최근 고품질 증거(주로 2020~2026년 무작위 대조 시험(RCT) 및 메타분석)에 기반한
COPD 환자의 보조 요법으로 유망한 영양제(nutraceuticals)를 정리한 내용입니다.
주요 개선 영역은
폐기능(FEV1, FEV1/FVC), 운동 능력(6MWD), 호흡곤란(mMRC 척도), 삶의 질(CAT 또는 SGRQ 점수) 등입니다.
Top Nutraceuticals with Strongest Evidence
- Nanocurcumin (Curcumin formulations): Highly effective for improving lung function. In a 2025 NMA of 37 RCTs (n=1975), nanocurcumin ranked highest for FEV1 % predicted (MD=13, 95% CrI 5.4–21) and FEV1/FVC (MD=12, 95% CrI 5.5–18) vs. placebo. A similar 2025 NMA of 48 RCTs (n=2481) confirmed nanocurcumin's superiority for FEV1 (MD=13, 95% CI 7.7–18). A 2025 meta-analysis of polyphenols (8 RCTs, n=894) showed curcumin (200–500 mg/day) reduced systolic blood pressure and improved FEV1 (SMD=-0.82, 95% CI -1.53 to -0.11). Typical dose: 80–500 mg/day; safe with no major adverse effects reported.
- Vitamin D: Improves quality of life and lung function in deficient patients. A 2024 meta-analysis of RCTs found vitamin D (alone or in compounds) enhanced FEV1 in COPD. The 2025 NMA noted reductions in CAT scores (MD=-2.5, 95% CrI -3.1 to -1.9). A 2022 meta-analysis (up to 2022 RCTs) showed benefits for asthma/COPD control, especially in deficient individuals. Dose: Often 1000–4000 IU/day; check serum levels first.
- Coenzyme Q10 + Creatine: Best for exercise endurance. The 2025 NMA ranked it highest for 6MWD (MD=63, 95% CrI 36–90). Confirmed in another 2025 NMA (MD=63, 95% CI 36–90). Creatine alone also reduced SGRQ scores (MD=-9.6, 95% CrI -17 to -2.8). Dose: CoQ10 100–200 mg + creatine 5–10 g/day.
- Melatonin: Enhances exercise, reduces dyspnea, and improves quality of life. In the 2025 NMA, it improved 6MWD (MD=46, 95% CrI 1.3–92), mMRC (-0.90, 95% CrI -1.6 to -0.21), and CAT (-8, 95% CrI -12 to -4.2; highest SUCRA 99.6%). Another 2025 NMA noted mMRC improvements (MD=-0.9, 95% CI -1.6 to -0.21). Dose: 3–10 mg/day.
- Yam-Epimedium (Traditional Chinese herbal mix): Top for dyspnea and quality of life. The 2025 NMA ranked it highest for mMRC (-1.3, 95% CrI -1.9 to -0.67; SUCRA 97.4%) and SGRQ (-24, 95% CrI -34 to -13; SUCRA 99.1%). Limited to specific formulations; consult for safety.
- Probiotics: Boosts lung function. The 2025 NMA showed FEV1 improvements (MD=7.1, 95% CrI 5.2–9.1). Another 2025 NMA confirmed (MD=7.1, 95% CI 5.2–9.1). Often multi-strain; dose: 10^9–10^10 CFU/day.
- Omega-3 Fatty Acids (e.g., Fish Oil): Mixed but positive for some outcomes. A 2025 systematic review of RCTs noted benefits from ω-3 in nutritional therapy for COPD. Reviews (2023–2024) link it to reduced inflammation. Dose: 1–2 g/day EPA+DHA.
- Resveratrol: Anti-inflammatory effects. The 2025 polyphenol meta-analysis (RCTs) showed reductions in TNF-α and IL-8 (p=0.003). Protective in reviews for COPD progression via epigenetics. Dose: 100–500 mg/day.
- 나노커큐민(Nanocurcumin, 커큐민 제제): 폐기능 개선에 가장 효과적. 2025년 NMA(37개 RCT, n=1975)에서 FEV1 % predicted(MD=13, 95% CrI 5.4–21)와 FEV1/FVC(MD=12, 95% CrI 5.5–18)에서 최고 순위. 또 다른 2025년 NMA(48개 RCT, n=2481)에서도 FEV1 개선(MD=13, 95% CI 7.7–18)에서 우수. 폴리페놀 관련 연구에서도 긍정적 효과 확인. 용량: 80~500 mg/일, 부작용 거의 없음.
- 비타민 D: 결핍 환자에서 삶의 질과 폐기능 개선. 2024년 메타분석에서 비타민 D가 COPD의 FEV1을 향상시켰고, 2025년 NMA에서 CAT 점수 감소(MD=-2.5). 결핍 환자에게 특히 효과적. 용량: 1000~4000 IU/일, 혈중 농도 확인 필수.
- 코엔자임 Q10 + 크레아틴: 운동 능력 향상에 최고. 2025년 NMA에서 6MWD 63m 증가(MD=63, 95% CrI 36–90)로 1위. 크레아틴만으로도 SGRQ 점수 감소(MD=-9.6). 용량: CoQ10 100~200 mg + 크레아틴 5~10 g/일.
- 멜라토닌: 운동 능력, 호흡곤란, 삶의 질 개선. 2025년 NMA에서 6MWD(MD=46m), mMRC(-0.90), CAT(-8점, SUCRA 99.6%)에서 높은 순위. 용량: 3~10 mg/일.
- 산약-음양곽(Yam-Epimedium, 한약 복합제): 호흡곤란과 삶의 질에서 최고. 2025년 NMA에서 mMRC(-1.3, SUCRA 97.4%), SGRQ(-24점, SUCRA 99.1%). 특정 제제에 한정, 안전성 상담 필요.
- 프로바이오틱스: 폐기능 향상. 2025년 NMA에서 FEV1(MD=7.1, 95% CrI 5.2–9.1) 개선. 다균주 제품 권장. 용량: 10^9~10^10 CFU/일.
- 오메가-3 지방산(생선 기름): 염증 감소 등 일부 긍정적 효과. 2025년 체계적 고찰에서 COPD 영양 요법으로 유용. 용량: EPA+DHA 1~2 g/일.
- 레스베라트롤: 항염증 효과. 2025년 폴리페놀 메타분석에서 TNF-α, IL-8 감소. 용량: 100~500 mg/일.
Additional Promising Ones
- Whey Proteins/Essential Amino Acids (EAA): Improve exercise (6MWD MD=11) and CAT; EAA reduces SGRQ. A 2024 meta-analysis (29 RCTs, n=1625) supported for sarcopenia in COPD (weight gain, 6MWD).
- Nitrate (e.g., Beetroot Juice): Enhances 6MWD (MD=30).
- Butyrate: FEV1 gains (MD=2.7).
- Salidroside: Reduces inflammation (TNF-α) and CAT scores (p<0.01).
- 유청단백/필수아미노산(EAA): 운동 능력(6MWD +11m) 및 CAT 개선, 근감소증 환자에서 체중·운동 능력 향상(2024년 29개 RCT 메타분석).
- 질산염(비트루트 주스): 6MWD +30m.
- 부티레이트: FEV1 +2.7%.
- 살리드로사이드: 염증(TNF-α) 및 CAT 점수 감소.

| 연구 목적 만성 폐쇄성 폐질환(COPD) 환자에서 다양한 영양 보충제(nutritional supplements)의 효과를 네트워크 메타분석(network meta-analysis)을 통해 체계적으로 비교 평가하여, 임상 의사결정에 근거를 제공하는 것. 방법
|
BMC Pulm Med
. 2025 May 8;25:220. doi: 10.1186/s12890-025-03667-0
The effects of various nutritional supplements in patients with chronic obstructive pulmonary disease: a network meta-analysis
Jia Zeng 1,#, Jing Cheng 1,#, Liangdong Zhu 2,✉, Sufang Tang 1
- Author information
- Article notes
- Copyright and License information
PMCID: PMC12060334 PMID: 40340688
AbstractObjective
To evaluate the effectiveness of various nutritional supplements as interventions for patients with Chronic Obstructive Pulmonary Disease (COPD) using network meta-analysis.
