD | NR |
| Wang | 2008 | 32/26 | 61.5 ± 4.8(15.6 ± 4.7) | 62.1 ± 5.3(16.3 ± 5.1) | 20/6 | 23/9 | BFTFD + CT | CT | NR | NSD | NR |
| Jiang | 2008 | 30/25 | 63.2 ± 5.3(13.12 ± 3.38) | 61.6 ± 6.1(15.25 ± 4.01) | 17/8 | 23/7 | SMYQHXD + CT | CT | 24 | NSD | NR |
| Hong | 2018 | 30/30 | 68.59 ± 7.72 (15.70 ± 5.59) | 67.93 ± 7.78 (14.57 ± 5.50) | 27/1 | 28/0 | YFN + CT | CT | 8 | NSD | YES |
NSD = not significantly different; NR = not reported; CT = conventional treatment. # Detailed description of CHM in Appendix 6.
3.2.1. Design
All studies are randomized, double-blind, and controlled clinical trials and report primary and secondary outcomes.
3.2.2. Sample Size
Sample sizes range from 15 participants per arm [44] to 83 participants per arm [45]. Ages range from 40.32 ± 3.12 (mean ± SE) [46] to 71.2 ± 5.7 [47] in the control group and from 40.65 ± 3.08 [46] to 72.35 ± 4.77 [47] in the intervention group. Ten studies do not report the mean age [48–57].
3.2.3. Participants
All studies recruit participants according to the GOLD guidelines [6], and all participants are in stable phase with stages between II and IV. The course of disease (in years) ranges from 3.03 ± 0.38 (mean ± SE) [58] to 33.57 ± 10.97 in the control group [28] and from 3.05 ± 0.37 [58] to 33.26 ± 9.41 [28] in the intervention group. Thirty studies do not report the course of years shown in Table 2.
3.2.4. Setting and Location
All studies are single-centered trials and based in hospital settings. The location of studies scatters across different provinces in China.
3.2.5. Interventions
Comparison Arms. There is no acupuncture plus conventional medicine versus conventional treatment (CT) identified. Ninety-nine studies compare one Chinese herbal medicine (CHM) formulae plus CT with conventional medicine. One study has three arms: conventional medicine, CHM decoction plus CT, and CHM powder plus CT [48].
Types of Chinese Medicine. One hundred different CHM formulae are used and detailed compositions and dosages of each formula are shown in Appendix 6. Conventional medicine is prescribed with reference to GOLD guidelines.
Duration of Treatment. The duration of treatment differs across studies, which ranges from 1 week [59] to 42 weeks [60]. The mean duration of treatment is 14 weeks. Two studies do not report the duration of treatment [61, 62].
3.2.6. Outcomes
Table 3 summarizes the availability of outcome measures reported. Forty studies report a change in FEV1 (mean ± SE). Thirteen studies report exacerbation rate (mean ± SE) as a continuous outcome at the study endpoint. Twenty studies report COPD assessment test (mean ± SE). Twenty-five studies report 6-minute walk test (mean ± SE). Thirty-five studies report TCM syndrome score (mean ± SE) and sixty-two studies report TCM effective rate as dichotomous outcome. For quality of life, seventeen studies report in different QoL scales (mean ± SE), including SGRQ and COPD quality of life. Only one study reports all withdrawals [63]. Eight studies report adverse events of any cause with reason provided. One study reports withdrawals due to lack of efficacy [56]. Thirteen studies report none of the primary and secondary outcomes.
Table 3.
Availability of outcome indexes.
AuthorYearAvailable outcome index
| Wang | 2019 | TCM syndrome score, TCM effective rate, FEV1, 6MWT |
| Zhu | 2019 | Nil |
| Chen | 2019 | FEV1 |
| Zhou | 2019 | TCM effective rate, CAT, QoL, exacerbation rate |
| Liu | 2019 | TCM syndrome score, TCM effective rate, FEV1, 6MWT, QoL, adverse events |
| Zhang | 2019 | TCM syndrome score, FEV1, CAT, TCM effective rate |
| Zhang | 2019 | TCM syndrome score, FEV1, 6MWT, exacerbation rate, TCM effective rate |
| Lin | 2019 | TCM syndrome score, CAT, TCM effective rate |
| Wang | 2019 | TCM syndrome score, FEV1, 6MWT, TCM effective rate, adverse events |
| Jin | 2019 | Nil |
| Li | 2019 | TCM syndrome score, FEV1, 6MWT |
| Liang | 2019 | TCM syndrome score, CAT |
| Wang | 2019 | Nil |
| Chen | 2019 | FEV1 |
| He | 2019 | TCM syndrome score, FEV1 |
| Yun | 2019 | TCM syndrome score, TCM effective rate, FEV1, CAT |
