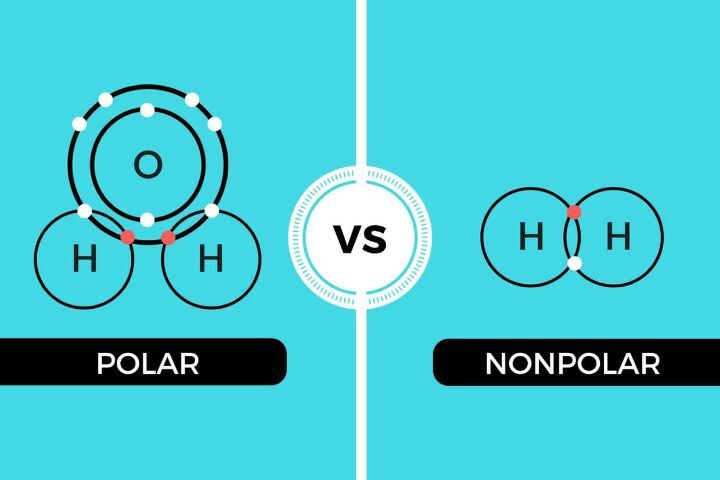
Definition of nonpolar
: not polar
especially : consisting of molecules not having a dipole
a nonpolar solvent
Examples of nonpolar in a Sentence
The reason is that ethanol is a polar molecule, while most other gasoline components are nonpolar.
— Robert Rapier, Forbes, 4 July 2021
This is, after all, the driest nonpolar place on Earth.
— Mark Johanson, chicagotribune.com, 8 Oct. 2019
nonpolar
adjective PHYSICS specialized
US /nɑːnˈpoʊ·lər/
used to refer to a substance whose molecules do not have opposite positive and negative poles
nonpolar
adjective [ not gradable ]
US /nɑnˈpoʊ·lər/
CHEMISTRY
describes a substance whose molecules do not have opposite positive and negative poles
How to Tell If Something Is Polar or Non-Polar
Updated March 13, 2018
By Kylene Arnold
Polarity describes the tendency of a substance to have a molecular dipole, or a positively and a negatively charged end. Polar molecules are made of elements with different electronegativities, or electron attractions, meaning that one element possesses the shared electrons more often than the other. This gives the more electronegative element a partially negative charge and the more electropositive element a partially positive charge. If these elements are arranged symmetrically, so that these charges cancel one another, the molecule is non-polar. If they are arranged asymmetrically, however, they form a polar molecule.
Stereochemical Method
Examining a diagram of the spatial arrangement of atoms that form a molecule can tell you if it is polar or non-polar. A molecule has polar bonds if there is a significant difference in electronegativity between the two elements. If the electronegativities of both elements are very similar or the same, the bonds are non-polar. If this is the case, the entire molecule is also non-polar. If it has polar bonds, you must examine the molecule further to determine whether it is polar or not.
Begin by drawing a Lewis diagram of the molecule. In this kind of diagram, the molecule's constituent elements are represented by their chemical symbols surrounded by dots representing their outer electrons. When properly drawn, Lewis diagrams show the number of bonds and lone pairs, or unbonded pairs of electrons, present in the molecule.
Examine the shape of the molecule, including the number of bonds and lone pairs around the central atom. For instance, two bonds and two lone pairs create a bent molecule. Four bonds and no lone pairs create a tetrahedral shape. You may need to refer to a molecular geometry chart if you are uncertain about the shape of your molecule.
Draw a shape diagram showing how the elements are spatially arranged. If the bonds are symmetrical, their polarities cancel each other and the molecule is non-polar. If the bonds are asymmetrical, so that the more electronegative element is on one end and the electropositive element is on the other, the molecule is polar.
Solution Method
Mixing a liquid of unknown polarity with water can tell you if the molecules in the liquid are polar or non-polar. Simply mix the liquid with an equal part of water and allow the mixture to sit undisturbed. Examine the mixture after the liquids have sat together for a time. If they have not separated, but have formed a solution, the unknown liquid is polar. If there is a clear boundary between the two liquids, it is non-polar. For instance, oil, a non-polar molecule, always separates out of a water-based solution. However, vinegar, a polar substance, does not.
Did you find this page helpful?
👍
👎
Related Articles
Differences Between Polar & Nonpolar in Chemistry
How to Know If a Compound Is Polar or Non-Polar?
How to Find Out If an Element Is an Ion
Three Ways That Polarity of Water Molecules Affect...
How to Explain Polarity
What Happens to Ionic & Covalent Compounds When They...
How to Find the Number of Ions in a Compound
How to Tell if an Atom Is Polar or Non-polar?
How to Determine if a Molecule Is Planar
How to Determine Polarity in Chemistry
Defining Characteristics of Lipid Molecules
Are Ions Hydrophobic Or Hydrophilic?