Methods
We searched PubMed, Embase, Cochrane Library, and Web of Science databases for randomized controlled trials on nutritional supplements for COPD patients, with the search updated to March 24, 2025. The risk of bias for each included study was assessed using appropriate tools, and the certainty of evidence was evaluated according to the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach. Data synthesis was performed using R software, employing network meta-analysis methods to compare the relative efficacy of different nutritional interventions.
Results
Thirty-seven studies involving 1975 COPD patients and 21 different nutritional supplements were analyzed. Butyrate [MD = 2.7, 95% CrI (1.0, 4.4)], Nanocurcumin [MD = 13, 95% CrI (5.4, 21)], and probiotics [MD = 7.1, 95% CrI (5.2, 9.1)] significantly improved Forced Expiratory Volume in One Second (FEV1). Nanocurcumin also slowed the decline in the ratio of Forced Expiratory Volume in One Second to Forced Vital Capacity (FEV1/FVC) [MD = 12, 95% CrI (5.5, 18)]. For exercise endurance, coenzyme Q10 combined with creatine [MD = 63, 95% CrI (36, 90)], Melatonin (MLT) [MD = 46, 95% CrI (1.3, 92)], Nitrate [MD = 30, 95% CrI (19, 41)], and whey proteins [MD = 11, 95% CrI (9.2, 13)] significantly improved 6-minute walk distance (6MWD). Regarding dyspnea reduction, MLT [MD = -0.90, 95% CrI (-1.6, -0.21)] and Yam-Epimedium [MD = -1.3, 95% CrI (-1.9, -0.67)] significantly lowered modified Medical Research Council (mMRC) scores. In terms of quality of life, MLT [MD = -8, 95% CrI (-12, -4.2)], Vitamin D (VD) [MD = -2.5, 95% CrI (-3.1, -1.9)], and whey proteins [MD = -0.70, 95% CrI (-0.99, -0.41)] reduced COPD Assessment Test (CAT) scores. Additionally, AKL1 [MD = -9.2, 95% CrI (-18, -0.41)], creatine [MD = -9.6, 95% CrI (-17, -2.8)], and Yam-Epimedium [MD = -24, 95% CrI (-34, -13)] lowered St. George’s Respiratory Questionnaire (SGRQ) scores. The Surface Under the Cumulative Ranking Curve (SUCRA) analysis showed Nanocurcumin ranked highest for improving FEV1/FVC, coenzyme Q10 combined with creatine for 6MWD, Yam-Epimedium for reducing dyspnea and SGRQ, and MLT for lowering CAT scores.
Conclusion
This analysis indicates that various nutritional supplements, including Nanocurcumin, butyrate, probiotics, coenzyme Q10 combined with creatine, MLT, nitrate, whey proteins, VD, AKL1, creatine, and Yam-Epimedium, can potentially benefit COPD patients, demonstrating differing levels of effectiveness. Specifically, Nanocurcumin may be the best choice for improving lung function; coenzyme Q10 combined with creatine may be the most effective for enhancing exercise endurance; Additionally, Yam-Epimedium is the most likely supplement to reduce the symptoms of dyspnea in COPD patients. Yam-Epimedium and MLT may be the most effective for improving quality of life and overall health status.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12890-025-03667-0.
Keywords: Chronic obstructive pulmonary disease, Dietary supplement, Network meta-analysis
Introduction
Chronic obstructive pulmonary disease (COPD) represents a heterogeneous group of lung diseases characterized by obstructive airway pathology [1, 2], including chronic bronchitis and bronchiectasis, as well as alveolar abnormalities, such as emphysema [3, 4]. These conditions are manifested by persistent respiratory symptoms, such as dyspnea, cough, and sputum production, which result in progressive and irreversible airflow limitation [5]. Epidemiological studies have indicated that, in 2019, approximately 210 million individuals globally were affected by COPD, with more than 3 million deaths attributed to the disease [6–8]. It is projected that COPD will become one of the leading causes of mortality worldwide, with an estimated 600 million cases and 6 million deaths by 2050 [9]. The disease burden is particularly pronounced in low- and middle-income countries, where the prevalence significantly exceeds that observed in high-income regions [10, 11].
COPD is not solely a respiratory disease but also has widespread systemic effects. Muscle dysfunction, malnutrition, and systemic inflammation are common sequelae in COPD patients [12, 13], contributing to disease progression, reduced exercise capacity, and a diminished quality of life [12–14]. While pharmacological therapy remains the cornerstone of COPD management, increasing insight into the underlying pathophysiological mechanisms has prompted a growing focus on the potential efficacy of nutritional supplements as adjunctive treatments [15–17]. Nutritional support has thus emerged as a critical component of COPD management.
Several clinical trials have investigated the therapeutic effects of various dietary supplements in COPD patients. For instance, oral nitrate supplementation has been shown to improve cardiovascular risk markers [18]; multistrain probiotics have been demonstrated to enhance handgrip strength and functional capacity [19]; vitamin D (VD) supplementation, particularly in deficient individuals, has been associated with improvements in lung function and quality of life [20]; omega-3 fatty acids supplementation has been shown to enhance endothelial function [21]; and supplementation with coenzyme Q10 and creatine has been found to improve functional performance [22].
Building on these findings, systematic reviews and meta-analyses have corroborated similar results to those observed in our study [23–25], with current evidence suggesting that dietary interventions can serve as an effective adjunct to conventional COPD treatments. However, the comparative effects of various nutritional supplements in COPD patients have not been comprehensively assessed. Network meta-analysis (NMA) is a robust statistical technique that enables the comparison of multiple interventions, even in the absence of direct head-to-head comparisons in clinical trials [26]. Therefore, this study aims to utilize network meta-analysis to systematically evaluate the efficacy of diverse dietary supplements in COPD patients, providing a more empirically grounded basis for clinical decision-making.
Materials and methodsStudy registration
This systematic evaluation has been registered with PROSPERO (registration number: CRD42024531862) and adheres to the Preferred Reporting Items for Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [27, 28] (S1 File).
Inclusion and exclusion criteria
Inclusion Criteria: (1) Participants diagnosed with COPD, defined as a Forced Expiratory Volume in One Second to Forced Vital Capacity (FEV1/FVC) ratio of less than 70% of the predicted value, aged 18 years or older; (2) Interventions involving dietary supplements; (3) Control groups receiving nonspecific interventions or standard diets, referred to as the placebo; (4) Outcome measures including FEV1 as a percentage of predicted values, FEV1/FVC ratio, 6-minute walk distance (6MWD), modified Medical Research Council (mMRC) Dyspnea Scale, COPD Assessment Test (CAT), and St. George’s Respiratory Questionnaire (SGRQ); (5) Study design being a randomized controlled trial (RCT).Exclusion Criteria: (1) Studies combining other organic diseases, such as diabetes mellitus, renal disease, lung cancer; (2) Duplicate literature; (3) Animal studies, conference abstracts; (4) Inaccessible full text or no data available.
Literature search
A systematic search was performed in PubMed, Embase, Cochrane Library, and Web of Science for randomized controlled trials evaluating nutritional supplements in COPD patients until March 24, 2025. The search terms included “Pulmonary Disease, Chronic Obstructive”, “Chronic Obstructive Lung Disease”, “Chronic Obstructive Pulmonary Diseases”, “COPD”, “Dietary Supplements”, “Dietary Supplementation”, “Vitamin”, “Minerals”, “Vitamin C”, “Vitamin D”, “Vitamin E”, “Creatine,” “Coenzyme Q10”, “β-Alanine”, “Nitrates”, and “Beet Protein”, which were searched in various combinations. The detailed search strategy is provided in Table S1 (S2 File).