| Zeng | 2019 | TCM effective rate, exacerbation rate, CAT |
| Ke | 2019 | FEV1, QoL |
| Feng | 2018 | TCM syndrome score, TCM effective rate, FEV1, 6MWT, QoL |
| Huang | 2018 | TCM effective rate, FEV1, CAT, 6MWT |
| Xu | 2018 | TCM syndrome score, TCM effective rate, FEV1, QoL |
| Liu | 2018 | TCM syndrome score, FEV1 |
| Kang | 2018 | TCM syndrome score |
| Wang | 2018 | TCM effective rate, QoL, FEV1 |
| Lu | 2018 | TCM effective rate, FEV1 |
| Yang | 2018 | TCM effective rate, FEV1, CAT, 6MWT, exacerbation rate |
| Li | 2018 | TCM effective rate, exacerbation rate, 6MWT, QoL |
| Yang | 2018 | TCM effective rate, FEV1 |
| Gao | 2018 | TCM effective rate |
| Zhang | 2018 | TCM effective rate, TCM syndrome score, CAT, FEV1 |
| Wu | 2018 | TCM syndrome score, 6MWT, CAT |
| Lu | 2018 | TCM syndrome score |
| Feng | 2018 | TCM effective rate |
| Yue | 2018 | TCM effective rate |
| Xu | 2018 | TCM effective rate, FEV1 |
| Tu | 2018 | CAT |
| Li | 2018 | TCM effective rate |
| Dai | 2017 | FEV1, CAT |
| Zhou | 2017 | TCM effective rate, exacerbation rate |
| Liu | 2017 | 6MWT, adverse events |
| Yu | 2017 | TCM effective rate, QoL, 6MWT |
| Zhang | 2017 | FEV1, CAT, TCM effective rate |
| Kong | 2017 | FEV1 |
| Wang | 2017 | TCM syndrome score, TCM effective rate, 6MWT, CAT |
| Wang | 2017 | FEV1, 6MWT |
| Zhao | 2017 | TCM syndrome score |
| Gong | 2017 | FEV1, TCM effective rate |
| Dong | 2017 | TCM syndrome score, TCM effective rate |
| Zjou | 2017 | TCM effective rate |
| Xiao | 2017 | FEV1, QoL, 6MWT |
| Li | 2017 | TCM effective rate, FEV1, adverse events |
| Wen | 2017 | TCM effective rate, FEV1 |
| You | 2017 | TCM effective rate, adverse events |
| Lu | 2017 | Nil |
| Yang | 2017 | Nil |
| Lu | 2017 | TCM syndrome score, TCM effective rate |
| Lu | 2017 | TCM effective rate |
| Li | 2017 | Nil |
| Guo | 2017 | FEV1, TCM effective rate |
| Zhao | 2017 | TCM effective rate |
| Lin | 2017 | TCM effective rate, TCM syndrome score, CAT |
| Lou | 2016 | TCM syndrome score, 6MWT, exacerbation rate |
| Ye | 2016 | TCM syndrome score, QoL, TCM effective rate, FEV1, 6MWT, exacerbation rate |
| Bian | 2016 | Nil |
| Li | 2016 | 6MWT |
| Tang | 2016 | QoL, TCM effective rate |
| Guo | 2016 | TCM effective rate |
| Fang | 2016 | TCM effective rate, 6MWT |
| Chen | 2016 | TCM effective rate |
| Hu | 2016 | TCM effective rate, 6MWT, CAT |
| Liang | 2015 | TCM syndrome score, TCM effective rate, 6MWT, QoL, FEV1, exacerbation rate |
| Wang | 2015 | TCM effective rate, FEV1 |
| Si | 2015 | QoL |
| Wang | 2015 | CAT |
| Li | 2015 | Nil |
| He | 2015 | TCM effective rate, FEV1, QoL |
| Huang | 2015 | FEV1, 6MWT, adverse events |
| Zhang | 2015 | CAT, 6MWT, TCM effective rate, exacerbation rate, adverse events |
| Luo | 2014 | 6MWT, exacerbation rate, TCM effective rate, adverse events |
| Wen | 2014 | TCM effective rate |
| Li | 2014 | FEV1, TCM effective rate |
| Qi | 2014 | TCM effective rate, TCM syndrome score |
| Zeng | 2014 | Nil |
| Zhang | 2014 | TCM effective rate, TCM syndrome score |
| Chen | 2013 | TCM syndrome score |
| Tan | 2013 | FEV1, TCM effective rate |
| Zhang | 2013 | TCM effective rate, TCM syndrome score, FEV1 |
| Liang | 2013 | TCM syndrome score, 6MWT |
| Jiang | 2013 | CAT, TCM syndrome score, FEV1 |
| Wang | 2013 | TCM effective rate |
| Dai | 2013 | Nil |
| Chen | 2012 | TCM effective rate, TCM syndrome score |
| Chen | 2011 | TCM effective rate, FEV1, TCM syndrome score, exacerbation rate |
| Gong | 2011 | FEV1, TCM effective rate, QoL |
| Tang | 2010 | TCM syndrome score, TCM effective rate, QoL |
| He | 2010 | TCM syndrome score, exacerbation rate |
| Wang | 2009 | Nil |
| Wang | 2008 | Nil |
| Jiang | 2008 | Nil |
| Hong | 2018 | TCM effective rate, CAT, 6MWT, adverse events, all withdrawals |
FEV1 = forced expiratory volume in 1 s; CAT = COPD assessment test; 6MWT = 6-minute walk test; QoL = quality of life.
3.2.7. Language
One full text is written in English. The remaining 99 full texts are written in Chinese and are translated by KH (myself).