What Are True Statements About a Compound?
Characteristics of Ionic and Covalent Compounds
What Happens When a Substance Dissolves in Water?
Does Bonding Exist in Substances That Consist of Discrete...
How to Find Out If a Compound Is a Strong Electrolyte
Understanding Chemical Formulas
How to Figure Out the Ionic Percentage Once You Get...
REFERENCES
ABOUT THE AUTHOR
Differences Between Polar & Nonpolar in Chemistry

•••
Updated February 10, 2020
By Riti Gupta
Reviewed by: Lana Bandoim, B.S.
Electronegativity determines how much an atom wants electrons. The more electronegative an atom, the more it wants electrons. This is important to keep in mind when looking at the different kinds of bonds.
If one atom is much more electronegative than another, then it can either completely take an electron from the other atom (ionic bond), or it can simply pull the electrons toward itself more (polar covalent bond). As a result, covalent bonds that contain atoms with very high electronegativities (like oxygen or fluorine) are polar. The oxygen or fluorine hog the electrons.
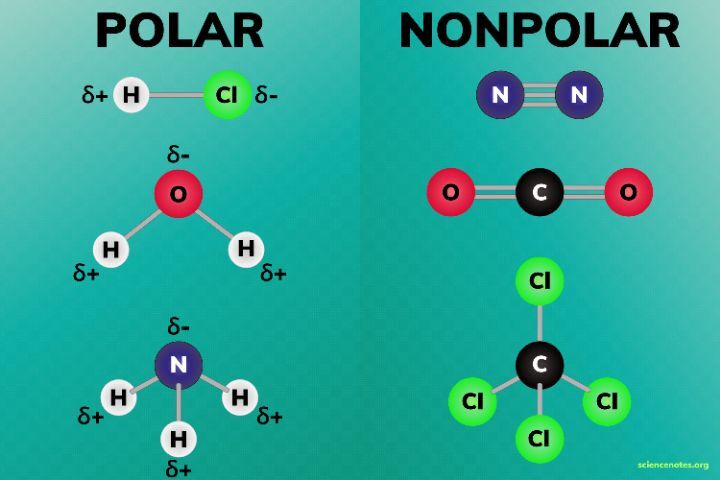
This is the basis for the difference between polar and nonpolar bonds. The unequal sharing of electrons results in the bond having a partially positive end and a partially negative end. The more electronegative atom is partially negative (denoted δ-) while the other end is partially positive (denoted δ+).
Classifying Chemical Bonds
Bonds can either be completely nonpolar or completely polar. A completely polar bond occurs when one of the atoms is so electronegative that it takes an electron from the other atom (this is called an ionic bond).
On the other hand, when the electronegativities are exactly the same the bond is considered to be a nonpolar covalent bond. The two atoms completely share electrons.
But what happens in between these two extremes?
Here is a table that demonstrates what kind of bond is likely forming based on the difference in electronegativity:
Determining Bond Type Based on Difference in ElectronegativityBond TypeElectronegative Difference
Pure Covalent
< 0.4
Polar Covalent
between 0.4 and 1.8
Ionic
> 1.8
https://chem.libretexts.org/Courses/Oregon_Institute_of_Technology/OIT%3A_CHE_202_-_General_Chemistry_II/Unit_6%3A_Molecular_Polarity/6.1%3A_Electronegativity_and_Polarity
Thus, the difference between polar and nonpolar bonds is due to the electronegativity difference of the atoms.
Polar vs. Nonpolar
A compound can have polar covalent bonds and yet still not be a polar compound. Why is that?
Polar compounds have a net dipole as a result of polar bonds that are arranged asymmetrically. This means that they have both a partial positive and partial positive charge that do not cancel out. An example of this is water.
Nonpolar compounds can either entirely share their electrons, or they can have symmetrical polar bonds that end up canceling out any sort of net dipole. An example of this is BF3. Because the polar bonds are arranged in a single plane they end up canceling out.
Why Does Polarity Matter?
Chemical polarity plays a huge role in how different molecules interact. For example, why does sugar dissolve in water while oil does not?
It's all about polar vs. nonpolar.
Water is a polar solvent. The oxygen atom contains two lone pairs and is more electronegative than hydrogen, thus pulling the electrons towards itself. As a result the oxygen atom has a partial negative charge associated with it. The hydrogens on the other hand are essentially protons and have a partial positive charge associated with them.
Sugar is also polar! It has many hydroxyl (OH) groups that readily make hydrogen bonds. Sugar thus has both partial positive and negative charges associated with it. As a result, there are hydrogen bond donors and acceptors in both water and in sugar. For this reason, sugar will dissolve in water.