Data extraction
Data were extracted separately by two researchers after a rigorous screening of the literature based on predetermined inclusion and exclusion criteria. In case of disagreement, consensus was reached after discussion or a third party was consulted to reach a consensus. Information extracted from the included studies included the following key elements: author, year of publication, country, sample size, gender, age, intervention, outcome.
Quality assessment
Two researchers used the Cochrane Risk of Bias Tool for randomized trials (RoB version 2.0) [29] to assessed the risk of bias in the included studies. The assessment categories included low risk of bias, unclear risk of bias and high risk of bias. The assessment covered seven key areas including: generation of randomization procedures, concealment of allocation, blinding of investigators and participants, blinding of outcome assessors, completeness of outcome data, selective reporting of study results, and other possible bias sources. Each study was independently assessed against these criteria. Additionally, the quality of evidence for each outcome was assessed using the GRADE(Grading of Recommendations Assessment, Development and Evaluation) framework [30].
Statistical analysis
Bayesian network meta-analysis was conducted using R software version 4.3.2 (R Foundation for Statistical Computing). The Markov chain Monte Carlo method was used to obtain the best combined estimates and probabilities for the effectiveness of nutritional supplements. To assess heterogeneity, we calculated I² statistics, with I² > 50% indicating substantial heterogeneity. When substantial heterogeneity was present (I² > 50%), random-effects models were used to analyze the associations between nutritional supplements and outcomes; otherwise, fixed-effects models were applied [31]. Model convergence was assessed using trajectory and Brooks-Gelman-Rubin plots, while continuous outcomes were reported as mean differences (MD) with their 95% credible interval (CrI). The Surface Under the Cumulative Ranking Curve (SUCRA) was calculated to estimate the probability of optimal intervention. Network and funnel plots were generated in Stara version 16.0 2 using specific commands. In network plots, each circle corresponds to a nutritional supplement and edges represent existing comparisons. The size of the circles is proportional to the number of included patients. Cumulative probability plots were created using the ggplot2 package.
ResultsData screening process and results
A total of 5954 records were initially identified through database searching. After removing 2447 duplicates, and excluding 1341 records flagged as ineligible by automation tools as well as 1018 records excluded for other reasons, 1146 records remained for title and abstract screening. Following this step, 1063 records were excluded based on title and abstract review, resulting in 83 full-text articles assessed for eligibility. Ultimately, 37 studies met the inclusion criteria and were included in the final review. The study selection process is illustrated in the PRISMA flow diagram (Fig. 1).
Fig. 1.
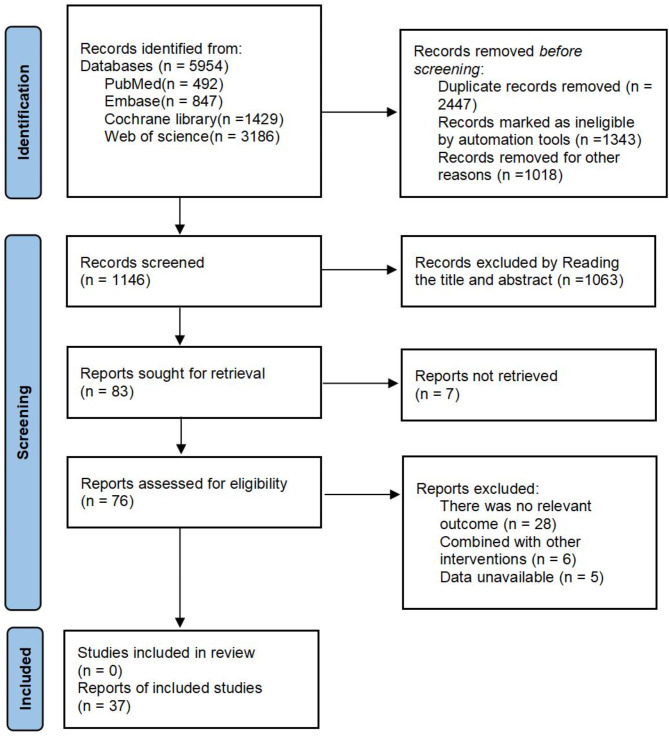
PRISMA flow diagram
Study characteristics
A total of 37 articles were included, all of which were parallel-group controlled studies, with a total population of 1975 patients with COPD, 998 in the experimental group and 977 in the control group, involving 21 nutritional supplements, and the specific characteristics of the literature are shown in Table 1.
Table 1.
Characteristics of included studies
StudyCountrySample sizeGender (M/F)Mean ageMean FEV1% predMean FEV1/FVCGOLD stageInterventionDuration of interventionOutcome
| Ahnfeldt-Mollerup et al. 2015 [32] | Denmark | EG: 28 CG: 25 | 23/30 | EG: 67 CG: 70 | / | / | I-IV | EG: complex (134.8 kcal, 9.3 g protein, 14.6 g CHO, 4.2 g fat) + exercise CG: placebo + exercise | 9 weeks | SGRQ |
| Aldhahir et al. 2021 [33] | UK | EG: 22 CG: 22 | 28/16 | EG: 75 CG: 70 | EG: 59 CG: 52 | EG: 53 CG: 54 | / | EG: complex (300 kcal, 24% protein, 41% CHO, 35% fat) + exercise CG: placebo + exercise | 6 weeks | CAT |
| Khan et al. 2016 [34] | India | EG: 30 CG: 30 | 54/6 | EG: 55.03 CG: 53.33 | EG: 52.87 CG: 53.31 | EG: 69.44 CG: 67.62 | I-IV | EG: complex (90 kcal, 45% protein, 55%CHO) CG: placebo | 12 weeks | 6MWD、FEV1/FVC FEV1 (% of predicted) |
| Zong et al. 2023 [35] | China | EG: 27 CG: 29 | 28/28 | EG: 80.5 CG: 81.1 | EG: 71.7 CG: 67.2 | EG: 63.3 CG: 61.8 | II-III | EG: whey proteins (10 g/day) CG: placebo | 12 weeks | mMRC、CAT、6MWD、FEV1 (% of predicted) |
| Gurgun et al. 2013 [36] | Turkey | EG: 15 CG: 16 | 31/0 | EG: 66.8 CG: 67.8 | EG: 41.9 CG: 41.9 | EG: 53.4 CG: 43.9 | III-IV | EG: complex (16.7% protein, 53.3% CHO, 30% fat) + exercise CG: placebo + exercise | 8 weeks | mMRC、6MWD、SGRQ |
| Zarei et al. 2024 [37] | Iran | EG: 30 CG: 30 | 29/31 | EG: 60.83 CG: 59.97 | EG: 36.77 CG: 44.87 | EG: 60.1 CG: 64.52 | III-IV | EG: Nanocurcumin (80 mg/day) CG: placebo | 3 months | FEV1/FVC、 FEV1 (% of predicted) |
| Cerda et al. 2006 [38] | Spain | EG: 15 CG: 15 | 30/0 | EG: 60 CG: 63.4 | EG:46 CG: 35.1 | EG: 46.59 CG: 43.24 | II-IV | EG: pomegranate juice (400 ml/day) CG: placebo | 5 weeks | FEV1/FVC |