3.3. Description of Excluded Studies
Two hundred and twenty-three studies are excluded after reading the full-text articles. Detailed exclusion reasons for each study are shown in Appendix 7.
Sixty-four studies (29%) are excluded because the participants are not stable COPD patients, or there is no evidence to indicate the disease phase.
Ninety-seven studies (43%) are excluded due to interventions. Reasons include that (1) CHM is not administered in the form of decoction or granules, (2) intervention group does not combine with conventional treatment, (3) intervention is not CHM or acupuncture, and (4) there is more than one CHM treatment.
Nine studies (4%) are excluded in the absence of control group or conventional treatment. Twelve studies (6%) do not report the wanted primary and secondary outcomes or do not use “intention-to-treat” analysis.
Forty-one studies (18%) are not included in the light of study designs, with reasons being not randomized or no such evidence.
3.4. Risk of Bias Summary of Included Studies
The risk of bias summary diagram shows the risk of bias for all included studies from low to unclear (Table 4). No study shows a low risk of bias in all six domains. Two studies (2%) show a high risk of bias in one domain [51, 56]. Ninety-nine studies (99%) display unclear risk of bias in four domains and one study (1%) shows unclear risk in two domains [63].
Table 4.
Risk of bias assessments of included studies.
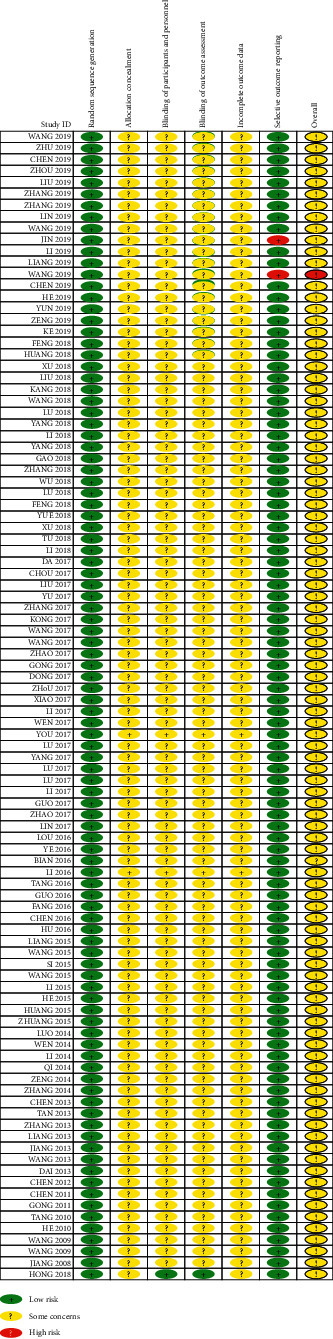 |
3.4.1. Random Sequence Generation (Selection Bias)
All studies describe their randomization methods which are mostly random number generation by 1 : 1 ratio (low risk).
3.4.2. Allocation Concealment (Selection Bias)
All studies do not report information about the concealment method of what types of treatment are given to participants (unclear risk).
3.4.3. Blinding of Participants and Personnel (Performance Bias)
Ninety-nine studies (99%) provide no information on how participants and research personnel are blinded (unclear risk). One study reports adequately the blinding procedures of both participants and personnel (low risk) [63].
3.4.4. Blinding of Outcome Assessors (Detection Bias)
Ninety-nine studies (99%) provide no information how participants and research personnel are blinded (unclear risk). One study reports adequately the blinding procedures of both participants and personnel (low risk) [63].
3.4.5. Incomplete Outcome Data (Attrition Bias)
Ninety-nine studies (99%) do not report dropouts and withdrawals (unclear risk). One study (1%) reports withdrawal numbers but no reasons provided (unclear risk) [63].
3.4.6. Selective Reporting (Reporting Bias)
Ninety-eight (98%) studies report all outcomes as prespecified in their protocol and two studies (2%) miss some outcome data (high risk) [51, 56].
3.5. Outcome Measures
One hundred studies are included with a total of 8,318. Forty studies report change in FEV1. Thirteen studies report exacerbation rate at the study endpoint. Twenty studies report COPD assessment test. Twenty-five studies report 6-minute walk test. Thirty-five studies report TCM syndrome score and sixty-two studies report TCM effective rate as dichotomous outcome. Seventeen studies report various QoL scales. Eleven studies report all withdrawals. Eight studies report adverse events of any cause with reason provided. One study reports withdrawals due to lack of efficacy. Thirteen studies report none of the primary and secondary outcomes.
3.5.1. Primary Outcome: Change in FEV1
Forty studies report the mean and standard error (SE) of FEV1 (in liters) at baseline and endpoint of the study. Data are converted to standard deviation (SD) and meta-analyzed in Figure 2. The results are presented as mean change in FEV1 and SD. The effect estimate is 0.18 (95% CI: 0.08, 0.28; p ≤ 0.001) with I2 = 0.0% (p=1.000), which means intervention significantly increases FEV1 by 0.18 liter with zero heterogeneity.
Figure 2.