On the other hand, something like oil is made up of primarily of C-H bonds. As discussed above, a C-H bond is not polar because the electronegativity between the two atoms in the bond is not that different. This means that overall, oil doesn't really have any sort of partial positive or negative charge. This lack of partial charges means that the oil molecule will not be able to hydrogen bond. Since water likes to hydrogen bond and stay with polar molecules, the water will not dissolve the oil.
Taking a look at the structure of the compound and the nature of the bonds it contains will tell you a lot about whether or not the molecule can have partial positive or partial negative charge. If it can, then it is likely polar. It if does not, then it is nonpolar.
Related Articles
How to Tell if an Atom Is Polar or Non-polar?
How to Tell If Something Is Polar or Non-Polar
What is a Polar Molecule?
How to Tell If Something Is Polar or Non-Polar
How to Explain Polarity
Are Ions Hydrophobic Or Hydrophilic?
Molecular Activity of Water Vs. Oil
What Dissolves Oil?
Characteristics of Ionic and Covalent Compounds
Defining Characteristics of Lipid Molecules
Why Oil Won't Mix in Water?
Examples of Immiscible Liquids
Characteristics of Hydrogen Bonding
Why Are Lipids Insoluble in Water?
How Does Alcohol Dissolve Oil?
How to Calculate a Fraction Covalent
Is Methane Nonpolar?
How to Find Out If an Element Is an Ion
How to Figure Out the Ionic Percentage Once You Get...
REFERENCES
ABOUT THE AUTHOR
How to Know If a Compound Is Polar or Non-Polar?

•••
Updated March 30, 2020
By Riti Gupta
Reviewed by: Lana Bandoim, B.S.
Before determining if a compound is polar, you need to determine whether or not the bonds in that compound are polar. You also have to determine the molecular geometry of the bonds and any electron lone pairs.
Before talking about whether or not an entire compound is polar, take a look at what determines whether or not a bond is polar. You can then apply these rules to determine if each molecule is polar or nonpolar.
What Makes a Bond Polar?
A molecule is polar if one part of it has a partial positive charge, and the other part has a partial negative charge.
When in a bond, atoms can either share electrons (covalent) or give them up (ionic). The atom that holds the electrons closer will thus be more negatively charged than the other atom.
Electronegativity is a measure of how much a particular element wants electrons. In the Resources section you will find a periodic table which reports the electronegativity of each element. The higher this number, the more an atom of that element will "hog" the electrons in a bond. For example, fluorine is the most electronegative element.
Electronegativity values can help you determine what kind of bond exists between two atoms. Is the bond likely to be ionic or covalent? To do this, find the absolute value of the difference between the electronegativities of the two atoms. Based on this value, the following table tells you if the bond is a polar covalent bond, covalent bond or ionic bond.
Bond Type
Electronegativity Difference
pure covalent
<0.4
polar covalent
between 0.4 and 1.8
ionic
>1.8
Think about water. What is the electronegativity difference between atoms in water? The electronegativity difference between H (2.2) and O (3.44) is 1.24. As such, the bond is polar covalent.
Bond Polarity and Molecule Polarity
As you saw above, a bond within a molecule can be polar. What does this mean for the whole molecule?
When determining molecule polarity, all bonds must be considered. This means that the vector partial charge from each bond must be added up. If they a cancel out, then the molecule may not be polar. If there are vector components left, then the bond is polar.
In order to find the direction of these vectors, you have to examine the molecular geometry of the bonds. You can find this via valence shell electron-pair repulsion (VSEPR) theory.
The theory starts with the idea that electron pairs in the valence shell of an atom repel each other (since like charges repel). As a result, the electron pairs around an atom will orient themselves to minimize repulsive forces.
Take a look at water again. Water is bound to two hydrogens and also has two lone pair electrons. It has a tetrahedral bent shape.
To determine whether or not the molecule is polar, you have to look at the partial charge vectors on the two bonds in the molecule.
First, there are two electron pairs on the molecule, which means there will be a large negative partial charge vector in that direction.
Next, oxygen is more electronegative than hydrogen and will hog the electrons. This means that the partial charge vector on each bond will have a negative component pointing toward the oxygen.
The inward component of the vector on each bond will cancel. The portion pointing toward the oxygen will not cancel. As a result, there is a net partial negative charge toward the oxygen side of the molecule. There is also a net partial position toward the hydrogen side of the molecule.
This analysis reveals that water is a polar molecule.
What about CH4?
First, CH4 has no lone pairs since all the electrons are involved in a single bond between C and H. CH4 has a tetrahedral molecular geometry.
Next, the C-H bond is covalent as the difference in electronegativities is 0.35. All the bonds are covalent, and there will not be a big dipole moment. Thus, CH4 is a nonpolar molecule.
The difference between polar and nonpolar molecules can thus be found by the vectors of partial charge resulting from each bond.