| Gouzi et al. 2019 [39] | France | EG:31 CG:26 | 28/29 | EG: 62.4 CG: 61.1 | EG: 57 CG: 62 | EG: 41 CG: 43 | / | EG: VE + VC (VE 30 mg/day, VC 180 mg/day) + exercise CG: placebo + exercise | 28 days | 6MWD |
| Alasmari et al. 2024 [19] | UK | EG: 36 CG: 34 | 48/22 | EG: 62.5 CG: 64.5 | EG: 45.2 CG: 39.6 | / | / | EG: nitrate (400 mg/day) CG: placebo | 3 months | CAT、6MWD |
| Beers et al. 2020 [40] | Netherland | EG: 42 CG: 39 | 41/40 | EG: 62.8 CG: 62.2 | EG: 57 CG: 53 | EG: 44.4 CG: 41.6 | / | EG: complex (protein, carbohydrates, fat and micronutrients, and was enriched with leucine, omega-3, and vitamin D ) CG: placebo | 4 months | SGRQ |
| Benedetto et al. 2018 [23] | Italy | EG: 45 CG: 45 | 68/22 | EG: 73 CG: 73 | EG: 52 CG: 57 | / | / | EG: 160 mg Coenzyme Q10+ 170 mg creatine CG: placebo | 2 months | 6MWD、mMRC、FEV1 (% of predicted) |
| Behnia et al. 2018 [41] | USA | EG:12 CG:13 | 13/12 | EG: 67 CG: 68 | / | EG: 57 CG: 61 | I-IV | EG: nitrate (70 ml of beetroot juice plus 180 ml of black currant juice) CG: placebo (70 ml of water and 180 ml of black currant juice) | 8 weeks | SGRQ、FEV1/FVC |
| Bool et al. 2017 [42] | Netherland | EG: 42 CG: 39 | 41/40 | EG: 62.8 CG: 62.2 | EG: 57 CG:53 | EG: 44.4 CG: 41.6 | / | EG: complex (187.5 kcal, 20% protein, 60% CHO, 20% fat and was enriched with leucine, Omega-3, and vitamin D) CG: placebo | 4 months | 6MWD |
| Brandt et al. 2022 [43] | Belgium | EG: 21 CG: 19 | 30/10 | EG: 66 CG: 65 | EG: 55.2 CG: 55.6 | EG: 46.1 CG: 50.4 | I-IV | EG: beta alanine (3.2 g/day ) CG: placebo | 12 weeks | 6MWD |
| Brockwell et al. 2014 [44] | UK | EG: 20 CG: 13 | 25/8 | EG: 66.9 CG: 67.3 | EG: 57.9 CG: 57.3 | / | / | EG: AKL1 (comprising standardized extracts of Picrorhiza kurroa, Ginkgo biloba, and Zingiber officinale) CG: placebo | 10 weeks | SGRQ、6MWD、mMRC、 FEV1 (% of predicted) |
| Faager et al. 2006 [45] | Solna | EG: 10 CG: 13 | 10/13 | EG: 66 CG: 66 | EG: 44 CG: 41.9 | / | / | EG: creatine (0.3 g/kg / body weight/day for one week, 0.07 g/kg / body weight/dayfor 7 weeks) + exercise CG: placebo + exercise | 8 weeks | SGRQ、 FEV1 (% of predicted) |
| Foumani et al. 2019 [21] | Iran | EG: 32 CG: 31 | 60/3 | EG: 67.9 CG: 68.4 | EG: 58.93 CG: 58.18 | EG: 57.74 CG: 58.9 | I-IV | EG: VD (50,000 IU ) CG: placebo (gelatin) | 4 months | CAT、FEV/FVC、FEV1 (% of predicted) |
| Fuld et al. 2005 [46] | UK | EG: 18 CG: 20 | 23/15 | EG: 61.7 CG: 63.7 | EG: 45.4 CG:45.4 | EG: 35.9 CG:40.9 | / | EG: creatine (5.7 g, three times daily ) + exercise CG: placebo + exercise | 2 weeks | SGRQ |
| Fulton et al. 2017 [47] | Australia | EG: 6 CG: 6 | 7/5 | EG: 68.5 CG: 70.5 | EG: 72 CG: 50 | EG: 59 CG: 46 | / | EG: fish oil (300 mg EPA + 200 mg DHA /day) CG: placebo (corn oil) | 16 weeks | FEV1/FVC、6MWD、FEV1 (% of predicted) |
| Ingadottir et al. 2019 [48] | Iceland | EG: 19 CG: 15 | 10/24 | EG: 72 CG: 73 | EG: 37.2 CG: 37.3 | / | / | EG: complex (300 kcal, 12 g Protein) CG: placebo (snacks ) | 12 months | SGRQ |
| Karim et al. 2022 [20] | United Arab Emirates | EG: 51 CG: 53 | / | EG: 66.9 CG: 68.3 | EG: 59.88 CG: 63.76 | / | / | EG: probiotic (one capsule a day, Each capsule contains 112 billion live bacteria) CG: placebo | 16 weeks | FEV1 (% of predicted) |
| Kim et al. 2021 [22] | USA | EG: 20 CG: 20 | 22/18 | EG: 67.5 CG: 62.2 | EG: 45.2 CG: 42.6 | EG: 44 CG: 40 | II-IV | EG: fish oil (650 mg EPA + 182 mg DHA /day) CG: placebo (corn oil) | 6 months | SGRQ、FEV1/FVC、6MWD、 FEV1 (% of predicted) |
| Lu et al. 2018 [49] | Taiwan | EG: 13 CG: 14 | / | EG: 71 CG: 72 | EG: 65.92 CG: 67.50 | EG: 52.85 CG: 51.50 | / | EG: OPC (50 mg/day) CG: placebo | 3 weeks | FEV1/FVC、 FEV1 (% of predicted) |
| Martínez et al. 2017 [50] | Peru | EG: 38 CG: 49 | 62/25 | EG: 52.18 CG: 35 | EG: 68.2 CG: 76.1 | / | I-III | EG: complex (258 kcal, 9.4 g protein, 4.2 g DHA, 3 g Prebiotics, 25–30% vitaminas and minerales) CG: placebo | 3 months | 6MWD、SGRQ |
| Marinari et al. 2013 [51] | Italy | EG: 30 CG: 25 | / | EG: 73.2 CG: 73.9 | / | / | / | EG: 320 mg Coenzyme Q10 + Creatine 340 mg CG: placebo | 2 months | 6MWD、mMRC |
| Negro et al. 2010 [52] | Italy | EG: 16 CG: 16 | 25/7 | EG: 75 CG: 75 | / | EG: 39 CG: 38 | / | EG: EAA (4 g/day) CG: placebo | 3 months | SGRQ |
| Negro et al. 2012 [53] | Italy | EG: 44 CG: 44 | 61/27 | EG: 75 CG: 73 | / | EG: 39.49 CG: 37.7 | / | EG: EAA (4 g/day) CG: placebo | 3 months | FEV1/FVC、SGRQ |
| Ogasawara et al. 2018 [54] | Japan | EG: 24 CG: 21 | 9/36 | EG: 77.4 CG: 79.1 | EG: 64.2 CG: 68.2 | / | I-IV | EG: EPA (300 mg/day) CG: placebo | 12 weeks | mMRC、CAT |
| Pavitt et al. 2020 [55] | UK | EG: 57 CG: 65 | 53/69 | EG: 70 CG: 68 | EG: 53 CG: 48 | / | II-III | EG: nitrate (0.8 g /day) CG: placebo | 8 weeks | CAT、mMRC |
| Qaisar et al. 2024 [56] | United Arab Emirates | EG: 52 CG: 59 | / | EG: 67.1 CG: 65.5 | EG:60.1 CG:61.9 | / | I-IV | EG: butyrate (300 mg, capsules, take one capsule a day) CG: placebo | 12 weeks | FEV1 (% of predicted) |
| Rafiq et al. 2017 [57] | Netherland | EG: 24 CG: 26 | 26/24 | EG: 64 CG: 61 | EG: 58.46 CG: 58.95 | EG: 48.76 CG: 48.46 | I-IV | EG: VD (1,200 IU colecalciferol ) CG: placebo | 6 months | FEV1/FVC、6MWD、FEV1 (% of predicted) |