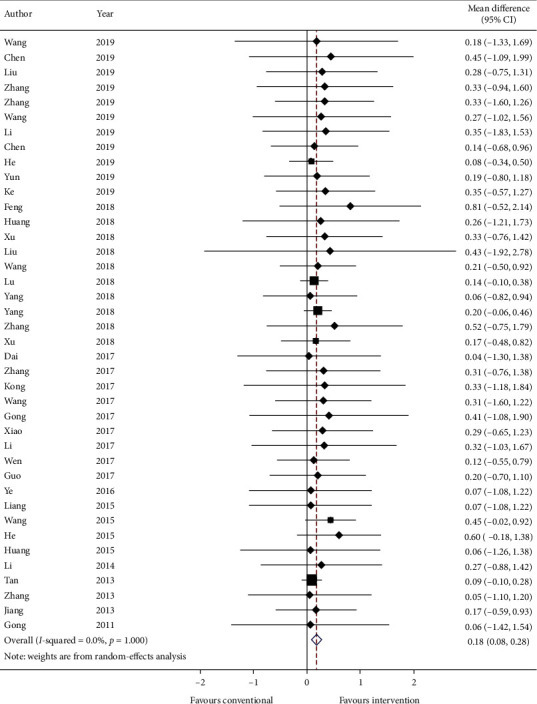
Forest plot of change in FEV1. CI: confidence intervals.
GRADE: the overall quality of this evidence is judged to be moderate, downgraded once for risk of bias and once for imprecision.
3.5.2. Primary Outcome: Exacerbation Rate
Thirteen studies reported the mean exacerbation rate and standard error at the endpoint of the study. Data are converted to standard deviation (SD) and meta-analyzed in Figure 3. The effect estimate was −0.29 (95% CI: −0.61, 0.03; p=0.075733) with I2 = 0.0% (p=0.455).
Figure 3.
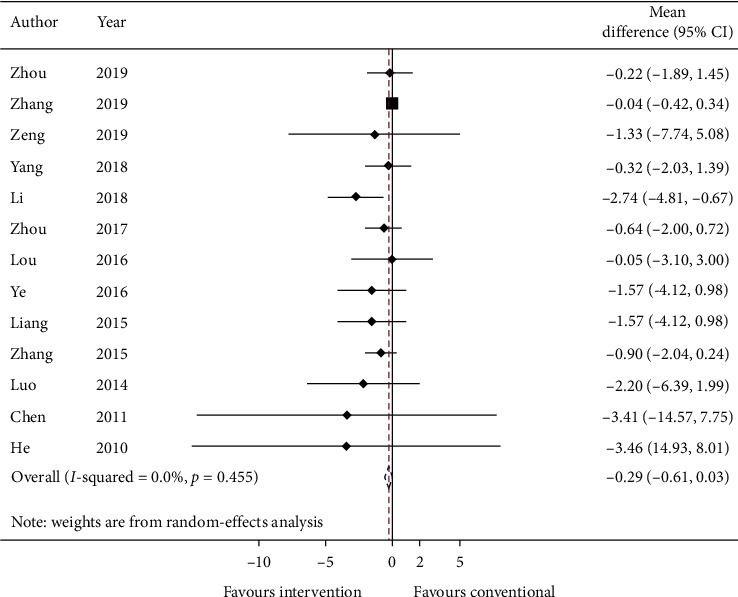
Forest plot of exacerbation rate. CI: confidence intervals.
GRADE: the overall quality of this evidence is judged to be very low, downgraded once for risk of bias, once for imprecision, and once for too few data from included studies.
3.5.3. Primary Outcome: COPD Assessment Test
Twenty studies report COPD assessment test (CAT) with mean and standard error. Data are converted to standard deviation (SD) and meta-analyzed in Figure 4. The effect estimate is −2.16 (95% CI: −3.44, −0.88; p ≤ 0.001) with I2 = 0.0% (p=0.982). Adding on CHM with conventional medicine significantly reduces the score of CAT.
Figure 4.
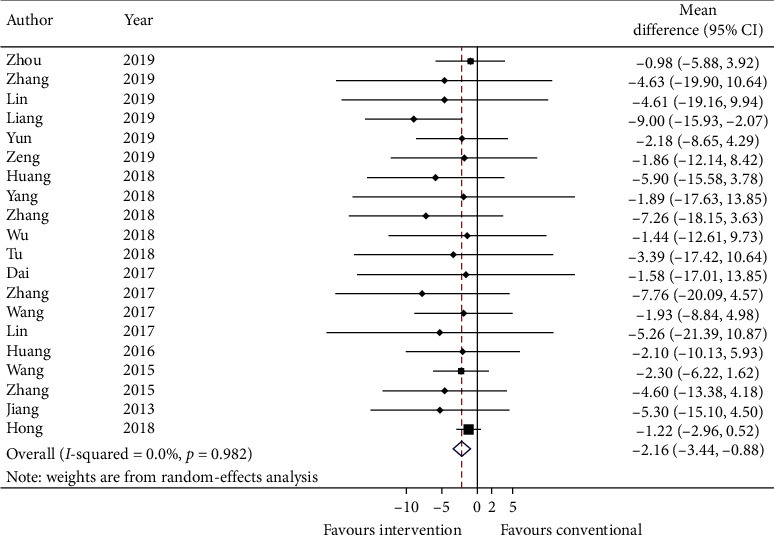
Forest plot of COPD assessment test. CI: confidence intervals.