| Sugawara et al. 2010 [58] | Japan | EG: 17 CG: 15 | / | EG: 77.3 CG: 78.2 | EG: 53.3 CG: 58.1 | EG: 42.8 CG: 45.0 | / | EG: complex (400Kcal, 60% CHO, 25% fat, 15% protein) CG: placebo | 12 weeks | 6MWD |
| Sugawara et al. 2012 [59] | Japan | EG: 17 CG: 14 | 29/2 | EG: 77.4 CG: 77.1 | EG: 43.4 CG: 48.6 | EG: 39.8 CG: 39 | / | EG: whey proteins + exercise CG: placebo + exercise | 12 weeks | 6MWD、mMRC |
| Viana et al. 2023 [60] | Brazil | EG: 18 CG: 21 | 24/15 | EG:67 CG:66.2 | EG: 41.3 CG: 43.9 | NR | / | EG: MLT (3 mg /day) + exercise CG: placebo + exercise | 12 weeks | 6MWD、mMRC、CAT |
| Vermeeren et al. 2004 [61] | Netherland | EG: 23 CG: 24 | 32/15 | EG:66 CG: 65 | EG: 32 CG: 34 | EG: 48 CG: 47 | I-IV | EG: complex (1046 kJ, 20%protein, 60%CHO, 20% fat ) CG: placebo | 6 days | FEV1(% of predicted) |
| Wu et al. 2007 [62] | Taiwan | EG1: 9 EG1: 9 EG1: 9 CG: 8 | 27/8 | EG1: 71 EG2: 72 EG3: 68 CG: 65.1 | EG1: 54 EG2: 51 EG3: 51 CG: 51 | EG1: 59 EG2: 52 EG3: 50 CG: 52 | / | EG1: VE400 (vitamin E 400 mg/day) EG2: VE200 (vitamin E 200 mg/day) EG3: VC250 (vitamin C 250 mg/day) CG: placebo | 12 weeks | FEV1 (% of predicted) 、FEV1/FVC |
| Zhao et al. 2012 [63] | China | EG: 26 CG: 23 | 46/3 | EG: 80.1 CG: 80.8 | EG: 39.7 CG: 38.2 | / | / | EG: Yam-Epimedium (30 g Rhizoma Dioscoreae and 12 g Herba Epimedii) CG: placebo | 3 months | FEV1 (% of predicted) 、6MWD、mMRC、SGRQ |
6MWD = 6-minute walk distance, FEV1 = Forced expiratory volume in 1 s, FVC: Forced vital capacity, CAT = COPD assessment test, mMRC = modified Medical Research Council, SGRQ = St. George’s Respiratory Questionnaire, EAA = Essential amino acid, EPA = Eicosapentaenoic acid, DHA = docosahexaenoic acid, CHO = Carbohydrate, MLT = Melatonin, VD = Vitamin D, VC = Vitamin C, VE = Vitamin E, OPC = Oligomeric proanthocyanidin, AKL1 = a patented botanical formulation containing extracts of Picrorhiza kurroa, Ginkgo biloba, and Zingiber officinale, EG = experimental group, CG = Control group. “ / ” means not reported
Risk of bias assessment
The results of the RoB analysis show that, 13 studies provided a thorough description of their randomization methods and were classified as having a low risk of bias, while the risk of bias in the remaining studies was unclear. Five studies exhibited elevated risks related to allocation concealment, investigator and participant blinding, and outcome assessment blinding. Additionally, 2 studies had incomplete outcome data.The assessment of the risk of bias for the included studies is illustrated in Figs. 2 and 3.
Fig. 2.
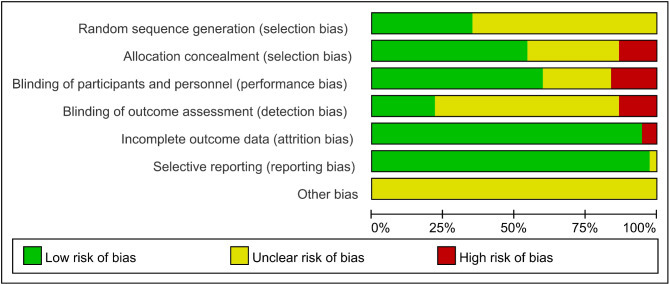
Risk of bias graph
Fig. 3.
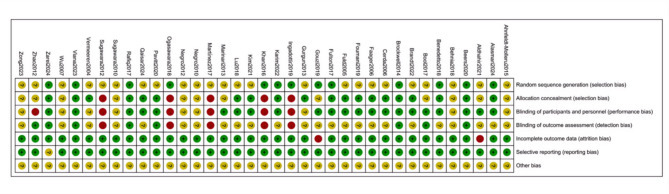
Risk of bias summary
GRADE evaluation on quality of evidence
According to the GRADE framework, the evidence quality for the outcomes of FEV1 (% of predicted) and FEV1/FVC was rated as “moderate,” while for the remaining four outcomes, the evidence quality was rated as “low.”
Results of the network Meta-AnalysisFEV1 (% of predicted)
Sixteen articles [20–23, 34, 35, 37, 42, 44, 46, 48, 51, 53, 60–62] addressed FEV1 (% of predicted). The network diagram (Fig. 4) demonstrated direct comparisons without forming a closed loop. The forest plot (Figure S1) showed that, compared to placebo, butyrate [MD = 2.7, 95% CrI (1.0, 4.4)], Nanocurcumin [MD = 13, 95% CrI (5.4, 21)], and probiotics [MD = 7.1, 95% CrI (5.2, 9.1)] significantly improved FEV1 (% of predicted) in COPD patients (p < 0.05). Indirect comparisons indicated that Nanocurcumin was superior to butyrate, fish oil, complex (Three or more ingredients or unspecified), Coenzyme Q10 combined with Creatine, AKL1 (a patented botanical formulation containing extracts of Picrorhiza kurroa, Ginkgo biloba, and Zingiber officinale), oligomeric proanthocyanidin (OPC), VD, whey proteins (p < 0.05); probiotics were superior to butyrate, coenzyme Q10 combined with creatine, complex, fish oil, OPC, whey proteins, and VD (p < 0.05); Butyrate was superior to whey proteins and VD (p < 0.05), and other comparisons did not reach statistical significance (Table S2). The cumulative ranking showed Nanocurcumin with the highest area (93.0%), followed by probiotics (83.2%), butyrate (65.5%), and OPC as the worst (21.5%) (Fig. 5; Table 2).
Fig. 4.
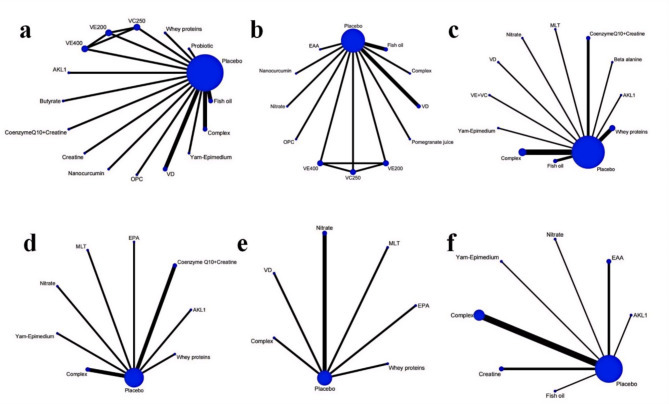
Network plots of the included nutritional supplements and their connections based on the efficacy outcomes. The size of the nodes corresponds to the number of studies involving that particular intervention. The thickness of the edges represents the number of studies comparing the two interventions. Panels (a–f): Each panel represents a specific outcome or analysis based on the included studies: a. Forced Expiratory Volume in One Second (FEV1) (% of predicted). b. The ratio of Forced Expiratory Volume in One Second to Forced Vital Capacity (FEV1/FVC). c. 6-minute walk distance (6MWD); d. modified Medical Research Council (mMRC) Dyspnea Scale. e. COPD Assessment Test (CAT). f. St. George’s Respiratory Questionnaire (SGRQ). EAA = Essential amino acid, EPA = Eicosapentaenoic acid, MLT = Melatonin, VD = Vitamin D, VC = Vitamin C, VE = Vitamin E, OPC = Oligomeric proanthocyanidin, AKL1 = a patented botanical formulation containing extracts of Picrorhiza kurroa, Ginkgo biloba, and Zingiber officinale
Fig. 5.
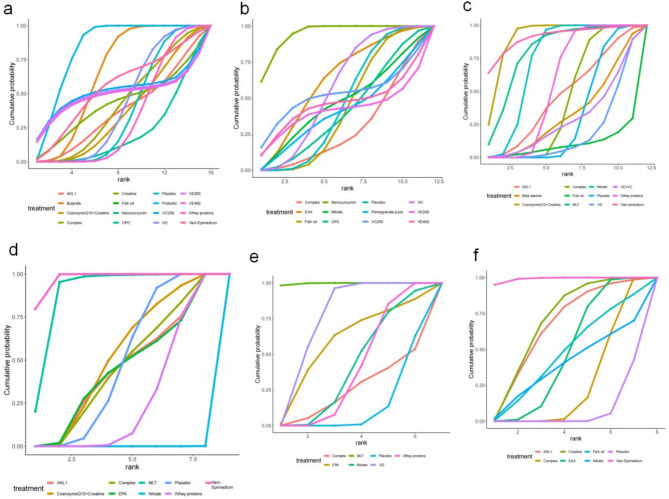
The cumulative sorting curve. Panels (a-f): Each panel represents the probability ranking of interventions for specific outcomes: a. Forced Expiratory Volume in One Second (FEV1) (% of predicted); b. The ratio of Forced Expiratory Volume in One Second to Forced Vital Capacity (FEV1/FVC); c. 6-minute walk distance (6MWD); d. modified Medical Research Council (mMRC) Dyspnea Scale; e. COPD Assessment Test (CAT); f. St. George’s Respiratory Questionnaire (SGRQ). EAA = Essential amino acid, EPA = Eicosapentaenoic acid, MLT = Melatonin, VD = Vitamin D, VC = Vitamin C, VE = Vitamin E, OPC = Oligomeric proanthocyanidin, AKL1 = a patented botanical formulation containing extracts of Picrorhiza kurroa, Ginkgo biloba, and Zingiber officinale
Table 2.
Efficacy ranking of nutritional supplements in COPD treatment
TreatmentFEV1% pred (%)FEV1/FVC (%)6MWD(%)mMRC(%)CAT(%)SGRQ(%)
| Yam-Epimedium | 54.8 | — | 91.0 | 97.4 | — | 99.1 |
| coenzyme Q10 + creatine | 37.0 | — | 90.8 | 52.2 | — | — |
| beta alanine | — | — | 32.6 | — | — | — |
| complex | 42.5 | 28.9 | 42.8 | 45.9 | — | 25.7 |
| fish oil | 57.0 | 42.8 | 9.4 | — | — | 46.8 |
| Nanocurcumin | 93.0 | 94.5 | — | — | — | — |
| MLT | — | — | 80.4 | 89.0 | 99.6 | — |
| nitrate | — | 46.7 | 72.8 | 0.00 | 40.5 | 38.8 |
| placebo | 37.5 | 45.9 | 31.5 | 48.3 | 12.8 | 6.9 |
| pomegranate juice | — | 41.2 | — | — | — | — |
| whey proteins | 33.1 | — | 55.4 | 26.9 | 39.2 | — |
| butyrate | 65.5 | — | — | — | — | — |
| creatine | 42.8 | — | — | — | — | 69.4 |
| EAA | — | 62.2 | — | — | — | 47.2 |
| probiotic | 83.2 | — | — | — | — | — |
| AKL1 | 37.4 | — | 46.0 | 45.0 | — | 65.7 |
| V E + V C | — | — | 28.6 | — | — | — |
| VD | 42.5 | 57.0 | 18.1 | — | 75.2 | — |
| OPC | 21.5 | 40.8 | — | — | — | — |
| VC 250 mg | 52.1 | 52.2 | — | — | — | — |
| V E 200 mg | 50.4 | 41.3 | — | — | — | — |
| VE 400 mg | 49.1 | 45.8 | — | — | — | — |
| EPA | — | — | — | 44.8 | 57.9 | — |
FEV1 = Forced expiratory volume in 1 s, FVC = Forced vital capacity, 6MWD = 6-minute walk distance,
CAT = COPD assessment test, mMRC = modified Medical Research Council, SGRQ = St. George’s Respiratory Questionnaire, coenzyme Q10 + creatine = coenzyme Q10 combined with creatine, MLT = Melatonin, AKL1 = a patented botanical formulation containing extracts of Picrorhiza kurroa, Ginkgo biloba, and Zingiber officinale. “—” means not applicable while. EAA = Essential amino acid, VD = Vitamin D, VC = Vitamin C, VE = Vitamin E, EPA = Eicosapentaenoic acid
FEV1/FVC
Eleven articles [21, 22, 34, 37, 38, 41, 46, 48, 52, 56, 61] investigated FEV1/FVC. The network diagram (Fig. 4) revealed direct comparisons without forming a closed loop. The forest plot (Figure S2) illustrated that compared with placebo, Nanocurcumin [MD = 12, 95% CrI (5.5, 18)] significantly improved FEV1/FVC in COPD patients (p < 0.05). Indirect comparison of different nutritional supplements showed that Nanocurcumin was superior to essential amino acid (EAA), nitrate, fish oil, pomegranate juice, OPC, VD, complex (p < 0.05), and other comparisons did not reach statistical significance (Table S3). The cumulative ranking was highest for Nanocurcumin (94.5%), followed by EAA (62.2%), VD (57%), and lowest for complex (28.9%) (Fig. 5; Table 2).
6MWD
Eighteen articles [19, 22, 23, 34–36, 39, 42, 43, 46, 48, 50, 56–59, 62] examined on 6MWD. The network diagram (Fig. 4) confirmed direct comparisons and without forming a closed loop. The forest plot (Figure S3) supported that, compared with placebo, coenzyme Q10 combined with creatine [MD = 63, 95% CrI (36, 90)], MLT [MD = 46, 95% CrI (1.3, 92)], nitrate [MD = 30, 95% CrI (19, 41)], whey proteins [MD = 11, 95% CrI (9.2, 13)] significantly improved 6MWD in COPD patients. Indirect comparison of different nutritional supplements showed that coenzyme Q10 combined with creatine was superior to AKL1, VE combined with VC, beta alanine, complex, fish oil, nitrate, VD, whey proteins (p < 0.05); whey proteins were superior to complex (p < 0.05); Yam-Epimedium was superior to fish oil, VD; MLT was superior to VD; nitrate was superior to VD, whey proteins, and the rest of the comparisons were not statistically significant (Table S4). Yam-Epimedium had the largest area under the cumulative ranking (91.0%), coenzyme Q10 combined with creatine was slightly next (90.8%), MLT (80.4%), and fish oil was the worst (9.4%) (Fig. 5; Table 2).
mMRC scores
Ten articles [23, 35, 36, 42, 50, 53, 54, 58, 59, 62] referred to the mMRC. The network diagram (Fig. 4) illustrated direct comparisons, failing to form a closed loop. The forest plot (Figure S4) showed that, compared with placebo, MLT [MD = -0.90, 95% CrI (-1.6, -0.21)], Yam-Epimedium [MD = -1.3, 95% CrI (-1.9, -0.67)] significantly reduced mMRC scores in COPD patients. Yam-Epimedium is superior to AKL1, coenzyme Q10 combined with creatine, complex, EPA, nitrate, whey proteins (p < 0.05); MLT is superior to AKL1, coenzyme Q10 combined with creatine, complex, EPA, whey proteins, nitrate (p < 0.05); whey proteins is superior to nitrate (p < 0.05), and the rest of the comparisons are not statistically significant (Table S5). The cumulative ranking was largest for Yam-Epimedium (97.4%), followed by MLT (89%), coenzyme Q10 combined with creatine (52.2%), and the worst for nitrate (0.0%) (Fig. 5; Table 2).