GRADE: the overall quality of this evidence is judged to be low, downgraded once for risk of bias and once for imprecision.
3.5.4. Primary Outcome: Adverse Events of Any Cause
Eight studies report adverse events of any cause and data are summarized in Table 5. Of these, four studies have no adverse events throughout the study period [28, 66–68]. Wang reports 2 cases of nausea, Liu reports 1 case of mouth dryness, and Huang reports 3 cases of acute exacerbation with hospitalization. Hong reports 2 epigastric discomfort and 1 diarrhea in the control group and 1 pale yellow phlegm in the intervention group.
Table 5.
Adverse events of any cause.
StudyGroup (number of events)Cause
| [56] | Control (0) | — |
| Intervention (2) | Nausea | |
| [64] | Control (0) | — |
| Intervention (1) | Mouth dryness | |
| [65] | Control (2) | Acute exacerbation |
| Intervention (1) | Acute exacerbation | |
| [63] | Control (3) | 2 epigastric discomfort, 1 diarrhea |
| Intervention (1) | 1 pale yellow phlegm | |
GRADE: the overall quality of this evidence is judged to be low, downgraded once for risk of bias and once for imprecision.
3.5.5. Secondary Outcome: Quality of Life
Seventeen studies reported quality of life using different scales. Of these, nine studies used St. George's Respiratory Questionnaire (SGRQ) [69–77]. Eight studies used COPD-Quality of Life (COPD-QoL) questionnaire [54, 67, 78–83].
Standardized mean difference (SMD) and standard deviation (SD) are calculated and meta-analyzed in Figure 5. The summary estimate was −0.01 (95% CI: −0.12, 0.10; p=0.858723) with heterogeneity = 0.0% (p=1.000).
Figure 5.
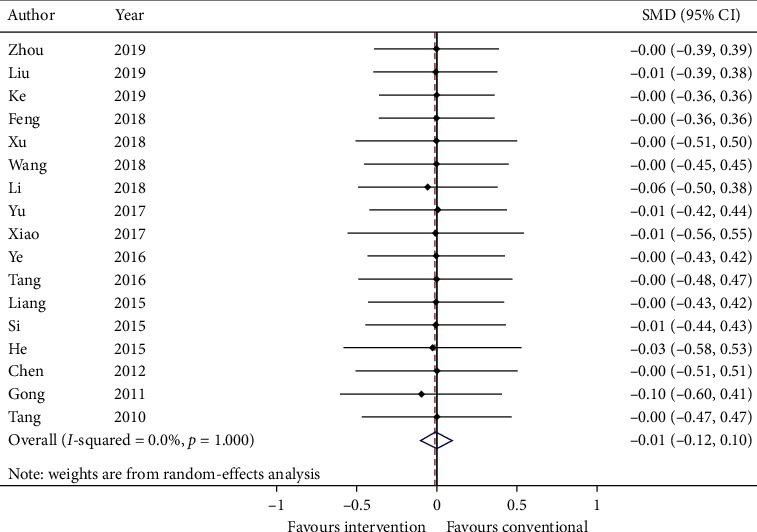
Forest plot of quality of life. CI: confidence intervals; SMD: standardized mean difference.
GRADE: the overall quality of this evidence is judged to be very low, downgraded once for risk of bias, once for inconsistency, and once for imprecision.
3.5.6. Secondary Outcome: TCM Syndrome Score and Effective Rate
Thirty-five studies reported total TCM syndrome score in mean plus standard error and sixty-two studies reported TCM effective rate as a number of events in each group. Data were converted to mean plus standard error and risk ratio. Meta-analysis results are presented in Figures 6 and 7, respectively. The total TCM syndrome score was reduced after adding CHM (effect estimate: MD: −3.96, 95% CI: −5.41, −2.51, p < 0.00001) with I2 = 0.0% (p=0.915). The effect estimate for TCM effective rate was 0.89 (95% CI: 0.86, 0.93, p < 0.00001) with heterogeneity = 0.0% (p=1.000).
Figure 6.
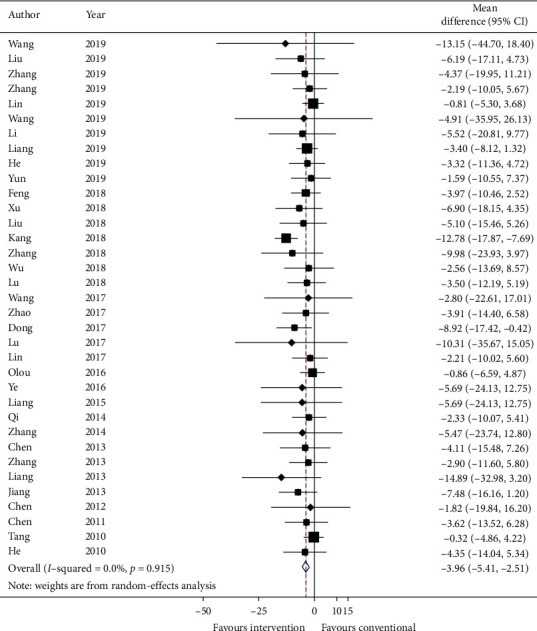
Forest plot of TCM syndrome score. CI: confidence intervals.
Figure 7.