CAT scores
Seven articles [19, 21, 33, 35, 53, 54, 59] referred to CAT. The network diagram (Fig. 4) indicated that the comparisons were direct, yet no closed loop was established. The forest plot (Figure S5) demonstrated that compared to placebo, MLT [MD = -8, 95% CrI (-12, -4.2)], VD [MD = -2.5, 95% CrI (-3.1, -1.9)], whey proteins [MD = -0.70, 95% CrI (-0.99, -0.41)] significantly reduced CAT scores in patients. Indirect comparison of different nutritional supplements showed that MLT was superior to complex, Eicosapentaenoic acid (EPA), nitrate, VD, and whey proteins (p < 0.05); VD was superior to nitrate, whey proteins (p < 0.05), and the rest of the comparisons were not statistically significant (Table S6). The cumulative ranking was largest for MLT (99.6%), followed by VD (75.2%), EPA (57.9%), and worst for placebo (12.8%) (Fig. 5; Table 2).
SGRQ scores
Thirteen articles [22, 32, 36, 40–42, 44, 45, 47, 49, 51, 52, 62] discussed the SGRQ. The network diagram (Fig. 4) revealed direct comparisons with no closed loop formed. The forest plot (Figure S6) verified that, compared with placebo, AKL1 [MD = -9.2, 95% CrI (-18, -0.41)], complex [MD = -2.3, 95% CrI (-3.3, -1.3)], creatine [MD = -9.6, 95% CrI (-17, -2.8)], EAA [MD = -5.2, 95% CrI (-8.1, -2.3), Yam-Epimedium [MD = -24, 95% CrI (-34, -13)], significantly reduced SGRQ scores in COPD patients. Indirect comparison of different nutritional supplements showed that Yam-Epimedium was superior to AKL1, complex, creatine, EAA, fish oil, and nitrate (p < 0.05); creatine was superior to complex (p < 0.05), and the rest of the comparisons were not statistically significant (Table S7). The cumulative ranking Yam-Epimedium was the largest (99.1%), creatine (69.4%) was the next largest, AKL1 (65.7%), and placebo was the worst (6.9%) (Fig. 5; Table 2).
Publish biased evaluations
By analyzing publication bias for FEV1 (%), FEV1/FVC%, 6MWD, CAT, mMRC, and SGRQ and plotting a funnel plot (Fig. 6), the results suggested that there was a higher likelihood of publication bias in 6MWD, SGRQ, and FEV1/FVC%.
Fig. 6.
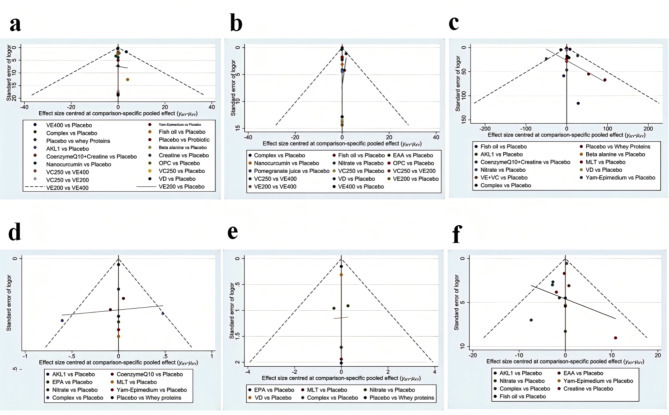
Funnel plots of the outcomes: (a) Forced Expiratory Volume in One Second (FEV1) (% of predicted); (b) The ratio of Forced Expiratory Volume in One Second to Forced Vital Capacity (FEV1/FVC); (c) 6-minute walk distance (6MWD); (d) modified Medical Research Council (mMRC) Dyspnea Scale; (e) COPD Assessment Test (CAT); (f) St. George’s Respiratory Questionnaire (SGRQ). EAA = Essential amino acid, EPA = Eicosapentaenoic acid, MLT = Melatonin, VD = Vitamin D, VC = Vitamin C, VE = Vitamin E, OPC = Oligomeric proanthocyanidin, AKL1 = a patented botanical formulation containing extracts of Picrorhiza kurroa, Ginkgo biloba, and Zingiber officinale
Inconsistency
Regarding inconsistency, most comparisons showed good agreement between direct and indirect evidence, supporting the overall coherence of the network. However, certain comparisons, such as VD versus placebo, complex interventions versus placebo, and coenzyme Q10 combined with creatine versus placebo, exhibited local inconsistency, as illustrated in the node-splitting analyses. These inconsistencies highlight the need for cautious interpretation of effect estimates within those nodes. Detailed local inconsistency results are presented in Supplementary File S3.
Discussion
The importance of nutrition in COPD management has been increasingly recognized in clinical practice. In this study, we assessed the efficacy of 21 nutritional supplements for COPD patients using a network meta-analysis system. Nanocurcumin, butyrate, and probiotics were associated with a slower decline in lung function, with Nanocurcumin demonstrating the most pronounced effect. Coenzyme Q10 combined with creatine was the most effective in enhancing exercise capacity (6MWD). MLT and Yam-Epimedium significantly reduced dyspnea, with the latter being the most promising for symptom relief. Both also contributed notably to improvements in quality of life and overall health status. Our findings may assist healthcare professionals in selecting appropriate supplements to improve patient outcomes. However, for most comparisons, the quality of the evidence was rated as low to moderate. There is still insufficient evidence to determine the optimal nutritional supplements for COPD patients.
Effects of nutritional supplements on lung function
Nanocurcumin, a formulation designed to overcome the limitations of traditional curcumin, emerged as the most significant supplement for improving lung function in COPD patients. Previous studies have underscored Curcumin’s therapeutic potential, particularly due to its antioxidant and anti-inflammatory properties, which can mitigate oxidative stress and slow airway remodeling in COPD [16, 63]. However, curcumin’s low solubility and poor bioavailability limit its clinical application. Nanocurcumin has shown promise in overcoming these barriers by enhancing biological and pharmacological efficacy, making it a more effective option for COPD patients [64, 65]. Despite these promising results, the limited number of available trials necessitates caution in interpreting these findings. Larger, well-designed clinical trials are essential to validate the efficacy of Nanocurcumin in COPD and to compare its advantages with other therapeutic options.
Probiotic supplementation has emerged as a potential therapeutic approach, largely due to its effects on gut-lung communication. Dysregulation of the gut microbiota has been identified as a key factor in the pathophysiology of COPD [66–68]. In our study, probiotics significantly attenuated the decline in lung function, likely through modulation of systemic inflammation via the gut-lung axis. This is consistent with previous research showing that probiotics reduce pro-inflammatory cytokines such as IL-6 and TNF-α while promoting anti-inflammatory mediators like IL-10 [69, 70]. Additionally, probiotics can enhance the production of short-chain fatty acids (SCFAs), which support immune homeostasis in the lungs [71]. However, our findings contrast with studies involving COPD patients with comorbid asthma [72], where probiotics did not yield the same improvements. This discrepancy may be due to the heterogeneity between COPD and asthma, suggesting that further investigations in homogenous COPD populations are needed to confirm the efficacy of probiotics in preserving lung function.