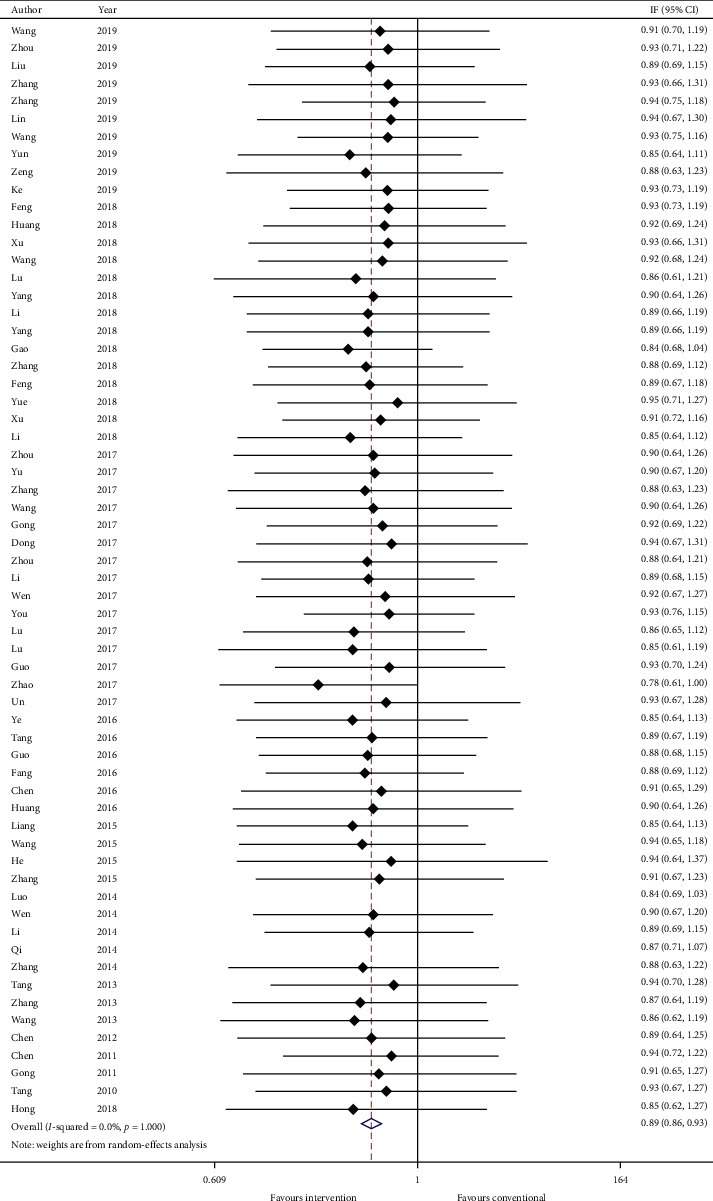
Forest plot of TCM effective rate. CI: confidence intervals.
GRADE: the overall quality of this evidence is judged to be moderate, downgraded once for risk of bias and once for imprecision.
3.5.7. Secondary Outcome: 6-Minute Walk Test
Twenty-five studies reported 6-minute walk test and data were reported as mean distance (in meters) and standard error. Data are transformed to standard deviation (SD) and meta-analyzed in Figure 8. The effect estimate was 37.81 (95% CI: 20.90, 54.73; p ≤ 0.001) favoring intervention. The heterogeneity is relatively low with I-squared = 14.6% (p=0.255).
Figure 8.
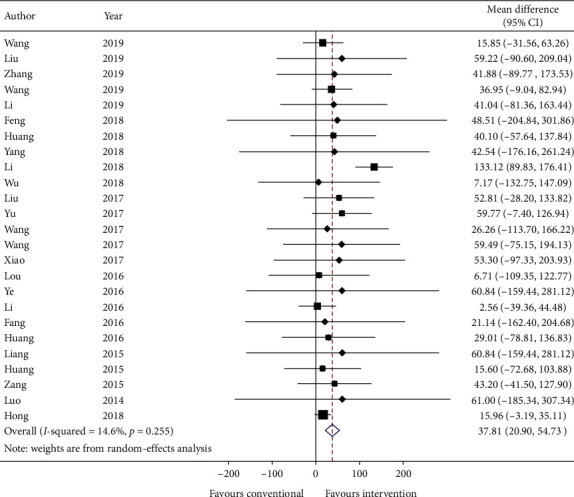
Forest plot of 6-minute walk test. CI: confidence intervals.
GRADE: the overall quality of this evidence is judged to be low, downgraded once for risk of bias and once for imprecision.
3.6. Publication Bias
Publication bias was examined using Egger's test in TCM effective rate from 62 studies. The p value is 0.011 (Figure 9), which means there was a small study effect and might influence the interpretation of the summary estimate.
Figure 9.