Butyric acid, an anti-inflammatory agent, has demonstrated its potential in reducing airway inflammation and improving lung function [73–75]. A recent study showed that butyric acid reduced inflammation in animal models of asthma and COPD [76]. Our results align with these findings, supporting the therapeutic potential of butyric acid supplementation in mitigating lung function decline. However, clinical trials focused on the specific impact of butyric acid supplementation in COPD patients remain sparse, and further studies are warranted to confirm its effects.
Effect of nutritional supplements on exercise endurance
COPD patients often experience a decline in exercise endurance due to prolonged hypoxia, oxidative damage, mitochondrial dysfunction, and chronic inflammation [77, 78]. Our study showed that coenzyme Q10 combined with creatine supplementation, as well as nitrate supplementation, significantly improved 6MWD in COPD patients. This suggests that these supplements may enhance exercise endurance.
Coenzyme Q10, as an antioxidant, attenuates oxidative stress and inflammation and improves mitochondrial function, thereby enhancing muscle cell energy production, reducing exercise fatigue and delaying muscle damage [79–81]. Creatine is widely recognized as a supplement that enhances muscular reserve and exercise endurance, and a large number of studies have been conducted to support its effects [82–84]. The combination of the two may have a synergistic effect and significantly enhance exercise capacity in COPD patients.
Nitrate supplementation also showed a significant improvement in exercise endurance, as evidenced by the increased 6MWD. However, a 2022 systematic review [85] did not report a significant improvement from nitrate supplementation in COPD patients. This discrepancy may be attributed to differences in the methods used to assess exercise endurance. Our study used 6MWD as the primary measure, while the systematic review utilized a different assessment method, which could have led to varying results. Additionally, patient heterogeneity, including COPD subtypes and disease severity, may influence the response to nitrate supplementation. It has been suggested that nitrate exerts its effects by converting into nitric oxide (NO), which improves blood flow and pulmonary circulation, potentially alleviating dyspnea, and thereby indirectly enhancing exercise endurance in COPD patients [85, 86].While our findings provide robust evidence for the efficacy of nitrate supplementation, further studies are needed to validate its impact across different COPD subtypes and disease stages.
Effects of nutritional supplements on dyspnea and quality of life
Relief of dyspnea and improvement in quality of life are key therapeutic goals in COPD management [87]. Our study demonstrated that both MLT and Yam-Epimedium supplementation were particularly effective in achieving these objectives. Both supplements significantly reduced patients’ mMRC scores, indicating an improvement in dyspnea. The ranked probability plots suggested that Yam-Epimedium was the most likely supplement to alleviate dyspnea symptoms. Regarding quality of life, as measured by the CAT and SGRQ scores, MLT exhibited the most significant effect, while Yam-Epimedium had the most pronounced impact on the SGRQ scores.
Yam-Epimedium’s effect may be attributed to the synergistic action of its components, particularly Icariin, which has antioxidant, anti-inflammatory, immunomodulatory, antitussive, and expectorant properties [88–90]. Icariin may help alleviate COPD symptoms by reducing airway inflammation and inhibiting bronchoconstriction. Additionally, yam contains polysaccharides with antioxidant properties, which may further enhance the anti-inflammatory effect [91]. While the potential of Yam-Epimedium in COPD treatment has not been extensively studied, our findings provide new evidence supporting its use in managing dyspnea and improving quality of life in COPD patients.
MLT, known for its role in regulating circadian rhythms [92, 93], has garnered attention for its potential in managing chronic diseases [94–96]. In this study, MLT significantly reduced mMRC and CAT scores, indicating its effectiveness in alleviating dyspnea and improving quality of life in COPD patients. MLT’s antioxidant and anti-inflammatory properties likely play a key role in reducing airway inflammation [97]. Moreover, its ability to improve sleep quality may contribute to the observed improvements in quality of life. Despite some studies on MLT’s efficacy in chronic diseases, research on its effects in COPD remains limited, and our study offers a promising direction for exploring MLT as a therapeutic option for COPD patients. However, it is important to note that the included trial evaluating MLT also incorporated physical exercise as part of the intervention. Given the well-established benefits of exercise in COPD management, the observed improvements may reflect a combined effect rather than that of MLT alone. Therefore, the contribution of MLT should be interpreted with caution.
Strengths and limitations
This study provides valuable insights into the use of nutritional supplements in COPD patients, but several limitations should be considered. First, the small sample sizes in most of the included studies may have introduced potential bias in the effect estimates. Second, significant heterogeneity in study methodologies, such as differences in intervention duration, dosage, and participant characteristics, may limit the generalizability of the findings. Third, many supplements were represented by only a single study, preventing meaningful sensitivity analyses. As a result, the findings for these supplements should be interpreted with caution. Additionally, potential publication bias was observed in outcomes like 6MWD, SGRQ, and FEV1/FVC%, as indicated by funnel plot asymmetry. This suggests that studies with negative or non-significant results may have been underreported, possibly leading to an overestimation of treatment effects. Consequently, the robustness of our findings may be compromised, and the evidence should be interpreted with caution. Future studies should aim for larger, standardized trials and efforts to reduce publication bias. Finally, adverse effects were not consistently reported in the included studies, which is another limitation. Future research should prioritize the reporting of safety data associated with nutritional supplements in COPD patients.
Conclusion
It was found that different nutritional supplements had varying effects on improving lung function, exercise tolerance, symptoms, and quality of life in patients with COPD. Nanocurcumin may be the best choice for improving lung function; Coenzyme Q10 combined with creatine may be the most effective for enhancing exercise endurance; Additionally, Yam-Epimedium is the most likely supplements to reduce the symptoms of dyspnea in COPD patients. Yam-Epimedium and MLT may be the most effective for improving quality of life and overall health status. Despite these promising findings, further research with larger, more rigorous trials is needed to validate these results and assess the long-term benefits and safety of these supplements.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12890_2025_3667_MOESM1_ESM.docx (32.2KB, docx)
Supplementary Material 1: PRISMA NMA checklist.
12890_2025_3667_MOESM2_ESM.docx (6.4MB, docx)
Supplementary Material 2: Table S1. Supplemental Material1 Search strategy. Table S2. League Table of FEV1%. Table S3. League Table of FEV1/FVC. Table S4. League Table of 6MWD. Table S5. League Table of mMRC. Table S6. League Table of CAT. Table S7. League Table of SGRQ. Figure S1 FEV1% forest plot. Figure S2 FEV1/FVC forest plot. Figure S3 6MWD forest plot. Figure S4 mMRC forest plot. Figure S5 CAT forest plot. Figure S6 SGRQ forest plot.
12890_2025_3667_MOESM3_ESM.docx (7.3MB, docx)
Supplementary Material 3: Local Inconsistency.
Acknowledgements
None.
Author contributions
ZJ and CJ conducted the literature retrieval and data extraction and wrote the main manuscript text. LDZ supervised the research process and provided guidance on the methodology and conceptualization of the study. FST assisted with the data extraction and validation. CJ contributed to the revision of the manuscript. All authors reviewed and approved the final manuscript.
Funding
None.
Data availability
The data supporting the findings of this network meta-analysis are available within the article and its supplementary materials. Additional information, including analysis scripts or raw data used in the synthesis, is available from the corresponding author upon reasonable request.
DeclarationsEthics approval and consent to participate
Not applicable.
Consent for publicatiosn
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Jia Zeng and Jing Cheng contributed equally to this work.