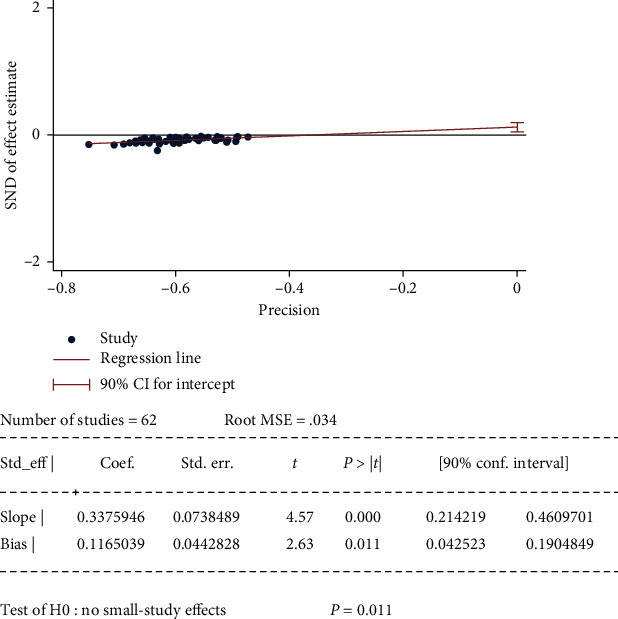
Egger's test for publication bias.
4. Discussion4.1. Summary of Evidence
This systematic review evaluates the available evidence in English and Chinese of combining Traditional Chinese medicine (including Chinese herbal medicine and acupuncture) with conventional medicine on treating stable chronic obstructive pulmonary disease patients. Although there is no high-quality evidence identified, we have low-to-moderate quality randomized controlled trials with 8291 participants to suggest that it might be beneficial to incorporate TCM into conventional treatment. This review included 100 double-blinded, randomized controlled trials (8291 participants), with one comparison arm: Chinese herbal medicine plus conventional medicine versus conventional medicine only. The overall risks of bias for these studies are low to unclear. Reasons include (1) unclear reporting of allocation concealment, (2) no detailed information on blinding of participants, personnel, and outcome assessors, and (3) lack of methods reporting how to deal with missing data.
For primary outcomes, there are 40 studies showing the addition of TCM improved the force expiratory volume in 1 s (mean change: 0.18 (L), 95% CI: 0.08, 0.28, I2: 0.0%). But there are few reports about exacerbation rate, only 13 studies were meta-analyzed and the summary effect showed no reduction in acute exacerbation (mean change: −0.29, 95% CI: −0.61, 0.03, I2: 0.0%). There are also limited data (20 studies), suggesting TCM is beneficial by reducing COPD assessment test score (mean change: −2.16, 95% CI: −3.44, −0.88, I2: 0.0%) when compared to conventional medicine only.
Only eight studies reported a total of 10 adverse events, including gastrointestinal symptoms such as nausea, mouth dryness, epigastric discomfort, diarrhea, and respiratory symptoms such as pale yellow phlegm and acute exacerbation with hospitalization. Ten studies were excluded from this review because they did not perform “intention-to-treat” analysis when facing withdrawals. Only one study reported 2 withdrawals in the TCM + CT group and 3 in the CT group, which were mostly lost during treatment. No studies reported adverse effects of either Chinese herbal medicine or acupuncture.
For secondary outcomes, there is limited evidence to show that TCM can improve patients' quality of life, with only 17 studies and different scales used. So we calculated the standardized mean difference and the effect estimate was −0.01 (95% CI: −0.12, 0.10, I2: 0.0%), which did not show any improvement. There are thirty-five studies reporting the change in TCM syndrome score. The symptoms and signs were less severe in the CHM + CT group (mean change: −3.96, 95%CI: -5.41, −2.51, I2 = 0.0%). Merely sufficient evidence (62 studies) showed that TCM was more effective combined with conventional treatment (RR: 0.89, 95% CI: 0.86, 0.93, I2 = 0.0%). The distance walked in 6 minutes was increased by 37.81 meters (95% CI: 20.90, 54.73, I2: 14.6%) in the intervention group when compared to the control group with 25 studies.
GRADE: the outcomes change in FEV1, TCM syndrome score, and effective rate were rated as moderate. This implies there is some confidence in the effect estimate, and the true effect is likely to be close to the estimate of effect, but there is a chance that it is substantially different. Future research is likely to have an important impact on the confidence in the estimate of effect and may change the estimate [40]. The outcomes COPD assessment test, adverse events of any cause, all withdrawals of study participants, and 6-minute walk test were judged to be low-quality evidence. This implies there is limited confidence in the effect estimate and the true effect may be substantially different from the estimate of effect. Future research is very likely to change the estimate of effect or impact the confidence in the estimate of effect [40]. The outcomes exacerbation rate, withdrawals due to a lack of efficacy, and quality of life were rated as very low-quality evidence. This implies there is very little confidence in the effect estimate between types of treatments and the true effect is likely to be substantially different from the estimate of effect. The results of these outcomes are very uncertain and the effect estimate is not accurate enough to recommend any use of Chinese herbal medicine [40]. In summary, the body of evidence suggests that adding Chinese herbal medicine to conventional treatment may be beneficial in stable COPD patients.
4.2. Strengths of This Review
A major strength of this review is we strictly included level 1 evidence of double-blinded RCTs only. This review does not include studies where participants have both acute exacerbated and stable chronic obstructive pulmonary disease and any complicated diseases. Previous systematic reviews report either one single herb extract or formulae, or one specific TCM syndrome-related treatment method [32, 67, 84–91]. However, this is not the usual practice of traditional Chinese medicine. In both outpatient and inpatient settings, TCM uses various formula or acupoints combination according to syndrome differentiation even in the same stable COPD population. This review identifies the add-on effect of any formation of Chinese herbal medicine with different TCM syndromes in comparison with conventional medicine only. This fills the gaps between the normal use of TCM and contemporary research in treating stable COPD patients.
This review identifies the need for high-quality double-blinded randomized controlled trials with more participants in each arm and more detailed reporting of research methods (randomization and blinding of participants and accessors).
4.3. Limitations of This Review
The key methodological limitation to this review was the language restriction, and only Chinese and English literature studies were searched. We believed there were studies written in other languages such as Korean and Japanese, where Chinese herbal medicine and acupuncture were often used. A more comprehensive review might be needed to include all languages.
A second limitation to this review was the broad inclusion criteria of herbal formulae or drugs and treatment duration. This would limit the specificity analysis of a certain herb or formulae, as we generally analyzed CHM as an adjunct treatment to conventional medicine. This might also limit the usage of CHM in clinical situation because we would need a registered Chinese medicine practitioner to diagnose with syndrome differentiation. Subgroup analyses were planned but could not be done in this review as there were too many combinations of herbal drugs and treatment durations. It was unable to investigate whether any of these variables affected treatment efficacy.
A third limitation of this review was lacking studies with sample sizes larger than or equal to 200 participants per arm.
5. Conclusions5.1. Implications for Clinical Practice
Traditional Chinese medicine has been used to treat COPD-related symptoms over decades, yet its effectiveness and safety remain uncertain. Previous clinical studies reported either one single Chinese herbal formulae or one specific Chinese medicine treatment method. Considering there are numerous formulae or combinations of herbs that might be beneficial to stable COPD patients, it is difficult to use current evidence to guide the use of TCM in addition to conventional medicine.
Although we saw some statistical significance in several outcome parameters, it did not mean that there are real treatment effects clinically. Our data suggested that TCM combined with conventional treatment was beneficial in FEV1, COPD assessment test, 6-minute walk distance, and TCM syndrome statistically. Clinicians may consider incorporating TCM into the mainstream medical system with reference to their own clinical experience.
5.2. Implications for Research
High-quality randomized controlled clinical trials or pragmatic trials are needed. In order to provide real information for Chinese medicine practitioners, TCM theories and diagnoses must be taken into account when designing clinical research protocols and conducting trials.
More advanced analyses, like individual participant data and network meta-analysis, can be applied to provide more information on different combinations of herbal drugs and/or acupuncture and generate personalized evidence with reference to TCM theories.
AbbreviationsBOLD:
Burden of Obstructive Lung Diseases
CATTM:
COPD assessment test
CBM:
Chinese Biomedical Database
CENTRAL:
Cochrane Central Register of Controlled Trials
CHM:
Chinese herbal medicine
CI:
Confidence interval
CNKI:
Chinese National Knowledge Infrastructure
COPD:
Chronic obstructive pulmonary disease
CT:
Conventional treatment
FEV1:
Forced expiratory volume in first second
FVC:
Forced vital capacity
GRADE:
Grading of Recommendations, Assessment, Development, and Evaluation system
GOLD:
Global Initiative for Chronic Obstructive Lung Disease
ICTRP:
The World Health Organization International Clinical Trials Registry Platform
ITT:
Intention-to-treat
LABA:
Long-acting beta2-agonist
LAMA:
Long-acting anticholinergic
MD:
Mean difference
MeSH:
Medical subject headings
mMRC:
Modified British Medical Research Council
mRCT:
metaRegister of controlled trials
PRISMA-P:
Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols
PROSPERO:
Prospective register of systematic review
QoL:
Quality of life
RCT:
Randomized controlled trial
SABA:
Short-acting beta2-agonist
SABD:
Short-acting bronchodilator
SAMA:
Short-acting anticholinergic
SD:
Standard deviation
SE:
Standard error
SGRQ:
St. George's Respiratory Questionnaire
SMD:
Standardized mean difference
TCM:
Traditional Chinese medicine
VIP:
Chinese Scientific and Technological Periodical Database
WHO:
World Health Organization.
Data Availability
The clinical data used to support the findings of this study are included within the supplementary information files.
Disclosure
This study is part of the MSc dissertation of the first author (K. H. Chan). K. H. Chan is the lead investigator of this project.
Conflicts of Interest
The authors declare that there are no conflicts of interest.
Authors' Contributions
K. H. designed the research protocol, screened databases, extracted data, performed meta-analysis, and wrote the dissertation. Y. Y. S. acted as a second reviewer to screen titles and abstracts for eligibility and extracted data from included studies.
Supplementary Materials
Supplementary Materials
Appendix 1: research protocol. Appendix 2: PRISMA statement reporting standards checklist. Appendix 3: data extraction form. Appendix 4: Cochrane risk of bias tool for randomized controlled trials. Appendix 5: GRADE guidelines. Appendix 6: description of Chinese medicine. Appendix 7: characteristics of excluded studies table.
Click here for additional data file. (113.2KB, docx)
References
- 1.Lozano R., Naghavi M., Foreman K., et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;280(9859):2095–2128. doi: 10.1016/S0140-6736(12)61728-0. [DOI] [PMC free article] [PubMed] [Google Scholar]